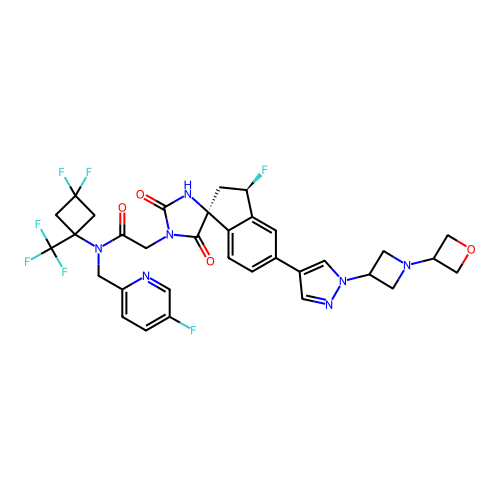
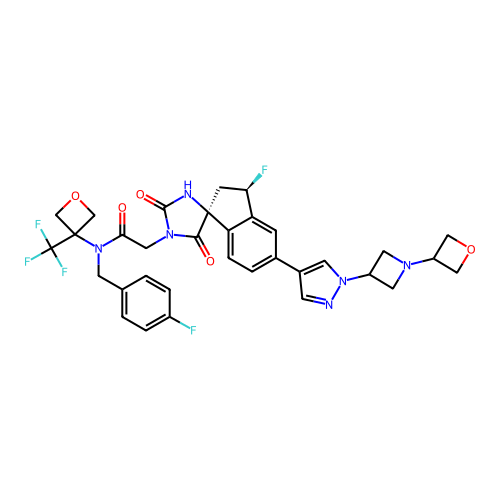
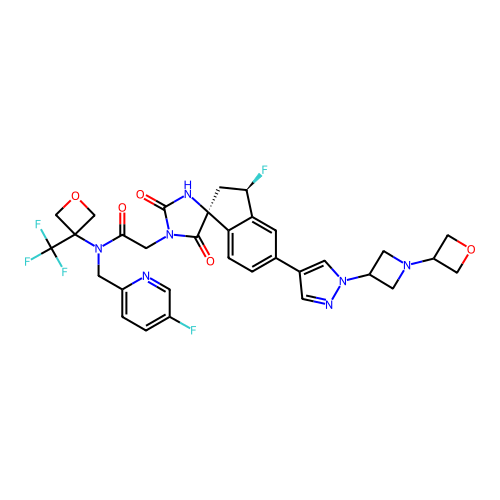
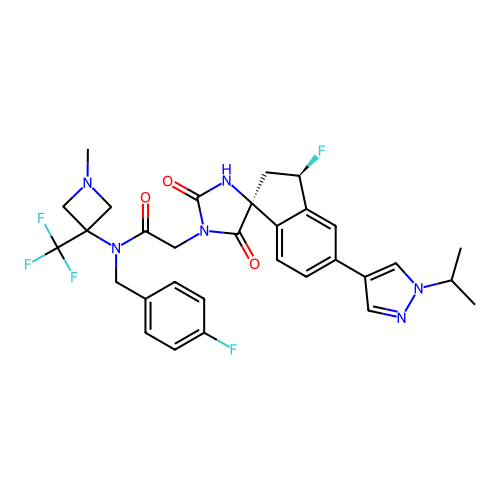
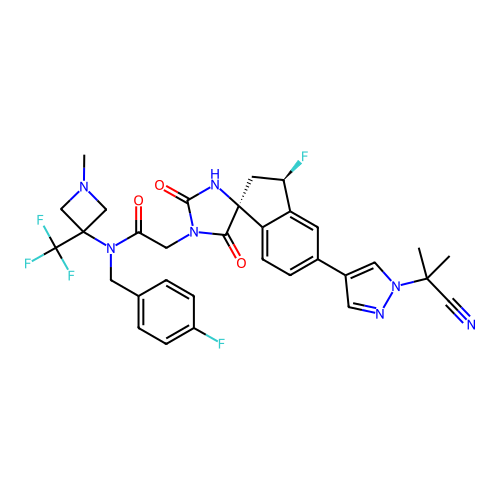
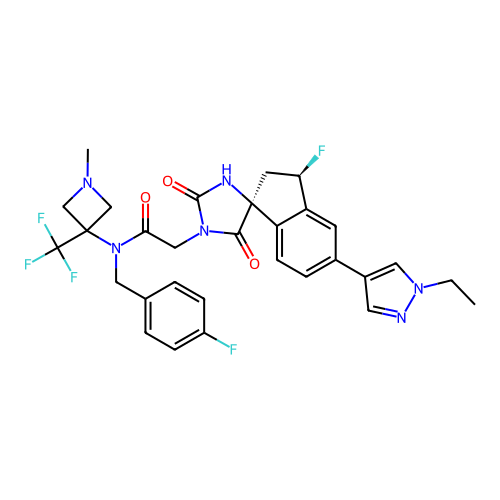
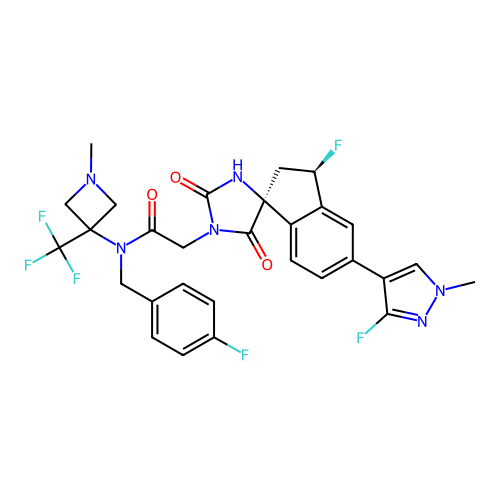
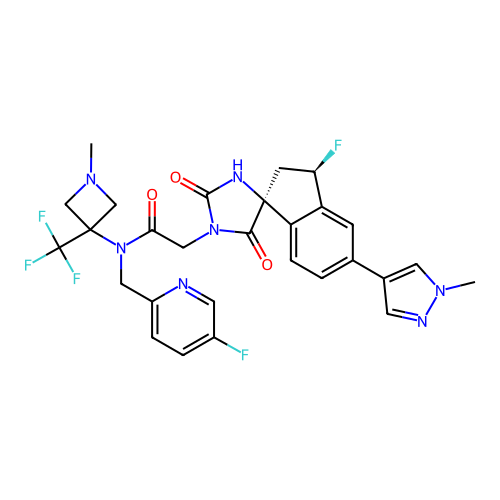
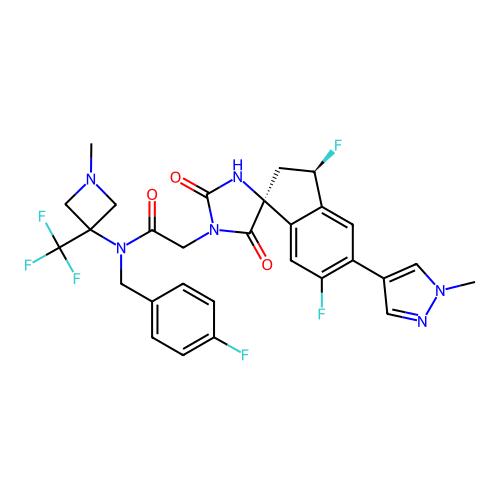
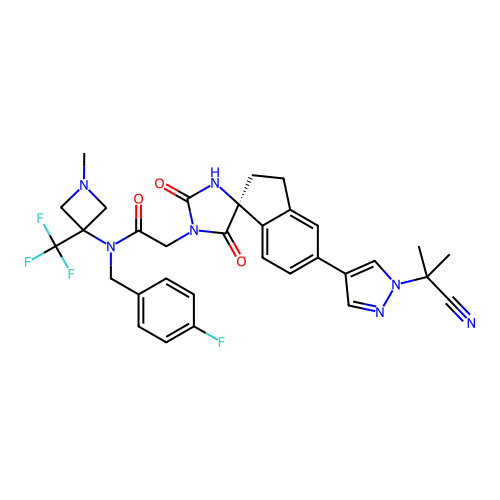
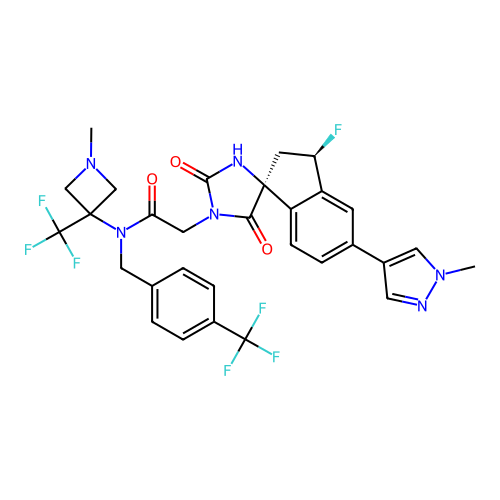
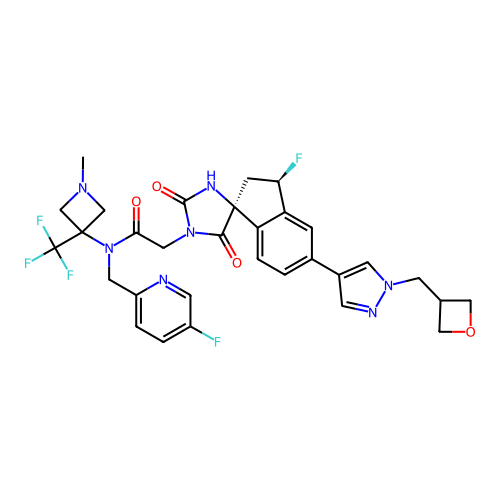
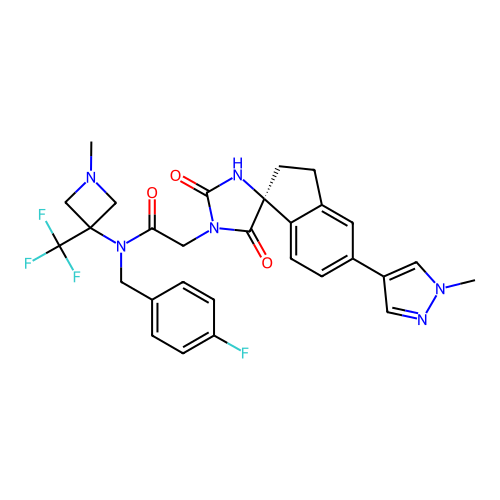
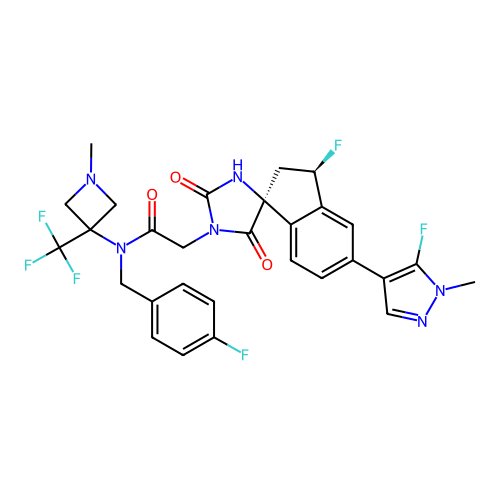
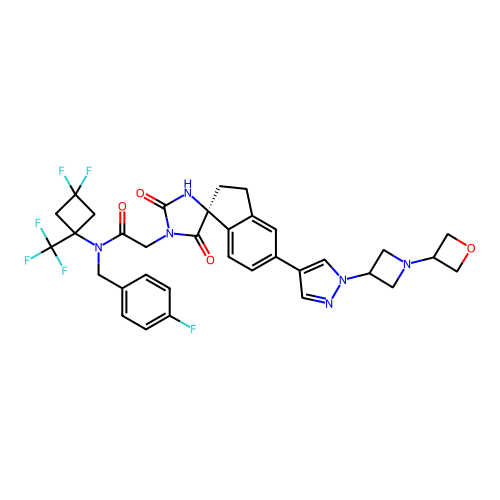
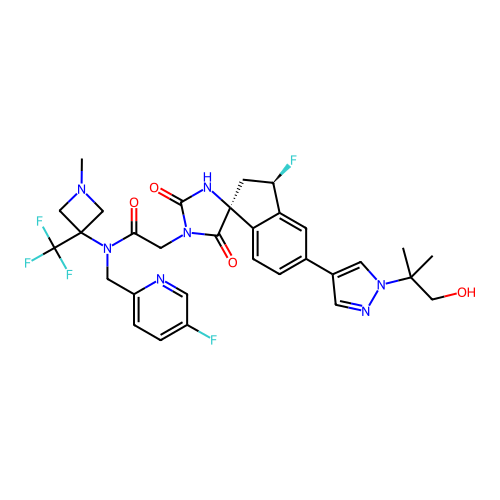
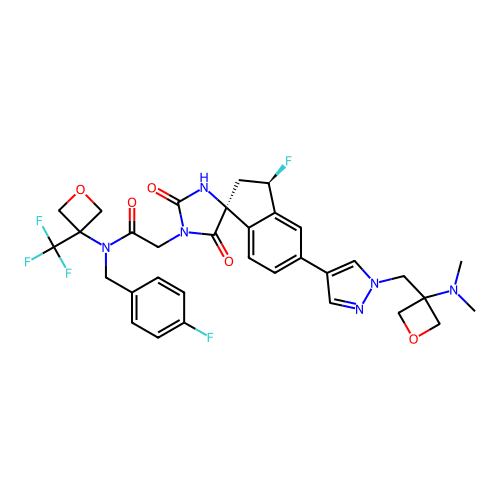
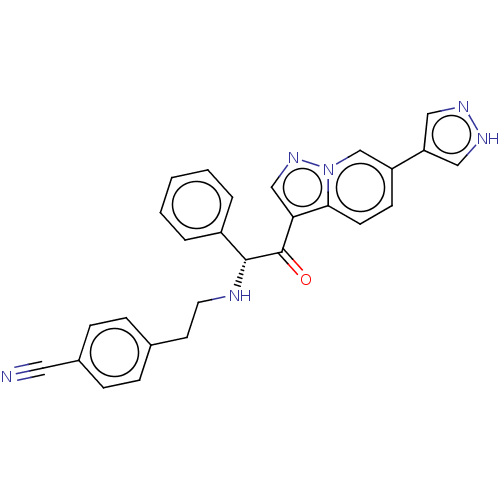
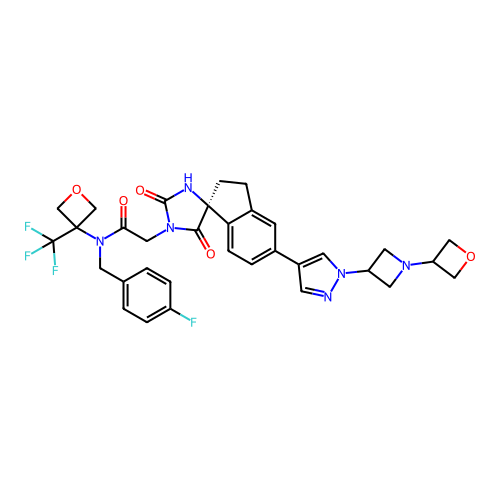
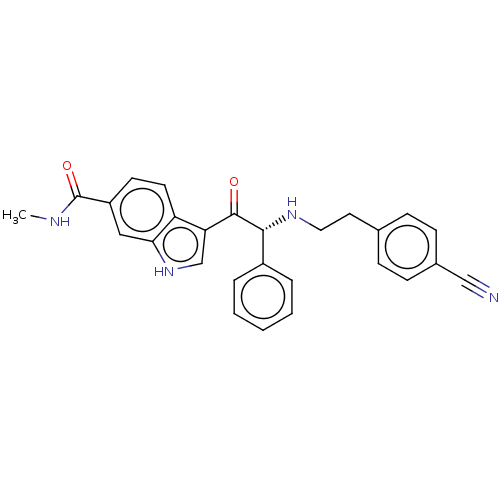
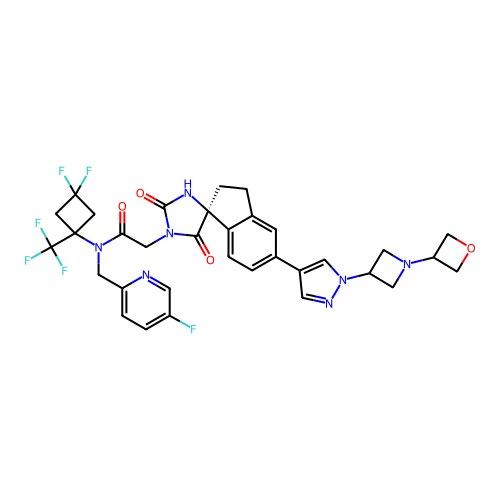
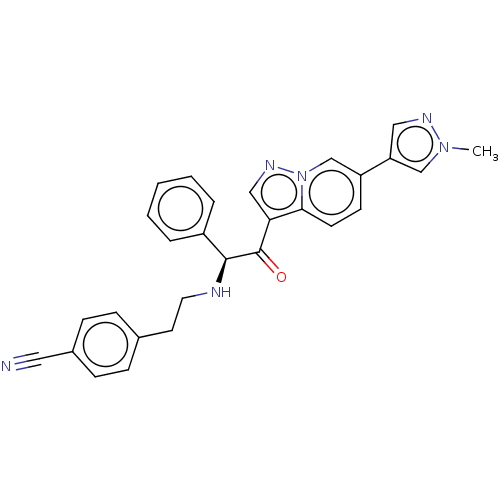
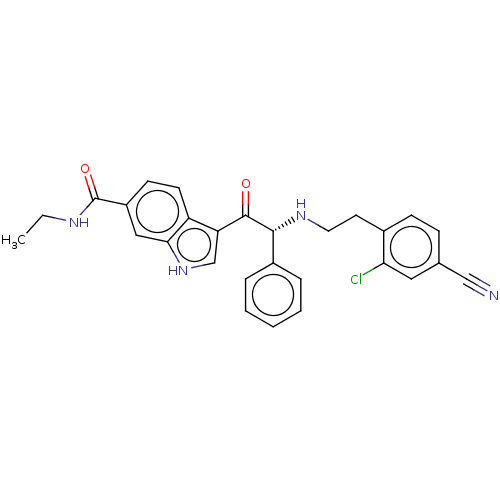
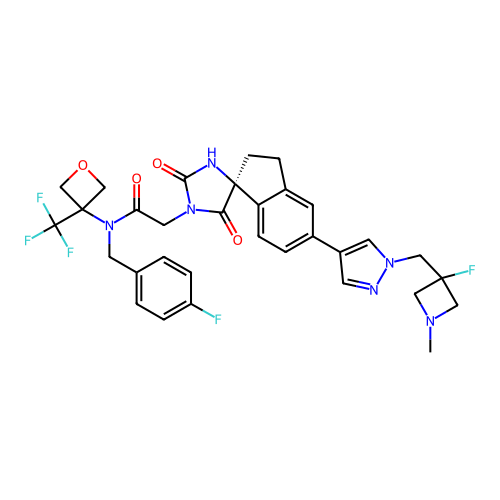
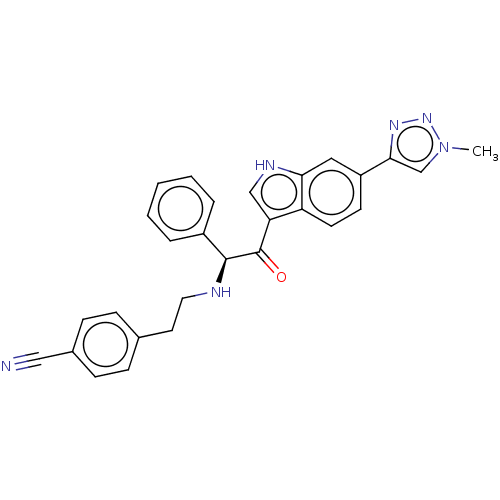
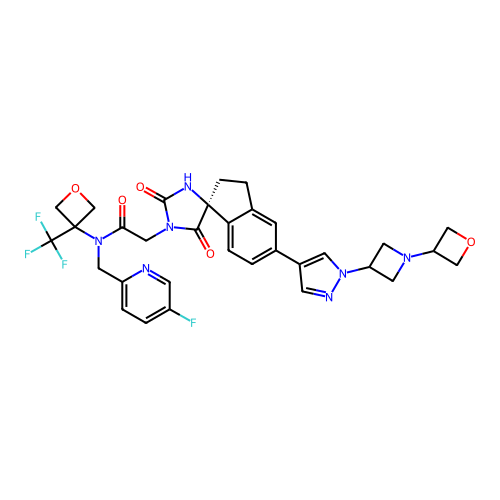
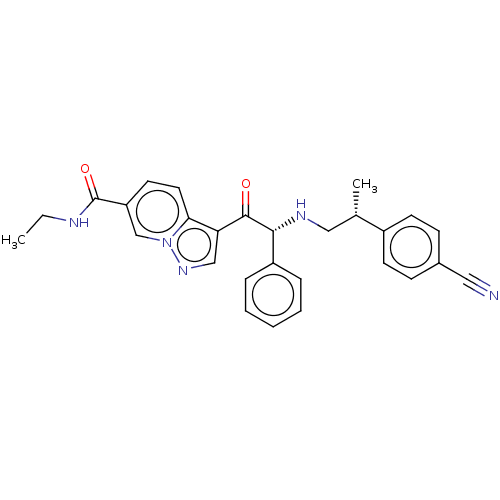
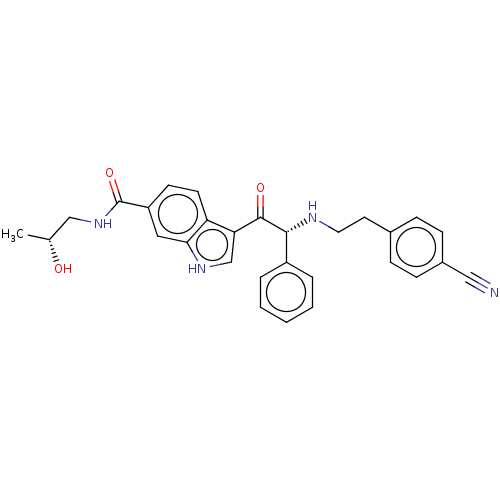
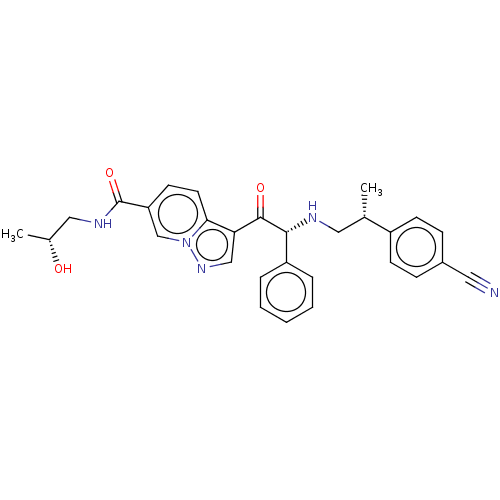
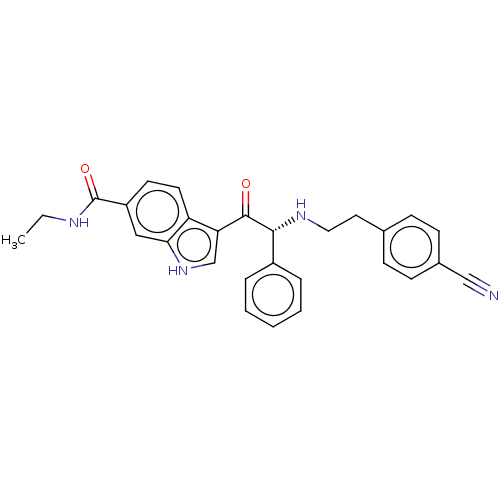
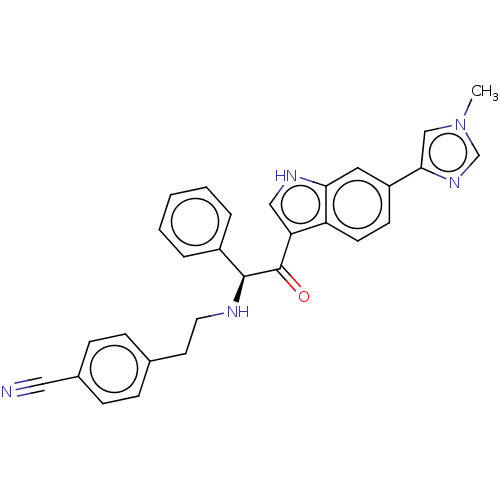
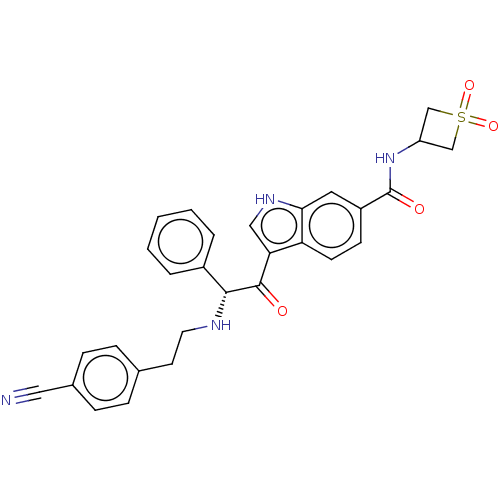
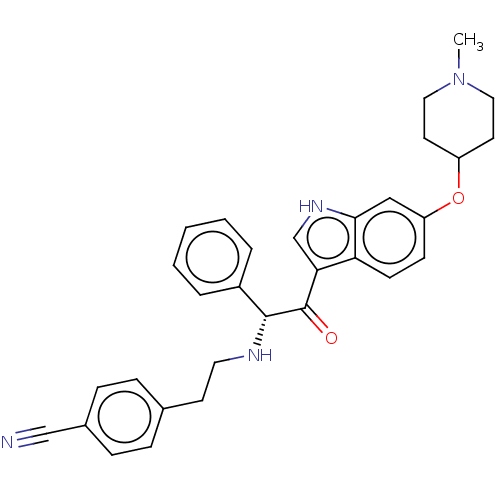
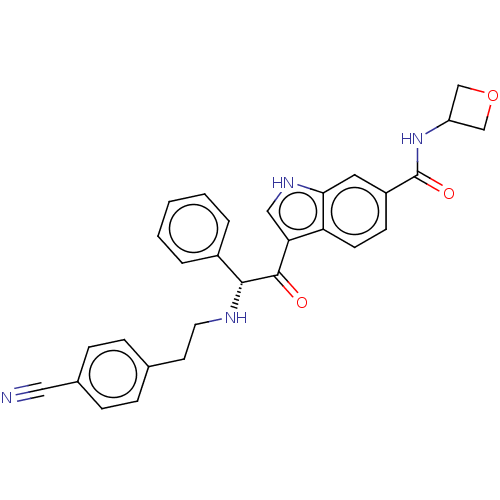
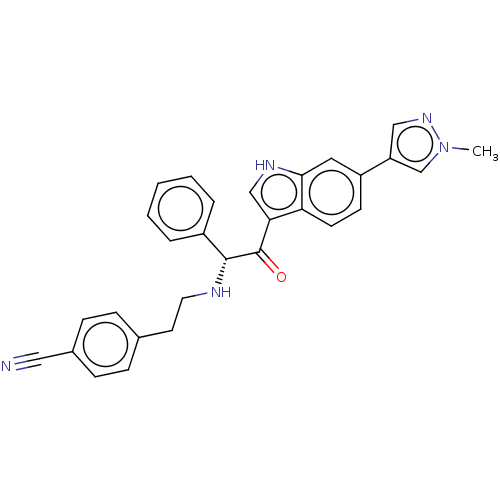
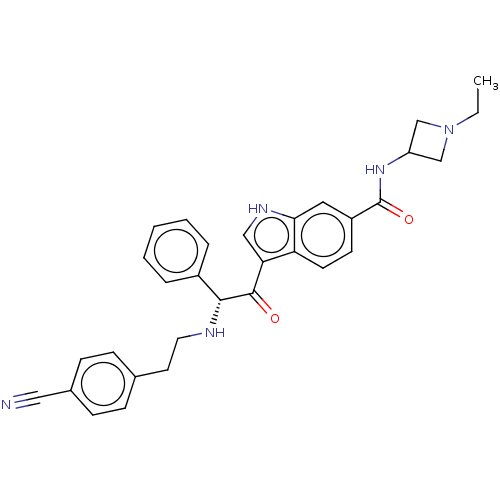
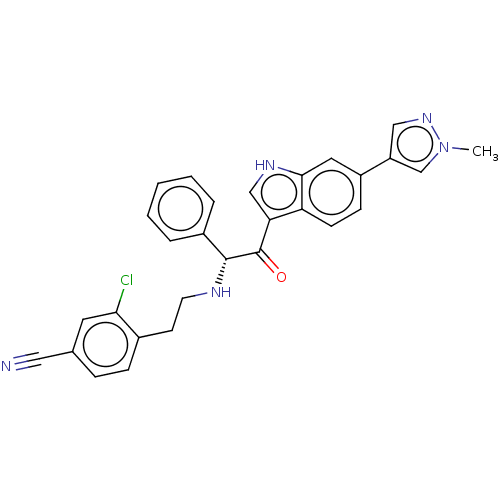
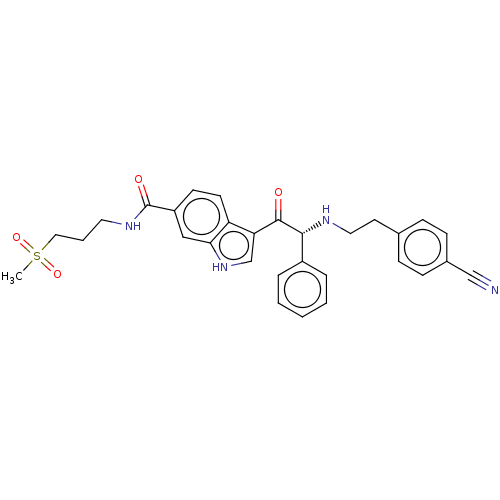
Report error Found 425 Enz. Inhib. hit(s) with Target = 'Histone acetyltransferase p300 [1287-1666]/Histone acetyltransferase type B catalytic subunit'
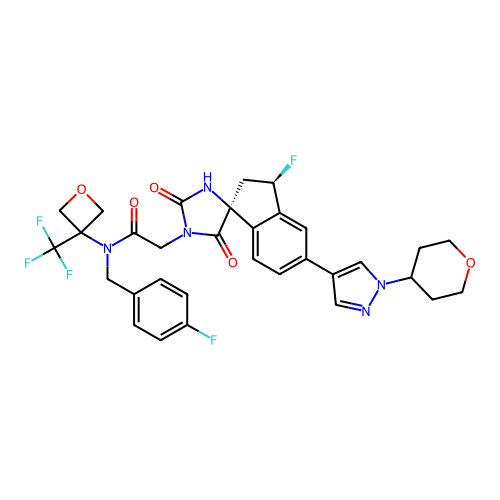
TargetHistone acetyltransferase p300 [1287-1666]/Histone acetyltransferase type B catalytic subunit(Human)
Sumitomo Pharma
US Patent
Sumitomo Pharma
US Patent
Affinity DataIC50: 0.200nMAssay Description:The HAT activity inhibitory ability of HAT inhibitors was evaluated using SensoLyte HAT(p300) Assay Kit (ANASPEC, AS-72172). Specifically, 7.5 μL of ...More data for this Ligand-Target Pair
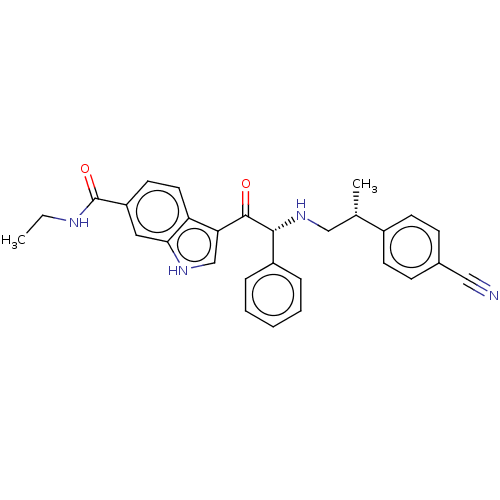
TargetHistone acetyltransferase p300 [1287-1666]/Histone acetyltransferase type B catalytic subunit(Human)
