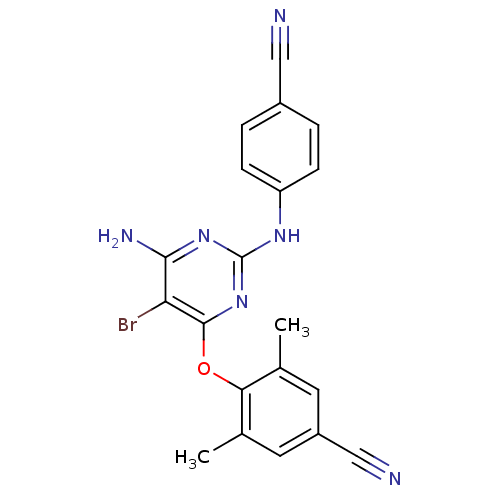
BDBM50103642 4-(6-amino-5-bromo-2-(4-cyanophenylamino)pyrimidin-4-ylamino)-3,5-dimethylbenzonitrile::4-(6-amino-5-bromo-2-(4-cyanophenylamino)pyrimidin-4-yloxy)-3,5-dimethylbenzonitrile::4-({6-AMINO-5-BROMO-2-[(4-CYANOPHENYL)AMINO]PYRIMIDIN-4-YL}OXY)-3,5-DIMETHYLBENZONITRILE::4-[6-amino-5-bromo-2-(4-cyano-phenylamino)-pyrimidin-4-yloxy]-3,5-dimethyl-benzonitrile::CHEMBL308954::ETRAVIRINE::Etravirine (ETR)::Etravirine (ETV)::Etravirine (TMC125)::TMC 125::TMC-125
SMILES Cc1cc(cc(c1Oc2c(c(nc(n2)Nc3ccc(cc3)C#N)N)Br)C)C#N
InChI Key InChIKey=PYGWGZALEOIKDF-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 153 hits for monomerid = 50103642
Found 153 hits for monomerid = 50103642
TargetReverse transcriptase(Human immunodeficiency virus type 1)
Merck Research Laboratories
Curated by ChEMBL
Merck Research Laboratories
Curated by ChEMBL
Affinity DataIC50: 0.600nMAssay Description:Inhibition of HIV1 isolate R8 reverse transcriptase after 90 minsMore data for this Ligand-Target Pair
TargetReverse transcriptase(Human immunodeficiency virus type 1)
Merck Research Laboratories
Curated by ChEMBL
Merck Research Laboratories
Curated by ChEMBL
Affinity DataIC50: 1.20nMAssay Description:Inhibition of HIV1 reverse transcriptase K103N mutant infected in human MT4 cells assessed as protection against virus-induced cytopathic effect by M...More data for this Ligand-Target Pair
TargetReverse transcriptase protein(Human immunodeficiency virus type 1)
Merck Research Laboratories
Curated by ChEMBL
Merck Research Laboratories
Curated by ChEMBL
Affinity DataIC50: 1.30nMAssay Description:Inhibition of HIV1 isolate R8 reverse transcriptase K103N mutant after 90 mins by electrochemiluminescence analysisMore data for this Ligand-Target Pair
TargetReverse transcriptase(Human immunodeficiency virus type 1)
Merck Research Laboratories
Curated by ChEMBL
Merck Research Laboratories
Curated by ChEMBL
Affinity DataIC50: 1.40nMAssay Description:Inhibition of HIV1 LAI reverse transcriptase infected in human MT4 cells assessed as protection against virus-induced cytopathic effect by MTT assayMore data for this Ligand-Target Pair
TargetReverse transcriptase(Human immunodeficiency virus type 1)
Merck Research Laboratories
Curated by ChEMBL
Merck Research Laboratories
Curated by ChEMBL
Affinity DataIC50: 2nMAssay Description:Inhibition of HIV1 isolate R8 reverse transcriptase Y181C mutant after 90 mins by electrochemiluminescence analysisMore data for this Ligand-Target Pair
TargetReverse transcriptase(Human immunodeficiency virus type 1)
Merck Research Laboratories
Curated by ChEMBL
Merck Research Laboratories
Curated by ChEMBL
Affinity DataEC50: 2nMAssay Description:Inhibition of HIV-1 3B reverse transcriptase K103N mutant infected in human MT4 cells assessed as inhibition of viral replication after 5 days by cel...More data for this Ligand-Target Pair
TargetReverse transcriptase(Human immunodeficiency virus type 1)
Merck Research Laboratories
Curated by ChEMBL
Merck Research Laboratories
Curated by ChEMBL
Affinity DataEC50: 2.30nMAssay Description:Inhibition of reverse transcriptase K103N mutant in HIV-1 3B infected in human MT4 cells assessed as reduction in virus-induced cytopathic effect inc...More data for this Ligand-Target Pair
