BDBM50260093 CHEMBL4078100
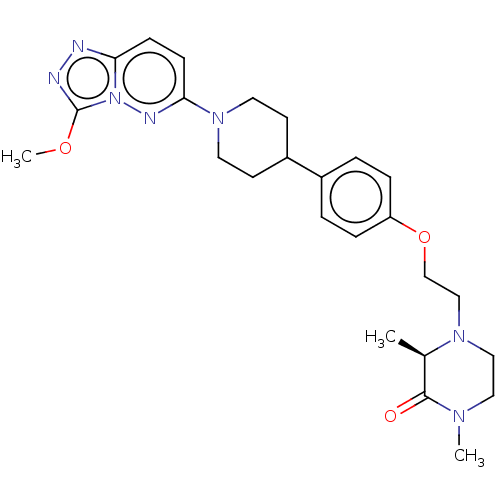
SMILES COc1nnc2ccc(nn12)N1CCC(CC1)c1ccc(OCCN2CCN(C)C(=O)[C@H]2C)cc1
InChI Key InChIKey=RSMYFSPOTCDHHJ-GOSISDBHSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 22 hits for monomerid = 50260093
Found 22 hits for monomerid = 50260093
Affinity DataKi: 5nMAssay Description:Inhibition of BRD4 (unknown origin)More data for this Ligand-Target Pair
Affinity DataKi: 5nMAssay Description:Displacement of Alexa647-labeled JQ1 derivative from wild type BRD4 tandem domain (44 to 460 residues) (unknown origin) incubated for 1 hr by fluores...More data for this Ligand-Target Pair
Affinity DataKi: 50nMAssay Description:Inhibition of human BRD4 bromodomain 1 expressed in bacterial expression systemMore data for this Ligand-Target Pair
Affinity DataKi: 63nMAssay Description:Inhibition of human BRD3 bromodomain 1 expressed in bacterial expression systemMore data for this Ligand-Target Pair
Affinity DataKi: 100nMAssay Description:Inhibition of human BRDT bromodomain 1 expressed in bacterial expression systemMore data for this Ligand-Target Pair
Affinity DataKi: 100nMAssay Description:Inhibition of human BRD2 bromodomain 1 expressed in bacterial expression systemMore data for this Ligand-Target Pair
Affinity DataKi: 126nMAssay Description:Displacement of Alexa647-labeled JQ1 derivative from wild type BRD4 bromodomain 2 (44 to 460 residues) N433A mutant (unknown origin) incubated for 1 ...More data for this Ligand-Target Pair
Affinity DataKi: 126nMAssay Description:Inhibition of human BRD4 bromodomain 2 expressed in bacterial expression systemMore data for this Ligand-Target Pair
Affinity DataKi: 158nMAssay Description:Displacement of Alexa647-labeled JQ1 derivative from wild type BRD4 bromodomain 1 (44 to 460 residues) N140A mutant (unknown origin) incubated for 1 ...More data for this Ligand-Target Pair
Affinity DataKi: 251nMAssay Description:Inhibition of human BRD2 bromodomain 2 expressed in bacterial expression systemMore data for this Ligand-Target Pair
Affinity DataKi: 398nMAssay Description:Inhibition of human BRD3 bromodomain 2 expressed in bacterial expression systemMore data for this Ligand-Target Pair
Affinity DataKi: 501nMAssay Description:Inhibition of human BRDT bromodomain 2 expressed in bacterial expression systemMore data for this Ligand-Target Pair
TargetTranscription initiation factor TFIID subunit 1(Homo sapiens (Human))
Astrazeneca
Curated by ChEMBL
Astrazeneca
Curated by ChEMBL
Affinity DataKi: 1.26E+3nMAssay Description:Inhibition of human TAF1 bromodomain 2 expressed in bacterial expression systemMore data for this Ligand-Target Pair
Affinity DataKi: 2.51E+3nMAssay Description:Inhibition of human BRD9 expressed in bacterial expression systemMore data for this Ligand-Target Pair
TargetTranscription initiation factor TFIID subunit 1-like(Homo sapiens (Human))
Astrazeneca
Curated by ChEMBL
Astrazeneca
Curated by ChEMBL
Affinity DataKi: 2.51E+3nMAssay Description:Inhibition of human TAF1L bromodomain 2 expressed in bacterial expression systemMore data for this Ligand-Target Pair
Affinity DataKi: 6.31E+3nMAssay Description:Inhibition of human CREBBP expressed in bacterial expression systemMore data for this Ligand-Target Pair
Affinity DataKi: >1.00E+4nMAssay Description:Inhibition of human EP300 expressed in bacterial expression systemMore data for this Ligand-Target Pair
Affinity DataIC50: 1.60E+3nMAssay Description:Displacement of Halo-tagged histone H3.3 from N-terminal NanoLuc-tagged BRD4 bromodomain 1 (44 to 168 residues) (unknown origin) expressed in HCT116 ...More data for this Ligand-Target Pair
Affinity DataIC50: 13nMAssay Description:Inhibition of BRD4 in human MM1S cells assessed as down regulation of c-Myc incubated for 16 hrs by flow cytometryMore data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:Inhibition of BRD4 in human LNCAP cells assessed as down regulation of nuclear AR level by immunofluorescence assayMore data for this Ligand-Target Pair
Affinity DataIC50: 25nMAssay Description:Inhibition of BRD4 in human MCF7 cells assessed as down regulation of ERalpha after 18 to 24 hrs by Hoechst staining based cellomics arrayscan analys...More data for this Ligand-Target Pair
Affinity DataIC50: 5nMAssay Description:Inhibition of recombinant human N-terminal His6-tagged BRD4 bromodomain 1 expressed in Escherichia coli BL21(DE3) using biotinylated histone peptide ...More data for this Ligand-Target Pair
 3D Structure (crystal)
3D Structure (crystal)