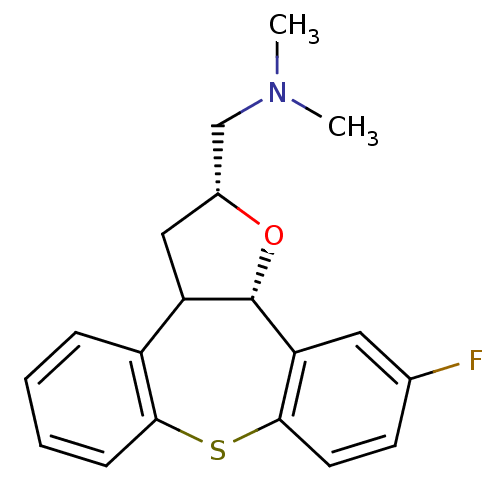
BDBM50162957 (11-Fluoro-2,3,3a,12b-tetrahydro-1-oxa-8-thia-dibenzo[e,h]azulen-2-ylmethyl)-dimethyl-amine::CHEMBL193639
SMILES CN(C)C[C@H]1CC2[C@H](O1)c1cc(F)ccc1Sc1ccccc21
InChI Key InChIKey=BSOLCASXROXPRZ-UHFFFAOYSA-N
Data 19 KI
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 19 hits for monomerid = 50162957
Found 19 hits for monomerid = 50162957
Target5-hydroxytryptamine receptor 2C(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 0.210nMAssay Description:Inhibition of [3H]mesulergine binding to human 5-hydroxytryptamine 2C receptorMore data for this Ligand-Target Pair
TargetHistamine H1 receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 0.520nMAssay Description:Inhibition of [3H]pyrilamine binding to human Histamine H1 receptor More data for this Ligand-Target Pair
TargetHistamine H1 receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 0.520nMAssay Description:Inhibition of [3H]pyrilamine binding to human Histamine H1 receptor More data for this Ligand-Target Pair
Target5-hydroxytryptamine receptor 2A(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 0.560nMAssay Description:Inhibition of [125I]R91150 binding to human 5-hydroxytryptamine 2A receptorMore data for this Ligand-Target Pair
Target5-hydroxytryptamine receptor 2A(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 0.560nMAssay Description:Inhibition of [125I]R91150 binding to human 5-hydroxytryptamine 2A receptorMore data for this Ligand-Target Pair
TargetD(3) dopamine receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 1.10nMAssay Description:Inhibition of [125I]iodosulpiride binding to human Dopamine receptor D3More data for this Ligand-Target Pair
TargetD(3) dopamine receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 1.10nMAssay Description:Inhibition of [125I]iodosulpiride binding to human Dopamine receptor D3More data for this Ligand-Target Pair
Affinity DataKi: 1.60nMAssay Description:Inhibition of [3H]nisoxetine binding to rat Norepinephrine transporterMore data for this Ligand-Target Pair
Affinity DataKi: 1.60nMAssay Description:Inhibition of [3H]nisoxetine binding to rat Norepinephrine transporterMore data for this Ligand-Target Pair
TargetD(2) dopamine receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 2.30nMAssay Description:Inhibition of [3H]-spiperone binding to human Dopamine receptor D2More data for this Ligand-Target Pair
TargetD(2) dopamine receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 2.30nMAssay Description:Inhibition of [3H]-spiperone binding to human Dopamine receptor D2More data for this Ligand-Target Pair
TargetD(1A) dopamine receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 5.20nMAssay Description:Inhibition of [3H]SCH-23390 binding to human Dopamine receptor D1More data for this Ligand-Target Pair
TargetD(1A) dopamine receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 5.20nMAssay Description:Inhibition of [3H]SCH-23390 binding to human Dopamine receptor D1More data for this Ligand-Target Pair
Target5-hydroxytryptamine receptor 7(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 6.20nMAssay Description:Inhibition of [3H]5-HT binding to human 5-hydroxytryptamine 7 receptorMore data for this Ligand-Target Pair
Target5-hydroxytryptamine receptor 7(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 6.60nMAssay Description:Inhibition of [3H]5-HT binding to human 5-hydroxytryptamine 7 receptorMore data for this Ligand-Target Pair
TargetAlpha-2C adrenergic receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 59nMAssay Description:Inhibition of [3H]rauwolscine binding to Alpha-2C adrenergic receptorMore data for this Ligand-Target Pair
TargetAlpha-2C adrenergic receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 59nMAssay Description:Inhibition of [3H]rauwolscine binding to Alpha-2C adrenergic receptorMore data for this Ligand-Target Pair
TargetAlpha-2A adrenergic receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 66nMAssay Description:Inhibition of [3H]rauwolscine binding to Alpha-2A adrenergic receptorMore data for this Ligand-Target Pair
TargetAlpha-2A adrenergic receptor(Human)
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Johnson & Johnson Pharmaceutical Research & Development
Curated by ChEMBL
Affinity DataKi: 66nMAssay Description:Inhibition of [3H]rauwolscine binding to Alpha-2A adrenergic receptorMore data for this Ligand-Target Pair