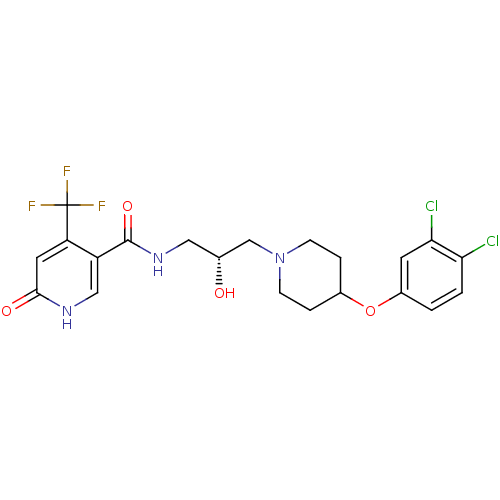
BDBM50402108 CHEMBL2207662
SMILES O[C@H](CNC(=O)c1c[nH]c(=O)cc1C(F)(F)F)CN1CCC(CC1)Oc1ccc(Cl)c(Cl)c1
InChI Key InChIKey=SKXJKSJHHFLVAP-GFCCVEGCSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 4 hits for monomerid = 50402108
Found 4 hits for monomerid = 50402108
Affinity DataKi: 3.16nMAssay Description:Displacement of [3H]-4-(2,4-dichloro-3-methylphenoxy)-l'-[4-(methylsulfonyl)benzoyl]-l,4'-bipiperidine from human recombinant CCR3 expressed in CHOK1...More data for this Ligand-Target Pair
Affinity DataKi: 3.98nMAssay Description:Displacement of [3H]-pyrilamine from human recombinant H1 histamine receptor expressed in CHOK1 cells after 1 hrMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
Astrazeneca
Curated by ChEMBL
Astrazeneca
Curated by ChEMBL
Affinity DataIC50: 5.01E+3nMAssay Description:Displacement of 3,7-Bis[2-(4-nitro[3,5]-[3H]phenyl)ethyl]-3,7-diazabicyclo[3.3.1]nonane from human ERG expressed in HEK cells after 3hrsMore data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Homo sapiens (Human))
Astrazeneca
Curated by ChEMBL
Astrazeneca
Curated by ChEMBL
Affinity DataIC50: 1.00E+4nMAssay Description:Inhibition of human ERG expressed in HEK cells by ion flux assayMore data for this Ligand-Target Pair