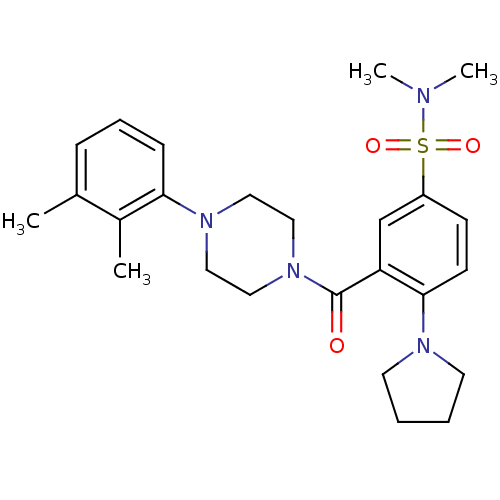
BDBM61095 3-[4-(2,3-dimethylphenyl)piperazin-1-yl]carbonyl-N,N-dimethyl-4-pyrrolidin-1-yl-benzenesulfonamide::3-[4-(2,3-dimethylphenyl)piperazine-1-carbonyl]-N,N-dimethyl-4-pyrrolidin-1-ylbenzenesulfonamide::3-[4-(2,3-dimethylphenyl)piperazine-1-carbonyl]-N,N-dimethyl-4-pyrrolidino-benzenesulfonamide::3-[[4-(2,3-dimethylphenyl)-1-piperazinyl]-oxomethyl]-N,N-dimethyl-4-(1-pyrrolidinyl)benzenesulfonamide::MLS001002737::SMR000369190::cid_2440433
SMILES CN(C)S(=O)(=O)c1ccc(N2CCCC2)c(c1)C(=O)N1CCN(CC1)c1cccc(C)c1C
InChI Key InChIKey=VRSJAHQGJHDACS-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 7 hits for monomerid = 61095
Found 7 hits for monomerid = 61095
TargetG-protein coupled receptor 55(Homo sapiens (Human))
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Affinity DataEC50: 508nMAssay Description:Data Source: Sanford-Burnham Center for Chemical Genomics (SBCCG) Source Affiliation: Sanford-Burnham Medical Research Institute (SBMRI, San Diego, C...More data for this Ligand-Target Pair
TargetG-protein coupled receptor 35(Homo sapiens (Human))
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Affinity DataIC50: 3.20E+4nMAssay Description:Data Source: Sanford-Burnham Center for Chemical Genomics Source Affiliation: Sanford-Burnham Medical Research Institute Network: NIH Molecular Lib...More data for this Ligand-Target Pair
TargetCannabinoid receptor 2(Homo sapiens (Human))
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Affinity DataIC50: 1.51E+4nMAssay Description:Data Source: Sanford-Burnham Center for Chemical Genomics Source Affiliation: Sanford-Burnham Medical Research Institute (SBMRI, San Diego, CA) Netw...More data for this Ligand-Target Pair
TargetCannabinoid receptor 2(Homo sapiens (Human))
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Affinity DataEC50: >3.00E+4nMAssay Description:Agonist activity at human CB2 receptor expressed in U2OS cells assessed as increase in GFP-tagged beta arrestin recruitment after 75 mins by fluoresc...More data for this Ligand-Target Pair
TargetG-protein coupled receptor 55(Homo sapiens (Human))
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Affinity DataEC50: 260nMAssay Description:Agonist activity at HA-epitope-tagged variant of GPR55E with serine enhanced C terminus (unknown origin) expressed in human U2OS cells assessed as in...More data for this Ligand-Target Pair
TargetCannabinoid receptor 1(Homo sapiens (Human))
University Of North Carolina At Greensboro
Curated by ChEMBL
University Of North Carolina At Greensboro
Curated by ChEMBL
Affinity DataEC50: >3.00E+4nMAssay Description:Agonist activity at human brain CB1 receptor expressed in U2OS cells assessed as increase in beta arrestin recruitment after 60 mins by fluorescence ...More data for this Ligand-Target Pair
TargetCannabinoid receptor 2(Homo sapiens (Human))
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Burnham Center For Chemical Genomics
Curated by PubChem BioAssay
Affinity DataEC50: >3.20E+4nMAssay Description:Data Source: Sanford-Burnham Center for Chemical Genomics Source Affiliation: Sanford-Burnham Medical Research Institute (SBMRI, San Diego, CA) Net...More data for this Ligand-Target Pair