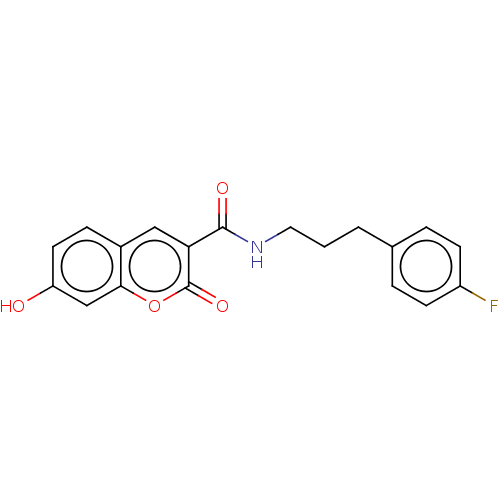
BDBM50241828 CHEMBL4089817
SMILES Oc1ccc2cc(C(=O)NCCCc3ccc(F)cc3)c(=O)oc2c1
InChI Key InChIKey=YNGXWVIJSUJZSG-UHFFFAOYSA-N
Data 8 IC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 8 hits for monomerid = 50241828
Found 8 hits for monomerid = 50241828
Affinity DataIC50: 3.5nMAssay Description:Inhibition of recombinant human AKR1B10 using pyridine-3-aldehyde as substrateMore data for this Ligand-Target Pair
Affinity DataIC50: 277nMAssay Description:Inhibition of recombinant human AKR1B1 using pyridine-3-aldehyde as substrateMore data for this Ligand-Target Pair
Affinity DataIC50: 350nMAssay Description:Inhibition of recombinant human AKR1C4 using S-tetralol as substrateMore data for this Ligand-Target Pair
Affinity DataIC50: 470nMAssay Description:Inhibition of recombinant human AKR1C3 using S-tetralol as substrateMore data for this Ligand-Target Pair
Affinity DataIC50: 610nMAssay Description:Inhibition of recombinant human AKR1C2 using S-tetralol as substrateMore data for this Ligand-Target Pair
Affinity DataIC50: 1.10E+3nMAssay Description:Inhibition of recombinant human CBR1 using isatin as substrateMore data for this Ligand-Target Pair
Affinity DataIC50: 1.70E+3nMAssay Description:Inhibition of recombinant human AKR1A1 using pyridine-3-aldehyde as substrateMore data for this Ligand-Target Pair
Affinity DataIC50: 1.20E+4nMAssay Description:Inhibition of recombinant human AKR1C1 using S-tetralol as substrateMore data for this Ligand-Target Pair