Sumitomo Pharma
US Patent
Sumitomo Pharma
US Patent
Affinity DataIC50: 0.200nMAssay Description:The HAT activity inhibitory ability of HAT inhibitors was evaluated using SensoLyte HAT(p300) Assay Kit (ANASPEC, AS-72172). Specifically, 7.5 μL of ...More data for this Ligand-Target Pair
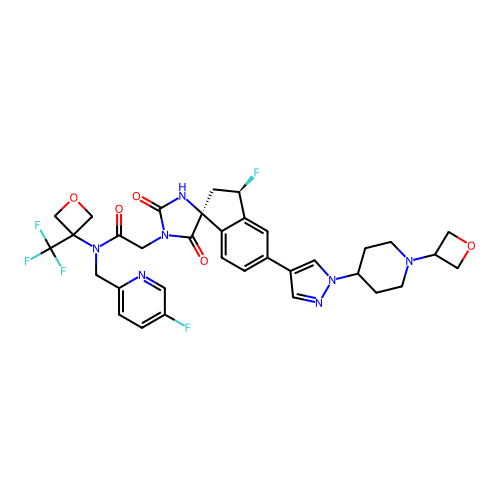
TargetHistone acetyltransferase p300 [1287-1666]/Histone acetyltransferase type B catalytic subunit(Human)
Sumitomo Pharma
US Patent
Sumitomo Pharma
US Patent
Affinity DataIC50: 0.400nMAssay Description:The HAT activity inhibitory ability of HAT inhibitors was evaluated using SensoLyte HAT(p300) Assay Kit (ANASPEC, AS-72172). Specifically, 7.5 μL of ...More data for this Ligand-Target Pair
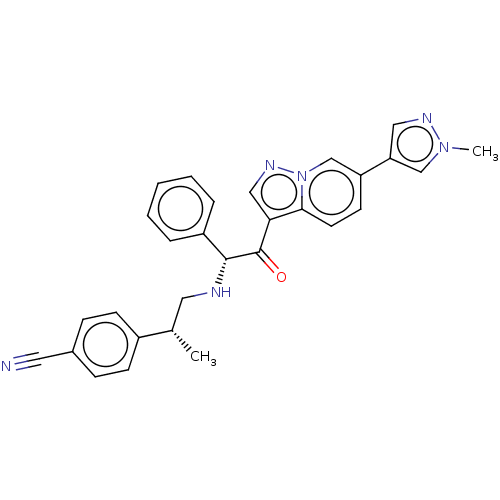
TargetHistone acetyltransferase p300 [1287-1666]/Histone acetyltransferase type B catalytic subunit(Human)
Sumitomo Pharma
US Patent
Sumitomo Pharma
US Patent
Affinity DataIC50: 0.400nMAssay Description:The HAT activity inhibitory ability of HAT inhibitors was evaluated using SensoLyte HAT(p300) Assay Kit (ANASPEC, AS-72172). Specifically, 7.5 μL of ...More data for this Ligand-Target Pair
TargetHistone acetyltransferase p300 [1287-1666]/Histone acetyltransferase type B catalytic subunit(Human)
Sumitomo Pharma
US Patent
Sumitomo Pharma
US Patent
Affinity DataIC50: 0.5nMAssay Description:The HAT activity inhibitory ability of HAT inhibitors was evaluated using SensoLyte HAT(p300) Assay Kit (ANASPEC, AS-72172). Specifically, 7.5 μL of ...More data for this Ligand-Target Pair
TargetHistone acetyltransferase p300 [1287-1666]/Histone acetyltransferase type B catalytic subunit(Human)
Sumitomo Pharma
US Patent
Sumitomo Pharma
US Patent
Affinity DataIC50: 0.600nMAssay Description:The HAT activity inhibitory ability of HAT inhibitors was evaluated using SensoLyte HAT(p300) Assay Kit (ANASPEC, AS-72172). Specifically, 7.5 μL of ...More data for this Ligand-Target Pair
TargetHistone acetyltransferase p300 [1287-1666]/Histone acetyltransferase type B catalytic subunit(Human)
Sumitomo Pharma
US Patent
Sumitomo Pharma
US Patent
Affinity DataIC50: 0.600nMAssay Description:The HAT activity inhibitory ability of HAT inhibitors was evaluated using SensoLyte HAT(p300) Assay Kit (ANASPEC, AS-72172). Specifically, 7.5 μL of ...More data for this Ligand-Target Pair