TargetReverse transcriptase(Human immunodeficiency virus type 1)
Merck Research Laboratories
Curated by ChEMBL
Merck Research Laboratories
Curated by ChEMBL
Affinity DataEC50: 3nMAssay Description:Inhibition of reverse transcriptase K103N mutant in HIV-1 infected in human MT4 cells assessed as reduction in virus-induced cytopathic effect incuba...More data for this Ligand-Target Pair
TargetReverse transcriptase(Human immunodeficiency virus type 1)
Merck Research Laboratories
Curated by ChEMBL
Merck Research Laboratories
Curated by ChEMBL
Affinity DataEC50: 3nMAssay Description:Inhibition of wild-type HIV1 3B reverse transcriptase infected in human MT4 cells assessed as protection against virus-induced cytopathic effect meas...More data for this Ligand-Target Pair
TargetReverse transcriptase(Human immunodeficiency virus type 1)
Merck Research Laboratories
Curated by ChEMBL
Merck Research Laboratories
Curated by ChEMBL
Affinity DataIC50: 3.30nMAssay Description:Inhibition of HIV1 reverse transcriptase L100I mutant infected in human MT4 cells assessed as protection against virus-induced cytopathic effect by M...More data for this Ligand-Target Pair
TargetReverse transcriptase/RNaseH(Human immunodeficiency virus type 1)
Fudan University
Curated by ChEMBL
Fudan University
Curated by ChEMBL
Affinity DataEC50: 3.40nMAssay Description:Inhibition of HIV-1 p66/p51 reverse transcriptase K103N mutant incubated for 40 mins by picogreen dye-based spectrofluorometric analysisMore data for this Ligand-Target Pair
TargetReverse transcriptase(Human immunodeficiency virus type 1)
Merck Research Laboratories
Curated by ChEMBL
Merck Research Laboratories
Curated by ChEMBL
Affinity DataEC50: 4nMAssay Description:Inhibition of wild-type HIV1 3B reverse transcriptase infected in human MT4 cells assessed as protection against virus-induced cytotoxicity by MTT as...More data for this Ligand-Target Pair
TargetReverse transcriptase(Human immunodeficiency virus type 1)
Merck Research Laboratories
Curated by ChEMBL
Merck Research Laboratories
Curated by ChEMBL
Affinity DataIC50: 4.30nMAssay Description:Inhibition of HIV1 reverse transcriptase K103N/Y188C double mutant infected in human MT4 cells assessed as protection against virus-induced cytopathi...More data for this Ligand-Target Pair
TargetReverse transcriptase(Human immunodeficiency virus type 1)
Merck Research Laboratories
Curated by ChEMBL
Merck Research Laboratories
Curated by ChEMBL
Affinity DataIC50: 4.60nMAssay Description:Inhibition of HIV1 reverse transcriptase Y188L mutant infected in human MT4 cells assessed as protection against virus-induced cytopathic effect by M...More data for this Ligand-Target Pair
TargetReverse transcriptase protein(Human immunodeficiency virus type 1)
Merck Research Laboratories
Curated by ChEMBL
Merck Research Laboratories
Curated by ChEMBL
Affinity DataEC50: 5nMAssay Description:Inhibition of HIV RT K103N/Y181C mutant by cell based assayMore data for this Ligand-Target Pair
TargetReverse transcriptase(Human immunodeficiency virus type 1)
Merck Research Laboratories
Curated by ChEMBL
Merck Research Laboratories
Curated by ChEMBL
Affinity DataEC50: 5.40nMAssay Description:Inhibition of HIV1 reverse transcriptase K103N mutant infected in human MT4 cells assessed as protection against virus-induced cytotoxicity by MTT as...More data for this Ligand-Target Pair
