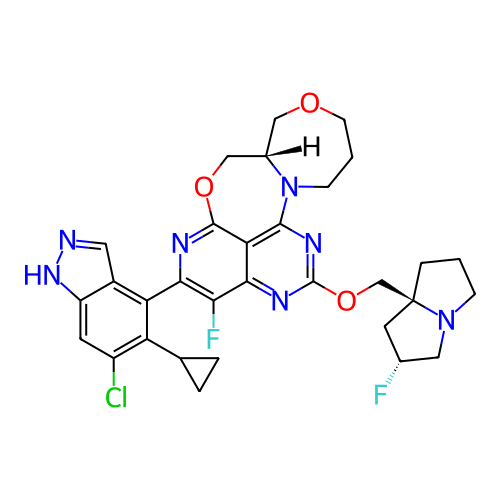
BDBM717613 (General Method A): (8aS)-5-(6-chloro-5-cyclopropyl-1H-indazol-4-yl)-4-fluoro-2-{[(2R,7aS)-2-fluorotetrahydro-1H-pyrrolizin-7a(5H)-yl]methoxy}-8a,9,12,13-tetrahydro-8H,11H-7,10-dioxa-1,3,6,13a-tetraazanaphtho[1,8-ab]heptalene::US12466840, Example Ex-3::US20250034173, Example 1
SMILES Fc1c(-c2c(C3CC3)c(Cl)cc3[nH]ncc23)nc2c3c(nc(OC[C@@]45CCCN4C[C@H](F)C5)nc13)N1CCCOC[C@H]1CO2
InChI Key InChIKey=FWEYEIDZVXMPKP-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 13 hits for monomerid = 717613
Found 13 hits for monomerid = 717613
Affinity DataKd: 0.0310nMAssay Description:Binding measurements were performed in parallel sets of either WT/G12D/G12C/G12V KRAS or WT K/H/N RAS proteins in GDP and/or GTPγS-loaded forms.Biaco...More data for this Ligand-Target Pair
Affinity DataKd: 0.0380nMAssay Description:Binding measurements were performed in parallel sets of either WT/G12D/G12C/G12V KRAS or WT K/H/N RAS proteins in GDP and/or GTPγS-loaded forms.Biaco...More data for this Ligand-Target Pair
Affinity DataKd: 0.0480nMAssay Description:Binding measurements were performed in parallel sets of either WT/G12D/G12C/G12V KRAS or WT K/H/N RAS proteins in GDP and/or GTPγS-loaded forms.Biaco...More data for this Ligand-Target Pair
Affinity DataKd: 0.105nMAssay Description:Binding measurements were performed in parallel sets of either WT/G12D/G12C/G12V KRAS or WT K/H/N RAS proteins in GDP and/or GTPγS-loaded forms.Biaco...More data for this Ligand-Target Pair
Affinity DataIC50: 1.30nMAssay Description:To assemble the preformed TR-FRET complexes, each biotinylated RAS protein is diluted to 2 μM in an EDTA Buffer (20 mM HEPES pH 7.5, 50 mM sodium chl...More data for this Ligand-Target Pair
Affinity DataIC50: 2.5nMAssay Description:To assemble the preformed TR-FRET complexes, each biotinylated RAS protein is diluted to 2 μM in an EDTA Buffer (20 mM HEPES pH 7.5, 50 mM sodium chl...More data for this Ligand-Target Pair
Affinity DataIC50: 3nMAssay Description:To assemble the preformed TR-FRET complexes, each biotinylated RAS protein is diluted to 2 μM in an EDTA Buffer (20 mM HEPES pH 7.5, 50 mM sodium chl...More data for this Ligand-Target Pair
Affinity DataKd: 8nMAssay Description:Binding measurements were performed in parallel sets of either WT/G12D/G12C/G12V KRAS or WT K/H/N RAS proteins in GDP and/or GTPγS-loaded forms.Biaco...More data for this Ligand-Target Pair
Affinity DataKd: 10nMAssay Description:Binding measurements were performed in parallel sets of either WT/G12D/G12C/G12V KRAS or WT K/H/N RAS proteins in GDP and/or GTPγS-loaded forms.Biaco...More data for this Ligand-Target Pair
Affinity DataKd: 12nMAssay Description:Binding measurements were performed in parallel sets of either WT/G12D/G12C/G12V KRAS or WT K/H/N RAS proteins in GDP and/or GTPγS-loaded forms.Biaco...More data for this Ligand-Target Pair
Affinity DataKd: 14nMAssay Description:Binding measurements were performed in parallel sets of either WT/G12D/G12C/G12V KRAS or WT K/H/N RAS proteins in GDP and/or GTPγS-loaded forms.Biaco...More data for this Ligand-Target Pair
Affinity DataKd: >1.00E+4nMAssay Description:Binding measurements were performed in parallel sets of either WT/G12D/G12C/G12V KRAS or WT K/H/N RAS proteins in GDP and/or GTPγS-loaded forms.Biaco...More data for this Ligand-Target Pair
Affinity DataKd: >1.00E+4nMAssay Description:Binding measurements were performed in parallel sets of either WT/G12D/G12C/G12V KRAS or WT K/H/N RAS proteins in GDP and/or GTPγS-loaded forms.Biaco...More data for this Ligand-Target Pair