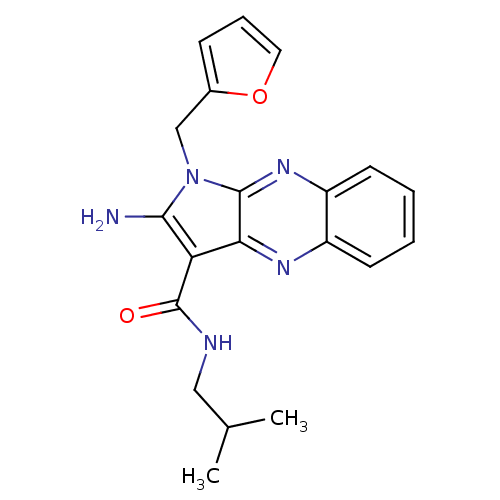
BDBM29661 2-Amino-1-furan-2-ylmethyl-1H-pyrrolo[2,3-b]quinoxaline-3-carboxylic acid isobutyl-amide::2-amino-1-(2-furanylmethyl)-N-(2-methylpropyl)-3-pyrrolo[3,2-b]quinoxalinecarboxamide::2-amino-1-(2-furfuryl)-N-isobutyl-pyrrolo[3,2-b]quinoxaline-3-carboxamide::2-amino-1-(furan-2-ylmethyl)-N-(2-methylpropyl)pyrrolo[3,2-b]quinoxaline-3-carboxamide::2-azanyl-1-(furan-2-ylmethyl)-N-(2-methylpropyl)pyrrolo[3,2-b]quinoxaline-3-carboxamide::MLS000556576::SMR000147893::cid_1881699
SMILES CC(C)CNC(=O)c1c(N)n(Cc2ccco2)c2nc3ccccc3nc12
InChI Key InChIKey=WSKCZZJHPRBJLI-UHFFFAOYSA-N
Data 3 IC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 2 hits for monomerid = 29661
Found 2 hits for monomerid = 29661
TargetRunt-related transcription factor 1(Homo sapiens (Human))
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Affinity DataIC50: 1.17E+4nMpH: 7.4 T: 2°CAssay Description:This assay is to use HTS to identify inhibitors of the protein-protein interaction between the RUNX1 Runt domain and CBFbeta-SMMHC, a potential thera...More data for this Ligand-Target Pair
TargetRunt-related transcription factor 1(Homo sapiens (Human))
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Affinity DataIC50: 1.17E+4nMpH: 7.4 T: 2°CAssay Description:This assay is to identify inhibitors of the protein-protein interaction between the RUNX1 Runt domain and CBFbeta-SMMHC. This is accomplished by usin...More data for this Ligand-Target Pair