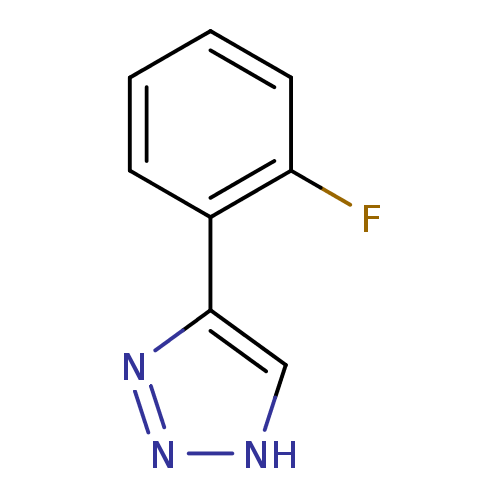
BDBM50391362 CHEMBL2148000
SMILES Fc1ccccc1-c1c[nH]nn1
InChI Key InChIKey=XQNLLPNGLMIYRT-UHFFFAOYSA-N
Data 5 IC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 5 hits for monomerid = 50391362
Found 5 hits for monomerid = 50391362
TargetIndoleamine 2,3-dioxygenase 1(Mus musculus)
Ludwig Center For Cancer Research Of The University Of Lausanne
Curated by ChEMBL
Ludwig Center For Cancer Research Of The University Of Lausanne
Curated by ChEMBL
Affinity DataIC50: 630nMAssay Description:Inhibition of mouse IDO1 in P815 clone 6 cells by HPLC analysisMore data for this Ligand-Target Pair
TargetIndoleamine 2,3-dioxygenase 1(Homo sapiens (Human))
Ludwig Center For Cancer Research Of The University Of Lausanne
Curated by ChEMBL
Ludwig Center For Cancer Research Of The University Of Lausanne
Curated by ChEMBL
Affinity DataIC50: 7.00E+4nMAssay Description:Inhibition of human IDO1 transfected in mouse P815B clone-6 cells by HPLC analysisMore data for this Ligand-Target Pair
TargetIndoleamine 2,3-dioxygenase 1(Homo sapiens (Human))
Ludwig Center For Cancer Research Of The University Of Lausanne
Curated by ChEMBL
Ludwig Center For Cancer Research Of The University Of Lausanne
Curated by ChEMBL
Affinity DataIC50: 8.00E+4nMpH: 6.5Assay Description:Inhibition of human recombinant N-terminal His-tagged IDO1 (Ala2 to Gly403) overexpressed in Escherichia coli BL21 at pH 6.5 after 60 mins by HPLC an...More data for this Ligand-Target Pair
TargetTryptophan 2,3-dioxygenase(Homo sapiens (Human))
Ludwig Center For Cancer Research Of The University Of Lausanne
Curated by ChEMBL
Ludwig Center For Cancer Research Of The University Of Lausanne
Curated by ChEMBL
Affinity DataIC50: >1.00E+5nMAssay Description:Inhibition of human TDO transfected in mouse P815B clone 19 cells by HPLC analysisMore data for this Ligand-Target Pair
TargetTryptophan 2,3-dioxygenase(Mus musculus)
Ludwig Center For Cancer Research Of The University Of Lausanne
Curated by ChEMBL
Ludwig Center For Cancer Research Of The University Of Lausanne
Curated by ChEMBL
Affinity DataIC50: >1.00E+5nMAssay Description:Inhibition of mouse TDO in P815 clone 12 cells by HPLC analysisMore data for this Ligand-Target Pair