TargetReverse transcriptase(Human immunodeficiency virus type 1)
Merck Research Laboratories
Curated by ChEMBL
Merck Research Laboratories
Curated by ChEMBL
Affinity DataIC50: 7nMAssay Description:Inhibition of HIV1 reverse transcriptase Y181C mutant infected in human MT4 cells assessed as protection against virus-induced cytopathic effect by M...More data for this Ligand-Target Pair
TargetReverse transcriptase/RNaseH(Human immunodeficiency virus type 1)
Fudan University
Curated by ChEMBL
Fudan University
Curated by ChEMBL
Affinity DataIC50: 8nMAssay Description:Inhibition of wild type HIV-1 reverse transcriptase assessed as reduction of biotin-dUTP incorporation into protein using ABTS as substrate incubated...More data for this Ligand-Target Pair
TargetReverse transcriptase(Human immunodeficiency virus type 1)
Merck Research Laboratories
Curated by ChEMBL
Merck Research Laboratories
Curated by ChEMBL
Affinity DataEC50: 8nMAssay Description:Inhibition of reverse transcriptase L100I mutant in HIV-1 infected in human MT4 cells assessed as reduction in virus-induced cytopathic effect incuba...More data for this Ligand-Target Pair
TargetReverse transcriptase(Human immunodeficiency virus type 1)
Merck Research Laboratories
Curated by ChEMBL
Merck Research Laboratories
Curated by ChEMBL
Affinity DataIC50: 8nMAssay Description:Inhibition of HIV1 RT V106A/F227L mutant in presence of reconstituted template and viral nucleotides [digoxigenin (DIG)-dUTP, biotin-dUTP and dTTP] i...More data for this Ligand-Target Pair
TargetReverse transcriptase(Human immunodeficiency virus type 1)
Merck Research Laboratories
Curated by ChEMBL
Merck Research Laboratories
Curated by ChEMBL
Affinity DataEC50: 9nMAssay Description:Inhibition of reverse transcriptase E138K mutant in HIV-1 infected in human MT4 cells assessed as reduction in virus-induced cytopathic effect incuba...More data for this Ligand-Target Pair
TargetReverse transcriptase(Human immunodeficiency virus type 1)
Merck Research Laboratories
Curated by ChEMBL
Merck Research Laboratories
Curated by ChEMBL
Affinity DataEC50: 9.20nMAssay Description:Inhibition of reverse transcriptase L100I mutant in HIV-1 3B infected in human MT4 cells assessed as reduction in virus-induced cytopathic effect inc...More data for this Ligand-Target Pair
TargetReverse transcriptase(Human immunodeficiency virus type 1)
Merck Research Laboratories
Curated by ChEMBL
Merck Research Laboratories
Curated by ChEMBL
Affinity DataIC50: 10nMAssay Description:Inhibition of wild type HIV1 reverse transcriptase RNA-dependent DNA polymerase activity using poly(rA)/oligo(dT)10:1 and [3H]-dTTP substrateMore data for this Ligand-Target Pair
TargetReverse transcriptase(Human immunodeficiency virus type 1)
Merck Research Laboratories
Curated by ChEMBL
Merck Research Laboratories
Curated by ChEMBL
Affinity DataIC50: 10nMAssay Description:Inhibition of wild type HIV1 reverse transcriptase assessed as reduction in enzyme activityMore data for this Ligand-Target Pair
TargetReverse transcriptase(Human immunodeficiency virus type 1)
Merck Research Laboratories
Curated by ChEMBL
Merck Research Laboratories
Curated by ChEMBL
