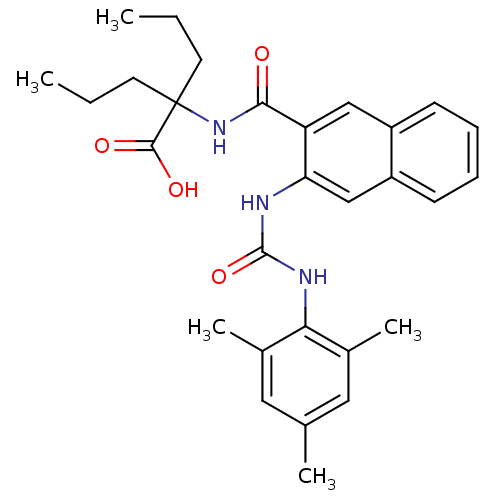
BDBM50256267 2-(3-(3-mesitylureido)-2-naphthamido)-2-propylpentanoic acid::CHEMBL472319
SMILES CCCC(CCC)(NC(=O)c1cc2ccccc2cc1NC(=O)Nc1c(C)cc(C)cc1C)C(O)=O
InChI Key InChIKey=ZUMUBJHONGJAGF-UHFFFAOYSA-N
Data 3 IC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 3 hits for monomerid = 50256267
Found 3 hits for monomerid = 50256267
Affinity DataIC50: 732nMAssay Description:Inhibition of human liver glycogen phosphorylase A by fluorescence intensity endpoint assay in presence of glucoseMore data for this Ligand-Target Pair
Affinity DataIC50: >3.30E+4nMAssay Description:Inhibition of CYP2C9 (unknown origin)More data for this Ligand-Target Pair
Affinity DataIC50: 2.52E+3nMAssay Description:Inhibition of human liver glycogen phosphorylase A in HepG2 cells assessed as forskolin-induced glycogenolysisMore data for this Ligand-Target Pair