TargetHistone acetyltransferase p300 [1287-1666]/Histone acetyltransferase type B catalytic subunit(Human)
Sumitomo Pharma
US Patent
Sumitomo Pharma
US Patent
Affinity DataIC50: 0.900nMAssay Description:The HAT activity inhibitory ability of HAT inhibitors was evaluated using SensoLyte HAT(p300) Assay Kit (ANASPEC, AS-72172). Specifically, 7.5 μL of ...More data for this Ligand-Target Pair
TargetHistone acetyltransferase p300 [1287-1666]/Histone acetyltransferase type B catalytic subunit(Human)
Sumitomo Pharma
US Patent
Sumitomo Pharma
US Patent
Affinity DataIC50: 1nMAssay Description:The HAT activity inhibitory ability of HAT inhibitors was evaluated using SensoLyte HAT(p300) Assay Kit (ANASPEC, AS-72172). Specifically, 7.5 μL of ...More data for this Ligand-Target Pair
TargetHistone acetyltransferase p300 [1287-1666]/Histone acetyltransferase type B catalytic subunit(Human)
Sumitomo Pharma
US Patent
Sumitomo Pharma
US Patent
Affinity DataIC50: 2nMAssay Description:The HAT activity inhibitory ability of HAT inhibitors was evaluated using SensoLyte HAT(p300) Assay Kit (ANASPEC, AS-72172). Specifically, 7.5 μL of ...More data for this Ligand-Target Pair
TargetHistone acetyltransferase p300 [1287-1666]/Histone acetyltransferase type B catalytic subunit(Human)
Sumitomo Pharma
US Patent
Sumitomo Pharma
US Patent
Affinity DataIC50: 2nMAssay Description:The HAT activity inhibitory ability of HAT inhibitors was evaluated using SensoLyte HAT(p300) Assay Kit (ANASPEC, AS-72172). Specifically, 7.5 μL of ...More data for this Ligand-Target Pair
TargetHistone acetyltransferase p300 [1287-1666]/Histone acetyltransferase type B catalytic subunit(Human)
Sumitomo Pharma
US Patent
Sumitomo Pharma
US Patent
Affinity DataIC50: 2nMAssay Description:The HAT activity inhibitory ability of HAT inhibitors was evaluated using SensoLyte HAT(p300) Assay Kit (ANASPEC, AS-72172). Specifically, 7.5 μL of ...More data for this Ligand-Target Pair
TargetHistone acetyltransferase p300 [1287-1666]/Histone acetyltransferase type B catalytic subunit(Human)
Sumitomo Pharma
US Patent
Sumitomo Pharma
US Patent
Affinity DataIC50: 2nMAssay Description:The HAT activity inhibitory ability of HAT inhibitors was evaluated using SensoLyte HAT(p300) Assay Kit (ANASPEC, AS-72172). Specifically, 7.5 μL of ...More data for this Ligand-Target Pair
