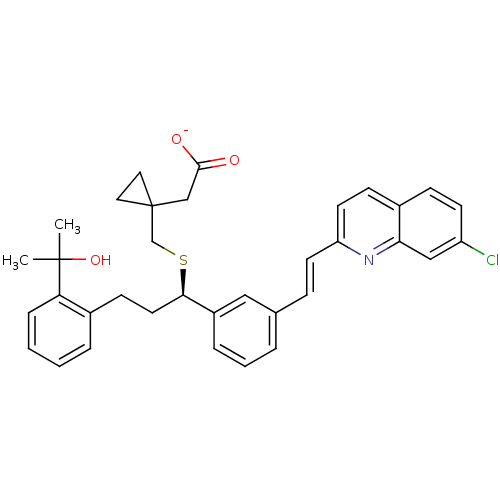
BDBM58922 MLS001304727::MONTELUKAST Na::SMR000469188::cid_23663996::sodium;2-[1-[[(1R)-1-[3-[(E)-2-(7-chloranylquinolin-2-yl)ethenyl]phenyl]-3-[2-(2-oxidanylpropan-2-yl)phenyl]propyl]sulfanylmethyl]cyclopropyl]ethanoate::sodium;2-[1-[[(1R)-1-[3-[(E)-2-(7-chloroquinolin-2-yl)ethenyl]phenyl]-3-[2-(2-hydroxypropan-2-yl)phenyl]propyl]sulfanylmethyl]cyclopropyl]acetate::sodium;2-[1-[[[(1R)-1-[3-[(E)-2-(7-chloro-2-quinolinyl)ethenyl]phenyl]-3-[2-(2-hydroxypropan-2-yl)phenyl]propyl]thio]methyl]cyclopropyl]acetate::sodium;2-[1-[[[(1R)-1-[3-[(E)-2-(7-chloro-2-quinolyl)vinyl]phenyl]-3-[2-(1-hydroxy-1-methyl-ethyl)phenyl]propyl]thio]methyl]cyclopropyl]acetate
SMILES CC(C)(O)c1ccccc1CC[C@@H](SCC1(CC([O-])=O)CC1)c1cccc(\C=C\c2ccc3ccc(Cl)cc3n2)c1
InChI Key InChIKey=UCHDWCPVSPXUMX-UHFFFAOYSA-M
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 6 hits for monomerid = 58922
Found 6 hits for monomerid = 58922
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay