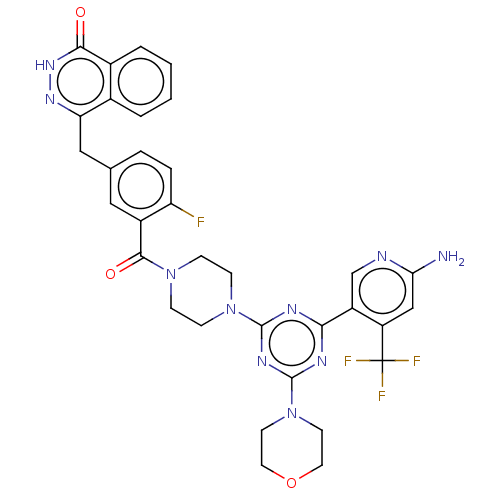
BDBM50520034 CHEMBL4541731
SMILES Nc1cc(c(cn1)-c1nc(nc(n1)N1CCN(CC1)C(=O)c1cc(Cc2n[nH]c(=O)c3ccccc23)ccc1F)N1CCOCC1)C(F)(F)F
InChI Key InChIKey=QEEHFLKQODURKT-UHFFFAOYSA-N
Data 4 IC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 4 hits for monomerid = 50520034
Found 4 hits for monomerid = 50520034
Affinity DataIC50: 9.5nMAssay Description:Inhibition of recombinant human PARP1 using histone as substrate after 1 hr in presence of biotinylated NAD+ by ELISAMore data for this Ligand-Target Pair
Affinity DataIC50: 9.60nMAssay Description:Inhibition of PARP1 (unknown origin) incubated for 1 hr by chemiluminescence assayMore data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform(Human)
Hubei Polytechnic University
Curated by ChEMBL
Hubei Polytechnic University
Curated by ChEMBL
Affinity DataIC50: 82nMAssay Description:Inhibition of PI3K-alpha (unknown origin) incubated for 40 mins in presence of ATP by ELISAMore data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform(Human)
Hubei Polytechnic University
Curated by ChEMBL
Hubei Polytechnic University
Curated by ChEMBL
Affinity DataIC50: 170nMAssay Description:Inhibition of PI3Kalpha (unknown origin) using PIP2 as substrate measured after 1 hr by ADP-glo plus luminescence assayMore data for this Ligand-Target Pair