Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
3-hydroxy-3-methylglutaryl-coenzyme A reductase
Ligand
BDBM50014826
Substrate
n/a
Meas. Tech.
ChEBML_80478
IC50
2.00±n/a nM
Citation
 Beck, G; Kesseler, K; Baader, E; Bartmann, W; Bergmann, A; Granzer, E; Jendralla, H; von Kerekjarto, B; Krause, R; Paulus, E Synthesis and biological activity of new HMG-CoA reductase inhibitors. 1. Lactones of pyridine- and pyrimidine-substituted 3,5-dihydroxy-6-heptenoic (-heptanoic) acids. J Med Chem 33:52-60 (1990) [PubMed] Article
Beck, G; Kesseler, K; Baader, E; Bartmann, W; Bergmann, A; Granzer, E; Jendralla, H; von Kerekjarto, B; Krause, R; Paulus, E Synthesis and biological activity of new HMG-CoA reductase inhibitors. 1. Lactones of pyridine- and pyrimidine-substituted 3,5-dihydroxy-6-heptenoic (-heptanoic) acids. J Med Chem 33:52-60 (1990) [PubMed] Article More Info.:
Target
Name:
3-hydroxy-3-methylglutaryl-coenzyme A reductase
Synonyms:
3-hydroxy-3-methylglutaryl-coenzyme A (HMG-CoA) reductase | 3-hydroxy-3-methylglutaryl-coenzyme A reductase (HMG-CoA) | HMDH_HUMAN | HMG-CoA Reductase | HMG-CoA reductase (HMGR) | HMGCR
Type:
Enzyme
Mol. Mass.:
97477.10
Organism:
Homo sapiens (Human)
Description:
P04035
Residue:
888
Sequence:
MLSRLFRMHGLFVASHPWEVIVGTVTLTICMMSMNMFTGNNKICGWNYECPKFEEDVLSSDIIILTITRCIAILYIYFQFQNLRQLGSKYILGIAGLFTIFSSFVFSTVVIHFLDKELTGLNEALPFFLLLIDLSRASTLAKFALSSNSQDEVRENIARGMAILGPTFTLDALVECLVIGVGTMSGVRQLEIMCCFGCMSVLANYFVFMTFFPACVSLVLELSRESREGRPIWQLSHFARVLEEEENKPNPVTQRVKMIMSLGLVLVHAHSRWIADPSPQNSTADTSKVSLGLDENVSKRIEPSVSLWQFYLSKMISMDIEQVITLSLALLLAVKYIFFEQTETESTLSLKNPITSPVVTQKKVPDNCCRREPMLVRNNQKCDSVEEETGINRERKVEVIKPLVAETDTPNRATFVVGNSSLLDTSSVLVTQEPEIELPREPRPNEECLQILGNAEKGAKFLSDAEIIQLVNAKHIPAYKLETLMETHERGVSIRRQLLSKKLSEPSSLQYLPYRDYNYSLVMGACCENVIGYMPIPVGVAGPLCLDEKEFQVPMATTEGCLVASTNRGCRAIGLGGGASSRVLADGMTRGPVVRLPRACDSAEVKAWLETSEGFAVIKEAFDSTSRFARLQKLHTSIAGRNLYIRFQSRSGDAMGMNMISKGTEKALSKLHEYFPEMQILAVSGNYCTDKKPAAINWIEGRGKSVVCEAVIPAKVVREVLKTTTEAMIEVNINKNLVGSAMAGSIGGYNAHAANIVTAIYIACGQDAAQNVGSSNCITLMEASGPTNEDLYISCTMPSIEIGTVGGGTNLLPQQACLQMLGVQGACKDNPGENARQLARIVCGTVMAGELSLMAALAAGHLVKSHMIHNRSKINLQDLQGACTKKTA
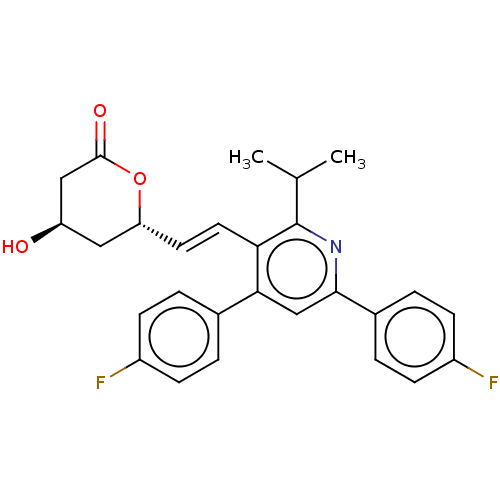
Inhibitor
Name:
BDBM50014826
Synonyms:
(E)-6-{2-[4,6-Bis-(4-fluoro-phenyl)-2-isopropyl-pyridin-3-yl]-vinyl}-4-hydroxy-tetrahydro-pyran-2-one | CHEMBL2368202
Type:
Small organic molecule
Emp. Form.:
C27H25F2NO3
Mol. Mass.:
449.4891
SMILES:
CC(C)c1nc(cc(-c2ccc(F)cc2)c1\C=C\[C@@H]1C[C@@H](O)CC(=O)O1)-c1ccc(F)cc1