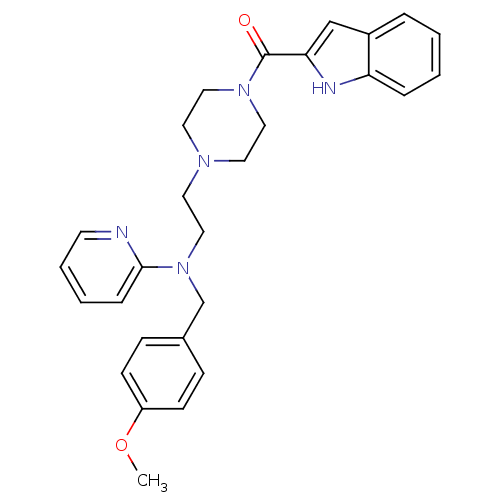
BDBM50419329 CHEMBL1910378
SMILES COc1ccc(CN(CCN2CCN(CC2)C(=O)c2cc3ccccc3[nH]2)c2ccccn2)cc1
InChI Key InChIKey=LAKQJOXTAMPPOO-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 4 hits for monomerid = 50419329
Found 4 hits for monomerid = 50419329
Affinity DataKi: 170nMAssay Description:Displacement of [3H]mepyramine from human H1R expressed in Sf9 cells co-expressing RGS4 after 90 mins by liquid scintillation countingMore data for this Ligand-Target Pair
Affinity DataKi: 5.89E+3nMAssay Description:Displacement of [3H]histamine from human H4R expressed in Sf9 cells co-expressing RGS19, Galphai2 and Gbeta1gamma2 after 60 mins by liquid scintillat...More data for this Ligand-Target Pair
Affinity DataEC50: 1.05E+5nMAssay Description:Partial agonist activity at human H4R expressed in Sf9 cells co-expressing RGS19, Galphai2 and Gbeta1gamma2 assessed as stimulation of [35S]GTPgammaS...More data for this Ligand-Target Pair
TargetHistamine H1 receptor(Cavia porcellus (domestic guinea pig))
University Of Regensburg
Curated by ChEMBL
University Of Regensburg
Curated by ChEMBL
Affinity DataKd: 10.7nMAssay Description:Antagonist activity at H1R in guinea pig ileum assessed as inhibition of histamine-induced muscle contraction after 15 minsMore data for this Ligand-Target Pair