TargetHistone acetyltransferase p300 [1287-1666]/Histone acetyltransferase type B catalytic subunit(Human)
Sumitomo Pharma
US Patent
Sumitomo Pharma
US Patent
Affinity DataIC50: 3nMAssay Description:The HAT activity inhibitory ability of HAT inhibitors was evaluated using SensoLyte HAT(p300) Assay Kit (ANASPEC, AS-72172). Specifically, 7.5 μL of ...More data for this Ligand-Target Pair
TargetHistone acetyltransferase p300 [1287-1666]/Histone acetyltransferase type B catalytic subunit(Human)
Sumitomo Pharma
US Patent
Sumitomo Pharma
US Patent
Affinity DataIC50: 5nMAssay Description:The HAT activity inhibitory ability of HAT inhibitors was evaluated using SensoLyte HAT(p300) Assay Kit (ANASPEC, AS-72172). Specifically, 7.5 μL of ...More data for this Ligand-Target Pair
TargetHistone acetyltransferase p300 [1287-1666]/Histone acetyltransferase type B catalytic subunit(Human)
Sumitomo Pharma
US Patent
Sumitomo Pharma
US Patent
Affinity DataIC50: 5nMAssay Description:The HAT activity inhibitory ability of HAT inhibitors was evaluated using SensoLyte HAT(p300) Assay Kit (ANASPEC, AS-72172). Specifically, 7.5 μL of ...More data for this Ligand-Target Pair
TargetHistone acetyltransferase p300 [1287-1666]/Histone acetyltransferase type B catalytic subunit(Human)
Sumitomo Pharma
US Patent
Sumitomo Pharma
US Patent
Affinity DataIC50: 6nMAssay Description:The HAT activity inhibitory ability of HAT inhibitors was evaluated using SensoLyte HAT(p300) Assay Kit (ANASPEC, AS-72172). Specifically, 7.5 μL of ...More data for this Ligand-Target Pair
TargetHistone acetyltransferase p300 [1287-1666]/Histone acetyltransferase type B catalytic subunit(Human)
Sumitomo Pharma
US Patent
Sumitomo Pharma
US Patent
Affinity DataIC50: 6nMAssay Description:The HAT activity inhibitory ability of HAT inhibitors was evaluated using SensoLyte HAT(p300) Assay Kit (ANASPEC, AS-72172). Specifically, 7.5 μL of ...More data for this Ligand-Target Pair
TargetHistone acetyltransferase p300 [1287-1666]/Histone acetyltransferase type B catalytic subunit(Human)
Sumitomo Pharma
US Patent
Sumitomo Pharma
US Patent
