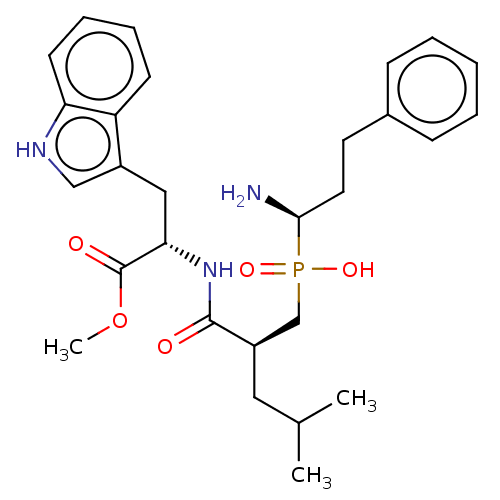
BDBM50570849 CHEMBL4854733
SMILES COC(=O)[C@H](Cc1c[nH]c2ccccc12)NC(=O)[C@H](CC(C)C)CP(O)(=O)[C@@H](N)CCc1ccccc1
InChI Key InChIKey=QWLYILUODIZPOK-ZSQFBXSQSA-N
Data 6 IC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 6 hits for monomerid = 50570849
Found 6 hits for monomerid = 50570849
Affinity DataIC50: 1.5nMAssay Description:Inhibition of APN (unknown origin) using A-AMC as substrateMore data for this Ligand-Target Pair
Affinity DataIC50: 1.5nMAssay Description:Inhibition of APN (unknown origin) using A-AMC as substrateMore data for this Ligand-Target Pair
TargetEndoplasmic reticulum aminopeptidase 2(Human)
The Institute of Cancer Research
Curated by ChEMBL
The Institute of Cancer Research
Curated by ChEMBL
Affinity DataIC50: 900nMAssay Description:Inhibition of ERAP2 (unknown origin) using R-AMC as substrateMore data for this Ligand-Target Pair
TargetEndoplasmic reticulum aminopeptidase 1(Human)
The Institute of Cancer Research
Curated by ChEMBL
The Institute of Cancer Research
Curated by ChEMBL
Affinity DataIC50: 140nMAssay Description:Inhibition of ERAP1 (unknown origin) using L-AMC as substrateMore data for this Ligand-Target Pair
TargetEndoplasmic reticulum aminopeptidase 2(Human)
The Institute of Cancer Research
Curated by ChEMBL
The Institute of Cancer Research
Curated by ChEMBL
Affinity DataIC50: 891nMAssay Description:Inhibition of ERAP2 (unknown origin) using R-AMC as substrateMore data for this Ligand-Target Pair
TargetEndoplasmic reticulum aminopeptidase 1(Human)
The Institute of Cancer Research
Curated by ChEMBL
The Institute of Cancer Research
Curated by ChEMBL
Affinity DataIC50: 141nMAssay Description:Inhibition of ERAP1 (unknown origin) using L-AMC as substrateMore data for this Ligand-Target Pair