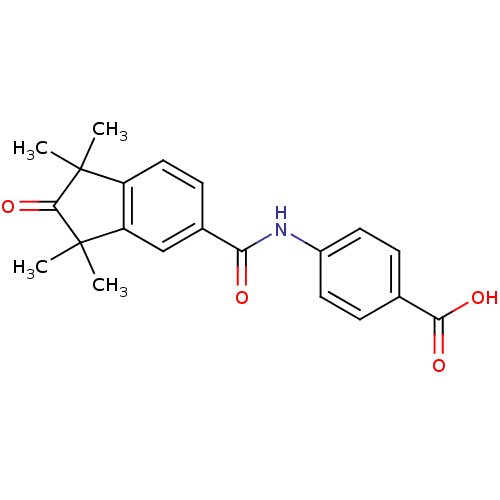
BDBM36810 BMS753::US9963439, BMS753
SMILES CC1(C)C(=O)C(C)(C)c2cc(ccc12)C(=O)Nc1ccc(cc1)C(O)=O
InChI Key InChIKey=KFBPBWUZXBYJDG-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 4 hits for monomerid = 36810
Found 4 hits for monomerid = 36810
TargetRetinoic acid receptor alpha [200-419]/gamma [183-417](Human)
Institute of Genetics and Molecular and Cellular Biology (Igbmc)
Institute of Genetics and Molecular and Cellular Biology (Igbmc)
Affinity DataKi: 1nM ΔG°: -11.4kcal/molepH: 8.0 T: 2°CAssay Description:Competitive assay were perform with 5nM tritiated all-trans retinoic acid (t-RA; 5nM) with or without 100-fold excess of non-radioactive t-RA (500nM)...More data for this Ligand-Target Pair
TargetRetinoic acid receptor alpha [200-419](Human)
Institute of Genetics and Molecular and Cellular Biology (Igbmc)
Institute of Genetics and Molecular and Cellular Biology (Igbmc)
Affinity DataKi: 2nM ΔG°: -11.0kcal/molepH: 8.0 T: 2°CAssay Description:Competitive assay were perform with 5nM tritiated all-trans retinoic acid (t-RA; 5nM) with or without 100-fold excess of non-radioactive t-RA (500nM)...More data for this Ligand-Target Pair
TargetCytochrome P450 3A4(Human)
University of Washington Through Its Center For Commercialization
US Patent
University of Washington Through Its Center For Commercialization
US Patent
Affinity DataIC50: 2.90E+4nMAssay Description:Compounds were assessed for inhibition (IC50, n=2) of CYP2C8, CYP2C9 and CYP3A4 in pooled human liver microsomes using selective probe substrates at ...More data for this Ligand-Target Pair
TargetCytochrome P450 26A1(Human)
University of Washington Through Its Center For Commercialization
US Patent
University of Washington Through Its Center For Commercialization
US Patent
Affinity DataIC50: 4.02E+4nMAssay Description:Eighteen compounds were tested as potential inhibitors of CYP26A1 and CYP26B1. The formation of 9-cis-4-OH-RA metabolite was monitored and the percen...More data for this Ligand-Target Pair