Affinity DataIC50: 6nMAssay Description:The HAT activity inhibitory ability of HAT inhibitors was evaluated using SensoLyte HAT(p300) Assay Kit (ANASPEC, AS-72172). Specifically, 7.5 μL of ...More data for this Ligand-Target Pair
TargetHistone acetyltransferase p300 [1287-1666]/Histone acetyltransferase type B catalytic subunit(Human)
Sumitomo Pharma
US Patent
Sumitomo Pharma
US Patent
Affinity DataIC50: 6nMAssay Description:The HAT activity inhibitory ability of HAT inhibitors was evaluated using SensoLyte HAT(p300) Assay Kit (ANASPEC, AS-72172). Specifically, 7.5 μL of ...More data for this Ligand-Target Pair
TargetHistone acetyltransferase p300 [1287-1666]/Histone acetyltransferase type B catalytic subunit(Human)
Sumitomo Pharma
US Patent
Sumitomo Pharma
US Patent
Affinity DataIC50: 7nMAssay Description:The HAT activity inhibitory ability of HAT inhibitors was evaluated using SensoLyte HAT(p300) Assay Kit (ANASPEC, AS-72172). Specifically, 7.5 μL of ...More data for this Ligand-Target Pair
TargetHistone acetyltransferase p300 [1287-1666]/Histone acetyltransferase type B catalytic subunit(Human)
Sumitomo Pharma
US Patent
Sumitomo Pharma
US Patent
Affinity DataIC50: 7nMAssay Description:The HAT activity inhibitory ability of HAT inhibitors was evaluated using SensoLyte HAT(p300) Assay Kit (ANASPEC, AS-72172). Specifically, 7.5 μL of ...More data for this Ligand-Target Pair
TargetHistone acetyltransferase p300 [1287-1666]/Histone acetyltransferase type B catalytic subunit(Human)
Sumitomo Pharma
US Patent
Sumitomo Pharma
US Patent
Affinity DataIC50: 8.76nMAssay Description:The p300 HAT domain (residues 1287-1666) was expressed and purified with an N-terminal His tag from Escherichia coli cells. The expressed protein was...More data for this Ligand-Target Pair
TargetHistone acetyltransferase p300 [1287-1666]/Histone acetyltransferase type B catalytic subunit(Human)
Sumitomo Pharma
US Patent
Sumitomo Pharma
US Patent
Affinity DataIC50: 9nMAssay Description:The HAT activity inhibitory ability of HAT inhibitors was evaluated using SensoLyte HAT(p300) Assay Kit (ANASPEC, AS-72172). Specifically, 7.5 μL of ...More data for this Ligand-Target Pair
TargetHistone acetyltransferase p300 [1287-1666]/Histone acetyltransferase type B catalytic subunit(Human)
Sumitomo Pharma
US Patent
Sumitomo Pharma
US Patent
