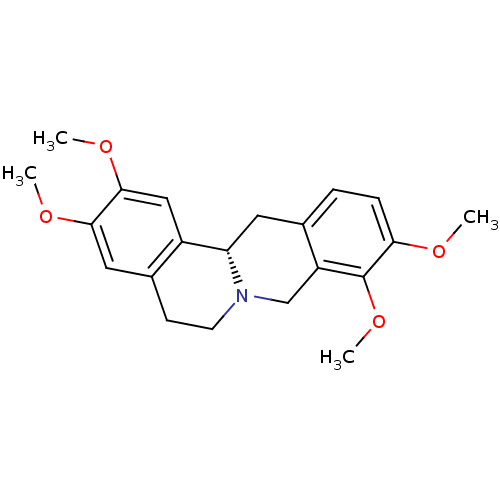
BDBM50424077 (-)-Tetrahydropalmatine::L-Tetrahydropalmatine::TETRAHYDROPALMATINE
SMILES COc1cc2CCN3Cc4c(C[C@H]3c2cc1OC)ccc(OC)c4OC
InChI Key InChIKey=AEQDJSLRWYMAQI-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 15 hits for monomerid = 50424077
Found 15 hits for monomerid = 50424077
Affinity DataIC50: 23nMAssay Description:Inhibition of tissue factor procoagulant activity in LPS-stimulated human THP1 cells preincubated for 1 hr before LPS addition measured after 5 hrsMore data for this Ligand-Target Pair
Affinity DataKi: 124nMAssay Description:Binding affinity to D1 receptor (unknown origin) assessed as inhibition constantMore data for this Ligand-Target Pair
Affinity DataKi: 153nMAssay Description:Displacement of [3H]SCH23390 from human 3HA-tagged dopamine D1 receptor expressed in HEK293 cells after 1 hrMore data for this Ligand-Target Pair
Affinity DataKi: 231nMAssay Description:Displacement of [3H]SCH23390 from dopamine D1 receptor (unknown origin) expressed in human HEK293 cells by liquid scintillation counterMore data for this Ligand-Target Pair
Affinity DataKi: 305nMAssay Description:Displacement of [3H]SCH23390 from human dopamine D5 receptor expressed in HEK293 cells after 1 hrMore data for this Ligand-Target Pair
Affinity DataKi: 388nMAssay Description:Binding affinity to D2 receptor (unknown origin) assessed as inhibition constantMore data for this Ligand-Target Pair
Affinity DataIC50: 450nMAssay Description:Antagonist activity at D2 receptor (unknown origin) expressed in CHOK1 cells assessed as inhibition of dopamine-induced calcium flux preincubated for...More data for this Ligand-Target Pair
Affinity DataKi: >1.00E+3nMAssay Description:Displacement of [3H]SCH23390 from human dopamine D4 receptor expressed in HEK293 cells after 1 hrMore data for this Ligand-Target Pair
Affinity DataKi: 1.13E+3nMAssay Description:Displacement of [3H]SCH23390 from human FLAG-tagged dopamine D2 receptor expressed in HEK293 cells after 1 hrMore data for this Ligand-Target Pair
Affinity DataKi: 1.37E+3nMAssay Description:Displacement of [3H]SCH23390 from human FLAG-tagged dopamine D3 receptor expressed in HEK293 cells after 1 hrMore data for this Ligand-Target Pair
Affinity DataKi: 1.42E+3nMAssay Description:Binding affinity to D3 receptor (unknown origin) assessed as inhibition constantMore data for this Ligand-Target Pair
Affinity DataIC50: 1.63E+3nMAssay Description:Antagonist activity at D1 receptor (unknown origin) expressed in CHOK1 cells assessed as inhibition of SKF38393-induced cAMP accumulation measured af...More data for this Ligand-Target Pair
Affinity DataKi: >5.00E+3nMAssay Description:Displacement of [3H]spiperone from dopamine D2 receptor (unknown origin) expressed in human HEK293 cells by liquid scintillation counterMore data for this Ligand-Target Pair
Affinity DataKi: >5.00E+3nMAssay Description:Displacement of [3H]8-OH-DPAT from 5-HT1A receptor (unknown origin) expressed in HEK293 cells by liquid scintillation counterMore data for this Ligand-Target Pair
TargetReverse transcriptase(Human immunodeficiency virus type 1)
University of Illinois
Curated by ChEMBL
University of Illinois
Curated by ChEMBL
Affinity DataIC50: 5.62E+5nMAssay Description:Inhibition of HIV1 RTMore data for this Ligand-Target Pair