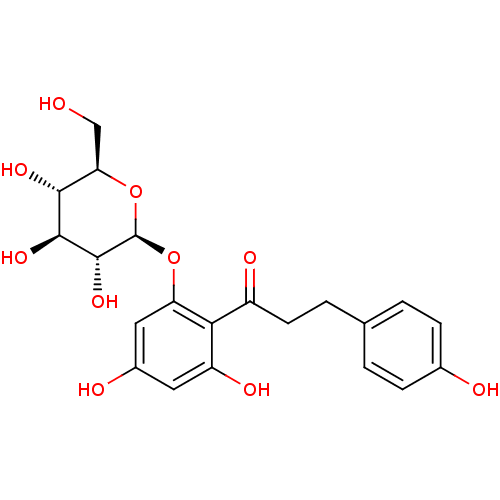
BDBM20875 1-(2,4-dihydroxy-6-{[(2S,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxy}phenyl)-3-(4-hydroxyphenyl)propan-1-one::CHEMBL241384::CHEMBL245067::JMC511145 Compound 1::O-glucoside, 1::Phlorizin
SMILES OC[C@H]1O[C@@H](Oc2cc(O)cc(O)c2C(=O)CCc2ccc(O)cc2)[C@H](O)[C@@H](O)[C@@H]1O
InChI Key InChIKey=IOUVKUPGCMBWBT-QNDFHXLGSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 5 hits for monomerid = 20875
Found 5 hits for monomerid = 20875
Affinity DataKi: 19nMAssay Description:Inhibition of human kidney SGLT2 assessed as renal glucose reabsorptionMore data for this Ligand-Target Pair
TargetSodium/glucose cotransporter 1(Human)
Max-Planck-Institut F£R Molekulare Physiologie
Curated by ChEMBL
Max-Planck-Institut F£R Molekulare Physiologie
Curated by ChEMBL
Affinity DataKi: 180nMAssay Description:Binding affinity against Sodium/glucose co-transporter of isolated renal brush border membranes.More data for this Ligand-Target Pair
TargetSolute carrier family 28 member 3(Human)
University of Tennessee Health Sciences Center
Curated by ChEMBL
University of Tennessee Health Sciences Center
Curated by ChEMBL
Affinity DataKi: 1.60E+4nMAssay Description:Binding affinity to human recombinant CNT3 expressed in pig PK15NTD cells assessed as [3H]uridine uptake by beta-scintillation counterMore data for this Ligand-Target Pair
Affinity DataKi: 6.43E+4nMAssay Description:Inhibition of diphenolase activity of mushroom tyrosinaseMore data for this Ligand-Target Pair
TargetEquilibrative nucleoside transporter 1(Human)
University of Tennessee Health Sciences Center
Curated by ChEMBL
University of Tennessee Health Sciences Center
Curated by ChEMBL
Affinity DataKi: >1.00E+6nMAssay Description:Binding affinity to human ENT1 assessed as [3H]uridine uptake by flow cytometryMore data for this Ligand-Target Pair
 3D Structure (crystal)
3D Structure (crystal)