Affinity DataIC50: 9nMAssay Description:The HAT activity inhibitory ability of HAT inhibitors was evaluated using SensoLyte HAT(p300) Assay Kit (ANASPEC, AS-72172). Specifically, 7.5 μL of ...More data for this Ligand-Target Pair
TargetHistone acetyltransferase p300 [1287-1666]/Histone acetyltransferase type B catalytic subunit(Human)
Sumitomo Pharma
US Patent
Sumitomo Pharma
US Patent
Affinity DataIC50: 9.36nMAssay Description:The p300 HAT domain (residues 1287-1666) was expressed and purified with an N-terminal His tag from Escherichia coli cells. The expressed protein was...More data for this Ligand-Target Pair
TargetHistone acetyltransferase p300 [1287-1666]/Histone acetyltransferase type B catalytic subunit(Human)
Sumitomo Pharma
US Patent
Sumitomo Pharma
US Patent
Affinity DataIC50: 10nMAssay Description:The HAT activity inhibitory ability of HAT inhibitors was evaluated using SensoLyte HAT(p300) Assay Kit (ANASPEC, AS-72172). Specifically, 7.5 μL of ...More data for this Ligand-Target Pair
TargetHistone acetyltransferase p300 [1287-1666]/Histone acetyltransferase type B catalytic subunit(Human)
Sumitomo Pharma
US Patent
Sumitomo Pharma
US Patent
Affinity DataIC50: 11nMAssay Description:The HAT activity inhibitory ability of HAT inhibitors was evaluated using SensoLyte HAT(p300) Assay Kit (ANASPEC, AS-72172). Specifically, 7.5 μL of ...More data for this Ligand-Target Pair
TargetHistone acetyltransferase p300 [1287-1666]/Histone acetyltransferase type B catalytic subunit(Human)
Sumitomo Pharma
US Patent
Sumitomo Pharma
US Patent
Affinity DataIC50: 15.6nMAssay Description:The p300 HAT domain (residues 1287-1666) was expressed and purified with an N-terminal His tag from Escherichia coli cells. The expressed protein was...More data for this Ligand-Target Pair
TargetHistone acetyltransferase p300 [1287-1666]/Histone acetyltransferase type B catalytic subunit(Human)
Sumitomo Pharma
US Patent
Sumitomo Pharma
US Patent
Affinity DataIC50: 16nMAssay Description:The HAT activity inhibitory ability of HAT inhibitors was evaluated using SensoLyte HAT(p300) Assay Kit (ANASPEC, AS-72172). Specifically, 7.5 μL of ...More data for this Ligand-Target Pair
TargetHistone acetyltransferase p300 [1287-1666]/Histone acetyltransferase type B catalytic subunit(Human)
