Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Genome polyprotein
Ligand
BDBM21398
Substrate
n/a
Meas. Tech.
ChEMBL_158047 (CHEMBL767017)
IC50
140000±n/a nM
Citation
 De Voss, JJ; Sui, Z; DeCamp, DL; Salto, R; Babé, LM; Craik, CS; Ortiz de Montellano, PR Haloperidol-based irreversible inhibitors of the HIV-1 and HIV-2 proteases. J Med Chem 37:665-73 (1994) [PubMed] Article
De Voss, JJ; Sui, Z; DeCamp, DL; Salto, R; Babé, LM; Craik, CS; Ortiz de Montellano, PR Haloperidol-based irreversible inhibitors of the HIV-1 and HIV-2 proteases. J Med Chem 37:665-73 (1994) [PubMed] Article More Info.:
Target
Name:
Genome polyprotein
Synonyms:
Human rhinovirus A protease | Human rhinovirus B 3A protease
Type:
PROTEIN
Mol. Mass.:
44361.04
Organism:
Human rhinovirus B
Description:
ChEMBL_158953
Residue:
401
Sequence:
AFRPCNVNTKIGNAKCCPFVCGKAVTFKDRSTCSTYNLSSSLHHILEEDKRRRQVVDVMSAIFQGPISLDAPPPPAIADLLQSVRTPRVIKYCQIIMGHPAECQVERDLNIANSIIAIIANIISIAGIIFVIYKLFCSLQGPYSGEPKPKTKVPERRVVAQGPEEEFGRSILKNNTCVITTGNGKFTGLGIHDRILIIPTHADPGREVQVNGVHTKVLDSYDLYNRDGVKLEITVIQLDRNEKFRDIRKYIPETEDDYPECNLALSANQDEPTIIKVGDVVSYGNILLSGNQTARMLKYNYPTKSGYCGGVLYKIGQILGIHVGGNGRDGFSAMLLRSYFTGQIKVNKHATECGLPDIQTIHTPSKTKLQPSVFYDVFPGSKEPAVLTDNDPRLEVNFKEA
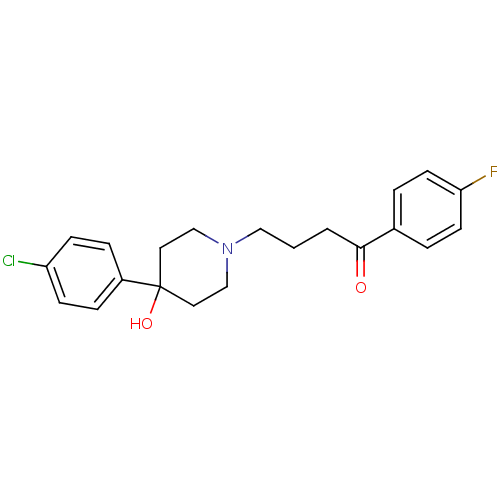
Inhibitor
Name:
BDBM21398
Synonyms:
4-[4-(4-Chloro-phenyl)-4-hydroxy-piperidin-1-yl]-1-(4-fluoro-phenyl)-butan-1-one;propionate(HCl) | 4-[4-(4-chlorophenyl)-4-hydroxypiperidin-1-yl]-1-(4-fluorophenyl)butan-1-one | CHEMBL54 | CHEMBL545608 | Haloperidol | Haloperidol, 1
Type:
Small organic molecule
Emp. Form.:
C21H23ClFNO2
Mol. Mass.:
375.864
SMILES:
OC1(CCN(CCCC(=O)c2ccc(F)cc2)CC1)c1ccc(Cl)cc1