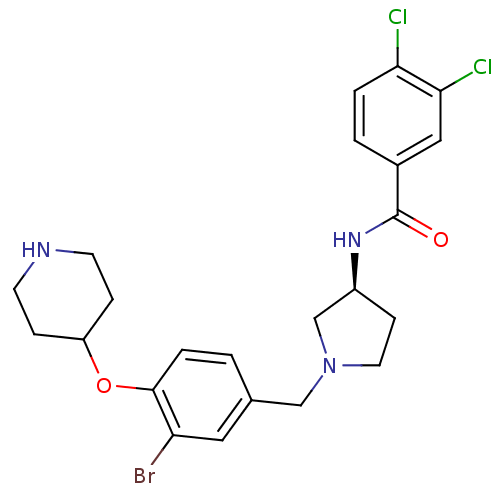
BDBM50252659 (S)-N-(1-(3-bromo-4-(piperidin-4-yloxy)benzyl)pyrrolidin-3-yl)-3,4-dichlorobenzamide::CHEMBL495075
SMILES Clc1ccc(cc1Cl)C(=O)N[C@H]1CCN(Cc2ccc(OC3CCNCC3)c(Br)c2)C1
InChI Key InChIKey=SXVZDDYARZWATR-KRWDZBQOSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 13 hits for monomerid = 50252659
Found 13 hits for monomerid = 50252659
Affinity DataKi: 4nMAssay Description:Displacement of [I125]hU2 from human recombinant urotensin 2 receptor expressed in HEK293 cellsMore data for this Ligand-Target Pair
Affinity DataKi: 38nMAssay Description:Binding affinity to 5HT1A receptorMore data for this Ligand-Target Pair
Affinity DataKi: 85nMAssay Description:Binding affinity at 5HT2C receptorMore data for this Ligand-Target Pair
Affinity DataKi: 100nMAssay Description:Binding affinity to dopamine D2 receptorMore data for this Ligand-Target Pair
Affinity DataKi: 140nMAssay Description:Binding affinity to 5HT1D receptorMore data for this Ligand-Target Pair
Affinity DataKi: 140nMAssay Description:Binding affinity at 5HT2B receptorMore data for this Ligand-Target Pair
Affinity DataKi: 4.10E+3nMAssay Description:Binding affinity to 5HT6 receptorMore data for this Ligand-Target Pair
Affinity DataKi: 7.20E+3nMAssay Description:Binding affinity to 5HT1F receptorMore data for this Ligand-Target Pair
Affinity DataKi: >1.00E+4nMAssay Description:Binding affinity to beta 2 adrenergic receptorMore data for this Ligand-Target Pair
Affinity DataKi: >1.00E+4nMAssay Description:Binding affinity to 5HT1E receptorMore data for this Ligand-Target Pair
Affinity DataIC50: 4nMAssay Description:Antagonist activity at human recombinant urotensin 2 receptor expressed in HEK293 cells assessed as inhibition of urotensin 2-induced calcium mobiliz...More data for this Ligand-Target Pair
Affinity DataIC50: 1.90E+3nMAssay Description:Inhibition of CYP3A4More data for this Ligand-Target Pair
Affinity DataIC50: 2.20E+3nMAssay Description:Inhibition of CYP2D6More data for this Ligand-Target Pair