Affinity DataIC50: 10nMAssay Description:Inhibition of HIV1 reverse transcriptase F227L/V106A double mutant using oligo(dT)16 as primer measured after 40 mins by picogreen-dye based spectrof...More data for this Ligand-Target Pair
TargetReverse transcriptase/RNaseH(Human immunodeficiency virus type 1)
Fudan University
Curated by ChEMBL
Fudan University
Curated by ChEMBL
Affinity DataIC50: 10nMAssay Description:Inhibition of HIV-1 reverse transcriptase incubated for 1 hrs in presence of biotin-labeled dNTPs by ELISA assayMore data for this Ligand-Target Pair
TargetReverse transcriptase(Human immunodeficiency virus type 1)
Merck Research Laboratories
Curated by ChEMBL
Merck Research Laboratories
Curated by ChEMBL
Affinity DataIC50: 10nMAssay Description:Inhibition of HIV1 reverse transcriptase L100I mutant using oligo(dT)16 as primer measured after 40 mins by picogreen-dye based spectrofluorimetric a...More data for this Ligand-Target Pair
TargetReverse transcriptase(Human immunodeficiency virus type 1)
Merck Research Laboratories
Curated by ChEMBL
Merck Research Laboratories
Curated by ChEMBL
Affinity DataIC50: 10nMAssay Description:Inhibition of HIV1 NL4-3 reverse transcriptase His-tagged p66/p51 associated RNA dependent DNA polymerase activity expressed in Escherichia coli asse...More data for this Ligand-Target Pair
TargetReverse transcriptase(Human immunodeficiency virus type 1)
Merck Research Laboratories
Curated by ChEMBL
Merck Research Laboratories
Curated by ChEMBL
Affinity DataIC50: 10nMAssay Description:Inhibition of wild type HIV1 reverse transcriptase V106A mutant assessed as reduction in enzyme activityMore data for this Ligand-Target Pair
TargetReverse transcriptase(Human immunodeficiency virus type 1)
Merck Research Laboratories
Curated by ChEMBL
Merck Research Laboratories
Curated by ChEMBL
Affinity DataIC50: 10nMAssay Description:Inhibition of HIV1 wild-type reverse transcriptase using [3H]dTTP by scintillation countingMore data for this Ligand-Target Pair
TargetReverse transcriptase protein(Human immunodeficiency virus type 1)
Merck Research Laboratories
Curated by ChEMBL
Merck Research Laboratories
Curated by ChEMBL
Affinity DataIC50: 10nMAssay Description:Inhibition of HIV1 reverse transcriptase L100I mutant RNA-dependent DNA polymerase activity using poly(rA)/oligo(dT)10:1 and [3H]-dTTP substrateMore data for this Ligand-Target Pair
TargetReverse transcriptase/RNaseH(Human immunodeficiency virus type 1)
Fudan University
Curated by ChEMBL
Fudan University
Curated by ChEMBL
Affinity DataIC50: 10nMAssay Description:Inhibition of RNA-dependent DNA polymerase activity of wild type recombinant HIV-1 His-tagged p66/p51 reverse transcriptase assessed as inhibition of...More data for this Ligand-Target Pair
TargetReverse transcriptase/RNaseH(Human immunodeficiency virus type 1)
Fudan University
Curated by ChEMBL
Fudan University
Curated by ChEMBL
Affinity DataIC50: 10nMAssay Description:Inhibition of wild type recombinant HIV-1 reverse transcriptase using biotin-labelled dNTPs as substrate assessed as inhibition of biotin-labelled UT...More data for this Ligand-Target Pair
TargetReverse transcriptase(Human immunodeficiency virus type 1)
Merck Research Laboratories
Curated by ChEMBL
Merck Research Laboratories
Curated by ChEMBL
