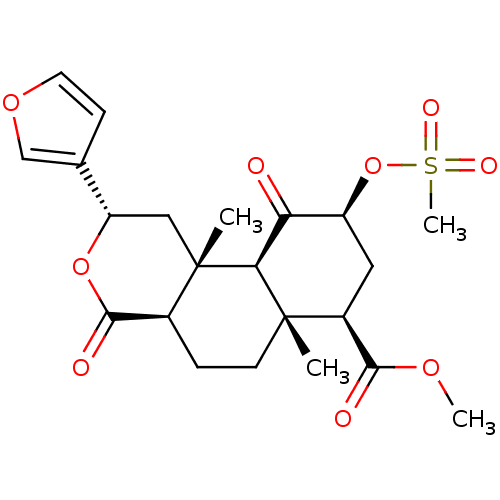
BDBM50170676 (2S,4aR,6aR,7R,9S,10aS,10bR)-methyl 2-(furan-3-yl)-6a,10b-dimethyl-9-(methylsulfonyloxy)-4,10-dioxo-dodecahydro-1H-benzo[f]isochromene-7-carboxylate::(3S,4aR,4bS,6S,8R,8aR,10aR)-3-Furan-3-yl-6-methanesulfonyloxy-4a,8a-dimethyl-1,5-dioxo-dodecahydro-2-oxa-phenanthrene-8-carboxylic acid methyl ester::CHEMBL365216
SMILES COC(=O)[C@@H]1C[C@H](OS(C)(=O)=O)C(=O)[C@H]2[C@@]1(C)CC[C@H]1C(=O)O[C@@H](C[C@]21C)c1ccoc1
InChI Key InChIKey=JPQCLBRBPPZECE-VOVNARLJSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 7 hits for monomerid = 50170676
Found 7 hits for monomerid = 50170676
Affinity DataKi: 2.30nMAssay Description:Inhibition of [125I]- IOXY binding to human Opioid receptor kappa1More data for this Ligand-Target Pair
Affinity DataKi: 2.30nMAssay Description:Displacement of [125]OXY from kappa opioid receptorMore data for this Ligand-Target Pair
Affinity DataKi: 6.82E+3nMAssay Description:Inhibition of [125I]- IOXY binding to human Opioid receptor mu1More data for this Ligand-Target Pair
Affinity DataKi: 6.82E+3nMAssay Description:Displacement of [125]OXY from mu opioid receptorMore data for this Ligand-Target Pair
Affinity DataKi: >1.00E+4nMAssay Description:Inhibition of [125I]- IOXY binding to human Opioid receptor delta1More data for this Ligand-Target Pair
Affinity DataKi: >1.00E+4nMAssay Description:Displacement of [125]OXY from delta opioid receptorMore data for this Ligand-Target Pair
Affinity DataEC50: 30nMAssay Description:Stimulation of [35S]-GTP-gammaS, binding to Opioid receptor kappa1 expressed in CHO cellsMore data for this Ligand-Target Pair