Sumitomo Pharma
US Patent
Sumitomo Pharma
US Patent
Affinity DataIC50: 17nMAssay Description:The p300 HAT domain (residues 1287-1666) was expressed and purified with an N-terminal His tag from Escherichia coli cells. The expressed protein was...More data for this Ligand-Target Pair
TargetHistone acetyltransferase p300 [1287-1666]/Histone acetyltransferase type B catalytic subunit(Human)
Sumitomo Pharma
US Patent
Sumitomo Pharma
US Patent
Affinity DataIC50: 17nMAssay Description:The HAT activity inhibitory ability of HAT inhibitors was evaluated using SensoLyte HAT(p300) Assay Kit (ANASPEC, AS-72172). Specifically, 7.5 μL of ...More data for this Ligand-Target Pair
TargetHistone acetyltransferase p300 [1287-1666]/Histone acetyltransferase type B catalytic subunit(Human)
Sumitomo Pharma
US Patent
Sumitomo Pharma
US Patent
Affinity DataIC50: 17.1nMAssay Description:The p300 HAT domain (residues 1287-1666) was expressed and purified with an N-terminal His tag from Escherichia coli cells. The expressed protein was...More data for this Ligand-Target Pair
TargetHistone acetyltransferase p300 [1287-1666]/Histone acetyltransferase type B catalytic subunit(Human)
Sumitomo Pharma
US Patent
Sumitomo Pharma
US Patent
Affinity DataIC50: 17.7nMAssay Description:The p300 HAT domain (residues 1287-1666) was expressed and purified with an N-terminal His tag from Escherichia coli cells. The expressed protein was...More data for this Ligand-Target Pair
TargetHistone acetyltransferase p300 [1287-1666]/Histone acetyltransferase type B catalytic subunit(Human)
Sumitomo Pharma
US Patent
Sumitomo Pharma
US Patent
Affinity DataIC50: 19nMAssay Description:The HAT activity inhibitory ability of HAT inhibitors was evaluated using SensoLyte HAT(p300) Assay Kit (ANASPEC, AS-72172). Specifically, 7.5 μL of ...More data for this Ligand-Target Pair
TargetHistone acetyltransferase p300 [1287-1666]/Histone acetyltransferase type B catalytic subunit(Human)
Sumitomo Pharma
US Patent
Sumitomo Pharma
US Patent
Affinity DataIC50: 19nMAssay Description:The p300 HAT domain (residues 1287-1666) was expressed and purified with an N-terminal His tag from Escherichia coli cells. The expressed protein was...More data for this Ligand-Target Pair