Affinity DataIC50: 10nMAssay Description:Inhibition of recombinant wild type HIV1 reverse transcriptase p66/p51 assessed as reduction in biotin-dUTP incorporation incubated for 40 mins using...More data for this Ligand-Target Pair
TargetReverse transcriptase(Human immunodeficiency virus type 1)
Merck Research Laboratories
Curated by ChEMBL
Merck Research Laboratories
Curated by ChEMBL
Affinity DataIC50: 10nMAssay Description:Inhibition of HIV1 wild-type reverse transcriptase V106A mutant using [3H]dTTP by scintillation countingMore data for this Ligand-Target Pair
TargetReverse transcriptase(Human immunodeficiency virus type 1)
Merck Research Laboratories
Curated by ChEMBL
Merck Research Laboratories
Curated by ChEMBL
Affinity DataIC50: 10nMAssay Description:Inhibition of HIV1 reverse transcriptase K103N/Y181C double mutant using oligo(dT)16 as primer measured after 40 mins by picogreen-dye based spectrof...More data for this Ligand-Target Pair
TargetReverse transcriptase(Human immunodeficiency virus type 1)
Merck Research Laboratories
Curated by ChEMBL
Merck Research Laboratories
Curated by ChEMBL
Affinity DataIC50: 10nMAssay Description:Inhibition of HIV1 wild type reverse transcriptase p66/p51 using poly(rA) template and measured after 40 mins by picogreen based spectrofluorometryMore data for this Ligand-Target Pair
TargetReverse transcriptase(Human immunodeficiency virus type 1)
Merck Research Laboratories
Curated by ChEMBL
Merck Research Laboratories
Curated by ChEMBL
Affinity DataIC50: 11nMAssay Description:Inhibition of recombinant wild-type HIV1 GST-fused reverse transcriptase p66/p51 RNA-dependent DNA polymerase activity expressed in Escherichia coli ...More data for this Ligand-Target Pair
TargetReverse transcriptase(Human immunodeficiency virus type 1)
Merck Research Laboratories
Curated by ChEMBL
Merck Research Laboratories
Curated by ChEMBL
Affinity DataIC50: 11nMAssay Description:Inhibition of HIV1 reverse transcriptase p66/p51 using poly(rA)/oligo(dT)16 as template/primer measured after 40 mins by pico-green based spectrofluo...More data for this Ligand-Target Pair
TargetReverse transcriptase(Human immunodeficiency virus type 1)
Merck Research Laboratories
Curated by ChEMBL
Merck Research Laboratories
Curated by ChEMBL
Affinity DataIC50: 11nMAssay Description:Inhibition of recombinant HIV-1 reverse transcriptase p66/p51 incubated for 40 mins by picogreen dye based spectrofluorometric assayMore data for this Ligand-Target Pair
TargetReverse transcriptase(Human immunodeficiency virus type 1)
Merck Research Laboratories
Curated by ChEMBL
Merck Research Laboratories
Curated by ChEMBL
Affinity DataIC50: 11nMAssay Description:Inhibition of recombinant wild type HIV1 p66/p51 using poly(rA) template/oligo(dT)16 primer after 40 mins by PicoGreen-based spectrofluorometric meth...More data for this Ligand-Target Pair
TargetReverse transcriptase/RNaseH(Human immunodeficiency virus type 1)
Fudan University
Curated by ChEMBL
Fudan University
Curated by ChEMBL
Affinity DataIC50: 11nMAssay Description:Inhibition of recombinant wild type p66/p51 HIV1 reverse transcriptase incubated for 40 mins by picogreen dye-based spectrofluorometric analysisMore data for this Ligand-Target Pair
TargetReverse transcriptase(Human immunodeficiency virus type 1)
Merck Research Laboratories
