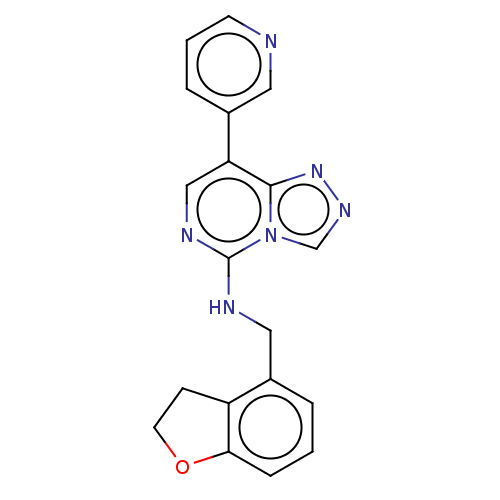
BDBM291701 N-((2,3-dihydrobenzofuran-4-yl)methyl)-8-(pyridin-3-yl)-[1,2,4]triazolo[4,3-c]pyrimidin-5-amine::US11207325, Example 16::US9580437, Example 16
SMILES C(Nc1ncc(-c2cccnc2)c2nncn12)c1cccc2OCCc12
InChI Key InChIKey=VFLXVDWCZHLLLM-UHFFFAOYSA-N
Data 11 IC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 11 hits for monomerid = 291701
Found 11 hits for monomerid = 291701
Affinity DataIC50: 10.8nMAssay Description:To assess the compounds potency in the EED-H3K27Me3 competition binding assay, compounds were serially diluted 3-fold in DMSO to obtain a total of tw...More data for this Ligand-Target Pair
TargetHistone-lysine N-methyltransferase EZH2/Polycomb protein EED/Polycomb protein SUZ12(Human)
Novartis
US Patent
Novartis
US Patent
Affinity DataIC50: 10.8nMpH: 8.0Assay Description:To assess the compounds potency in the EED-H3K27Me3 competition binding assay, compounds were serially diluted 3-fold in DMSO to obtain a total of tw...More data for this Ligand-Target Pair
Affinity DataIC50: 24nMAssay Description:Representative compounds of the present disclosure were serially and separately diluted 3-fold in DMSO to obtain a total of eight or twelve concentra...More data for this Ligand-Target Pair
TargetHistone-lysine N-methyltransferase EZH2/Polycomb protein EED/Polycomb protein SUZ12(Human)
Novartis
US Patent
Novartis
US Patent
Affinity DataIC50: 24nMpH: 8.0Assay Description:Representative compounds of the present disclosure were serially and separately diluted 3-fold in DMSO to obtain a total of eight or twelve concentra...More data for this Ligand-Target Pair
Affinity DataIC50: 60.4nMAssay Description:Representative compounds of the present disclosure were serially and separately diluted 3-fold in DMSO to obtain a total of eight or twelve concentra...More data for this Ligand-Target Pair
TargetHistone-lysine N-methyltransferase EZH2/Polycomb protein EED/Polycomb protein SUZ12(Human)
Novartis
US Patent
Novartis
US Patent
Affinity DataIC50: 60.4nMAssay Description:Cell lysates were transferred to the wells of a 384-well plate and the final volume was adjusted to 50 μL per well with PBS. The plate was seale...More data for this Ligand-Target Pair
Affinity DataIC50: 100nMAssay Description:Inhibition of CYP2D6 in human liver microsomes using bufuralol as substrate incubated for 10 mins in presence of NADPH by LC-MS/MS analysisMore data for this Ligand-Target Pair
TargetcAMP-specific 3',5'-cyclic phosphodiesterase 4D(Human)
Novartis Institutes For Biomedical Research
Curated by ChEMBL
Novartis Institutes For Biomedical Research
Curated by ChEMBL
Affinity DataIC50: 2.60E+3nMAssay Description:Inhibition of human Phosphodiesterase 4DMore data for this Ligand-Target Pair
TargetAlpha-2C adrenergic receptor(Human)
Novartis Institutes For Biomedical Research
Curated by ChEMBL
Novartis Institutes For Biomedical Research
Curated by ChEMBL
Affinity DataIC50: 3.90E+3nMAssay Description:Inhibition of alpha2c adrenergic receptor (unknown origin)More data for this Ligand-Target Pair
Affinity DataIC50: 1.20E+4nMAssay Description:Inhibition of CYP2C9 in human liver microsomes using diclofenac as substrate incubated for 10 mins in presence of NADPH by LC-MS/MS analysisMore data for this Ligand-Target Pair
Affinity DataIC50: 2.90E+4nMAssay Description:Inhibition of CYP3A4 in human liver microsomes using midazolam as substrate incubated for 10 mins in presence of NADPH by LC-MS/MS analysisMore data for this Ligand-Target Pair