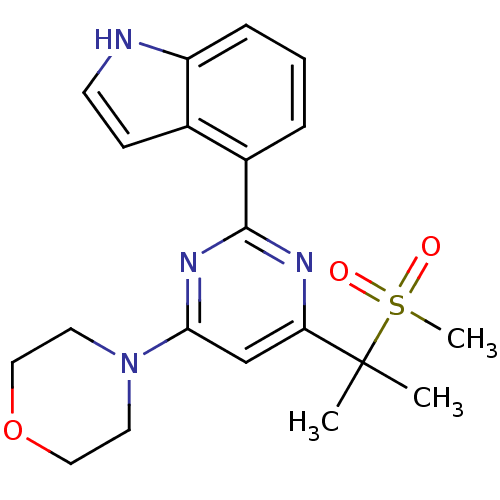
BDBM50427322 CHEMBL2325703
SMILES CC(C)(c1cc(nc(n1)-c1cccc2[nH]ccc12)N1CCOCC1)S(C)(=O)=O
InChI Key InChIKey=TYPBASYBSGLAOL-UHFFFAOYSA-N
Data 6 IC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 6 hits for monomerid = 50427322
Found 6 hits for monomerid = 50427322
Affinity DataIC50: 2.70E+3nMAssay Description:Inhibition of mTOR-mediated AKT phosphorylation at serine 473 in human MDA-MB-468 cellsMore data for this Ligand-Target Pair
Affinity DataIC50: 280nMAssay Description:Inhibition of ATR-mediated CHK1 phosphorylation at serine 345 in human HT29 cells after 1 hr in presence of 4-nitroquinoline 1-oxideMore data for this Ligand-Target Pair
Affinity DataIC50: 190nMAssay Description:Inhibition of mTOR-mediated AKT phosphorylation at serine 473 in human MDA-MB-468 cellsMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
Astrazeneca
Curated by ChEMBL
Astrazeneca
Curated by ChEMBL
Affinity DataIC50: >3.30E+4nMAssay Description:Inhibition of human ERG by whole-cell electrophysiology assayMore data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform(Homo sapiens (Human))
Astrazeneca
Curated by ChEMBL
Astrazeneca
Curated by ChEMBL
Affinity DataIC50: 1.60E+3nMAssay Description:Inhibition of PI3Kalpha-mediated AKT phosphorylation at threonine 308 in human BT474 cellsMore data for this Ligand-Target Pair
Affinity DataIC50: 3nMAssay Description:Inhibition of ATR-mediated CHK1 phosphorylation at serine 345 in human HT29 cells after 1 hr in presence of 4-nitroquinoline 1-oxideMore data for this Ligand-Target Pair