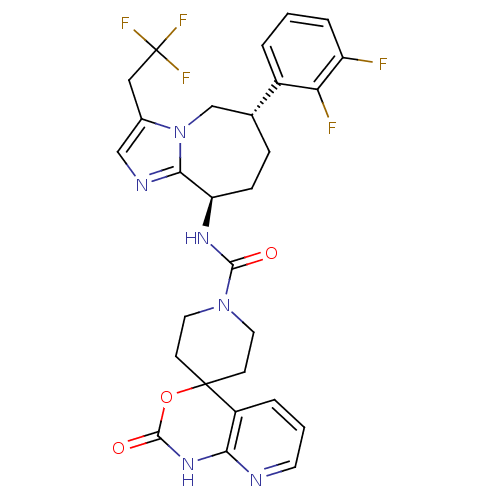
BDBM50342630 CHEMBL1770720::N-((6S,9R)-6-(2,3-difluorophenyl)-3-(2,2,2-trifluoroethyl)-6,7,8,9-tetrahydro-5H-imidazo[1,2-a]azepin-9-yl)-2'-oxo-1',2'-dihydrospiro[piperidine-4,4'-pyrido[2,3-d][1,3]oxazine]-1-carboxamide
SMILES Fc1cccc([C@@H]2CC[C@@H](NC(=O)N3CCC4(CC3)OC(=O)Nc3ncccc43)c3ncc(CC(F)(F)F)n3C2)c1F
InChI Key InChIKey=FTBFNPPRRNOXIX-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 2 hits for monomerid = 50342630
Found 2 hits for monomerid = 50342630
TargetCalcitonin gene-related peptide type 1 receptor(Human)
Merck Research Laboratories
Curated by ChEMBL
Merck Research Laboratories
Curated by ChEMBL
Affinity DataKi: 0.0700nMAssay Description:Displacement of [125I]-CGRP from human recombinant CGRP receptorMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Human)
Merck Research Laboratories
Curated by ChEMBL
Merck Research Laboratories
Curated by ChEMBL
Affinity DataIC50: 3.30E+3nMAssay Description:Inhibition of human ERGMore data for this Ligand-Target Pair