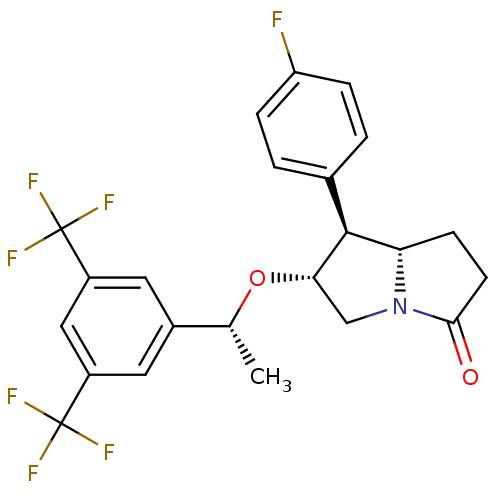
BDBM50234162 (6R,7S,7aS)-6-((R)-1-(3,5-bis(trifluoromethyl)phenyl)ethoxy)-7-(4-fluorophenyl)tetrahydro-1H-pyrrolizin-3(2H)-one::(6R,7S,7aS)-6-{(1R)-1-[3,5-bis(trifluoromethyl)phenyl]ethoxy}-7-(4-fluorophenyl)hexahydro-3H-pyrrolizin-3-one::CHEMBL272649
SMILES C[C@@H](O[C@H]1CN2[C@@H](CCC2=O)[C@@H]1c1ccc(F)cc1)c1cc(cc(c1)C(F)(F)F)C(F)(F)F
InChI Key InChIKey=SUITYIWBXSZXIQ-UHFFFAOYSA-N
Data 6 IC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 6 hits for monomerid = 50234162
Found 6 hits for monomerid = 50234162
Affinity DataIC50: 0.0600nMAssay Description:Displacement of [125I]L-703606 from human NK1 expressed in CHO cellsMore data for this Ligand-Target Pair
Affinity DataIC50: 0.0600nMAssay Description:Displacement of [125I]substance P from human NK1 receptor expressed in CHO cellsMore data for this Ligand-Target Pair
Affinity DataIC50: 0.0600nMAssay Description:Displacement of [125I]substance P from human NK1 receptor expressed in CHO cellsMore data for this Ligand-Target Pair
Affinity DataIC50: 2.20nMAssay Description:Displacement of [125I]substance P from human NK1 receptor expressed in CHO cells in the presence of 50 percent human serumMore data for this Ligand-Target Pair
Affinity DataIC50: 2.20nMAssay Description:Displacement of [125I]substance P from human NK1 receptor expressed in CHO cells in presence of 50% human serumMore data for this Ligand-Target Pair
TargetNuclear receptor subfamily 1 group I member 2(Human)
Merck Research Laboratories
Curated by ChEMBL
Merck Research Laboratories
Curated by ChEMBL
Affinity DataIC50: 4.80E+3nMAssay Description:Inhibition of human PXR inductionMore data for this Ligand-Target Pair