Curated by ChEMBL
Merck Research Laboratories
Curated by ChEMBL
Affinity DataIC50: 11nMAssay Description:Inhibition of recombinant HIV1 Reverse transcriptase p66/p51 using poly (rA)-oligo (dT) as template primer after 40 mins by spectrofluorometric analy...More data for this Ligand-Target Pair
TargetReverse transcriptase/RNaseH(Human immunodeficiency virus type 1)
Fudan University
Curated by ChEMBL
Fudan University
Curated by ChEMBL
Affinity DataIC50: 11nMAssay Description:Inhibition of wild type HIV1 p66/p51 reverse transcriptase using poly(rA) as template, oligo(dT)16 as primer and RNA/DNA as substrate measured after ...More data for this Ligand-Target Pair
TargetReverse transcriptase/RNaseH(Human immunodeficiency virus type 1)
Fudan University
Curated by ChEMBL
Fudan University
Curated by ChEMBL
Affinity DataIC50: 11nMAssay Description:Inhibition of HIV1 reverse transcriptase assessed as reduction in biotin-dUTP incorporation into template incubated for 1 hr by ELISAMore data for this Ligand-Target Pair
TargetReverse transcriptase(Human immunodeficiency virus type 1)
Merck Research Laboratories
Curated by ChEMBL
Merck Research Laboratories
Curated by ChEMBL
Affinity DataIC50: 11nMAssay Description:Inhibition of HIV1 reverse transcriptase p66/p51 using poly(rA)/oligo(dT)16 as template/primer measured after 40 mins by pico-green based spectrofluo...More data for this Ligand-Target Pair
TargetReverse transcriptase/RNaseH(Human immunodeficiency virus type 1)
Fudan University
Curated by ChEMBL
Fudan University
Curated by ChEMBL
Affinity DataIC50: 11nMAssay Description:Inhibition of recombinant wild type HIV-1 reverse transcriptase using DNA/RNA as substrate incubated for 40 mins by Pico-green dye based spectrofluor...More data for this Ligand-Target Pair
TargetReverse transcriptase(Human immunodeficiency virus type 1)
Merck Research Laboratories
Curated by ChEMBL
Merck Research Laboratories
Curated by ChEMBL
Affinity DataIC50: 11nMAssay Description:Inhibition of recombinant HIV1 reverse transcriptase using (DIG)-dUTP and biotin-labeled dNTPs as substrate after 1 hr by ELISAMore data for this Ligand-Target Pair
TargetReverse transcriptase/RNaseH(Human immunodeficiency virus type 1)
Fudan University
Curated by ChEMBL
Fudan University
Curated by ChEMBL
Affinity DataEC50: 11nMAssay Description:Inhibition of HIV-1 p66/p51 reverse transcriptase L100I mutant incubated for 40 mins by picogreen dye-based spectrofluorometric analysisMore data for this Ligand-Target Pair
TargetReverse transcriptase(Human immunodeficiency virus type 1)
Merck Research Laboratories
Curated by ChEMBL
Merck Research Laboratories
Curated by ChEMBL
Affinity DataIC50: 11nMAssay Description:Inhibition of recombinant wild type HIV1 reverse transcriptase assessed as decrease in biotin-dUTP incorporation using DIG-labeled dUTP/biotin-labele...More data for this Ligand-Target Pair
Activity Spreadsheet -- ITC Data from BindingDB
 Found 1 hit for monomerid = 50103642
Found 1 hit for monomerid = 50103642
CellReverse transcriptase (HIV-1 RT)(1-350)(Human immunodeficiency virus type 1)
Chulalongkorn University
Chulalongkorn University
ITC DataΔG°: -36.8kcal/mole −TΔS°: 23.0kcal/mole ΔH°: -59.7kcal/mole
T: 25.00°C
T: 25.00°C
 3D Structure (crystal)
3D Structure (crystal)