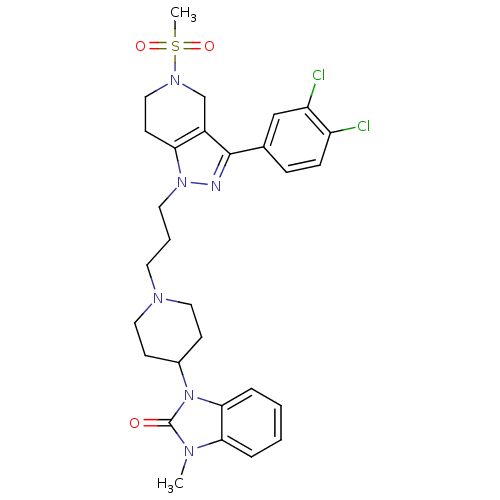
BDBM50162828 1-(1-(3-(3-(3,4-dichlorophenyl)-5-(methylsulfonyl)-4,5,6,7-tetrahydropyrazolo[4,3-c]pyridin-1-yl)propyl)piperidin-4-yl)-3-methyl-1H-benzo[d]imidazol-2(3H)-one::1-(1-{3-[3-(3,4-Dichloro-phenyl)-5-methanesulfonyl-4,5,6,7-tetrahydro-pyrazolo[4,3-c]pyridin-1-yl]-propyl}-piperidin-4-yl)-3-methyl-1,3-dihydro-benzoimidazol-2-one::CHEMBL180108
SMILES Cn1c2ccccc2n(C2CCN(CCCn3nc(c4CN(CCc34)S(C)(=O)=O)-c3ccc(Cl)c(Cl)c3)CC2)c1=O
InChI Key InChIKey=YQQYXWVMTOUJTM-UHFFFAOYSA-N
Data 3 IC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 3 hits for monomerid = 50162828
Found 3 hits for monomerid = 50162828
Affinity DataIC50: 10nMAssay Description:Inhibition of human cathepsin SMore data for this Ligand-Target Pair
Affinity DataIC50: 50nMAssay Description:Inhibitory concentration against cathepsin SMore data for this Ligand-Target Pair
Affinity DataIC50: 800nMAssay Description:Inhibition of cathepsin S in human JY cells by invariant chain degradation assayMore data for this Ligand-Target Pair