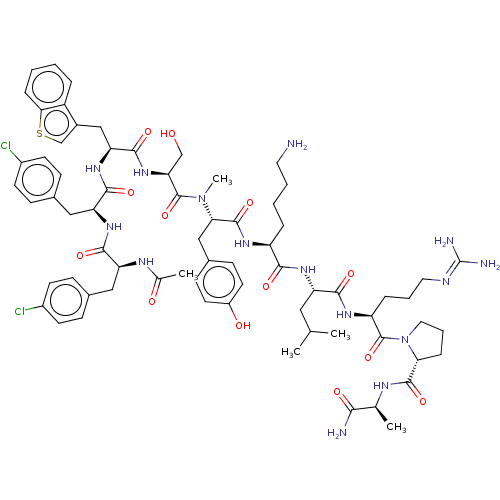
BDBM50469858 CHEMBL268397
SMILES [#6]-[#6](-[#6])-[#6]-[#6@H](-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#7]-[#6](=O)-[#6@H](-[#6]-c1ccc(-[#8])cc1)-[#7](-[#6])-[#6](=O)-[#6@H](-[#6]-[#8])-[#7]-[#6](=O)-[#6@H](-[#6]-c1csc2ccccc12)-[#7]-[#6](=O)-[#6@H](-[#6]-c1ccc(Cl)cc1)-[#7]-[#6](=O)-[#6@H](-[#6]-c1ccc(Cl)cc1)-[#7]-[#6](-[#6])=O)-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6](=O)-[#7]-1-[#6]-[#6]-[#6]-[#6@@H]-1-[#6](=O)-[#7]-[#6@@H](-[#6])-[#6](-[#7])=O
InChI Key InChIKey=OAOCKSGJCWTPMY-UHFFFAOYSA-N
Data 1 KI
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 1 hit for monomerid = 50469858
Found 1 hit for monomerid = 50469858
Affinity DataKi: 0.0234nMAssay Description:The negative logarithm of the concentration of antagonist that inhibits 50% of the binding of 125 I-labeled leuprolide to the rat pituitary LHRH rece...More data for this Ligand-Target Pair