TargetHistone acetyltransferase p300 [1287-1666]/Histone acetyltransferase type B catalytic subunit(Human)
Sumitomo Pharma
US Patent
Sumitomo Pharma
US Patent
Affinity DataIC50: 20nMAssay Description:The HAT activity inhibitory ability of HAT inhibitors was evaluated using SensoLyte HAT(p300) Assay Kit (ANASPEC, AS-72172). Specifically, 7.5 μL of ...More data for this Ligand-Target Pair
TargetHistone acetyltransferase p300 [1287-1666]/Histone acetyltransferase type B catalytic subunit(Human)
Sumitomo Pharma
US Patent
Sumitomo Pharma
US Patent
Affinity DataIC50: 20.7nMAssay Description:The p300 HAT domain (residues 1287-1666) was expressed and purified with an N-terminal His tag from Escherichia coli cells. The expressed protein was...More data for this Ligand-Target Pair
TargetHistone acetyltransferase p300 [1287-1666]/Histone acetyltransferase type B catalytic subunit(Human)
Sumitomo Pharma
US Patent
Sumitomo Pharma
US Patent
Affinity DataIC50: 21.1nMAssay Description:The p300 HAT domain (residues 1287-1666) was expressed and purified with an N-terminal His tag from Escherichia coli cells. The expressed protein was...More data for this Ligand-Target Pair
TargetHistone acetyltransferase p300 [1287-1666]/Histone acetyltransferase type B catalytic subunit(Human)
Sumitomo Pharma
US Patent
Sumitomo Pharma
US Patent
Affinity DataIC50: 21.8nMAssay Description:The p300 HAT domain (residues 1287-1666) was expressed and purified with an N-terminal His tag from Escherichia coli cells. The expressed protein was...More data for this Ligand-Target Pair
TargetHistone acetyltransferase p300 [1287-1666]/Histone acetyltransferase type B catalytic subunit(Human)
Sumitomo Pharma
US Patent
Sumitomo Pharma
US Patent
Affinity DataIC50: 23.2nMAssay Description:The p300 HAT domain (residues 1287-1666) was expressed and purified with an N-terminal His tag from Escherichia coli cells. The expressed protein was...More data for this Ligand-Target Pair
TargetHistone acetyltransferase p300 [1287-1666]/Histone acetyltransferase type B catalytic subunit(Human)
Sumitomo Pharma
US Patent
Sumitomo Pharma
US Patent
Affinity DataIC50: 26nMAssay Description:The p300 HAT domain (residues 1287-1666) was expressed and purified with an N-terminal His tag from Escherichia coli cells. The expressed protein was...More data for this Ligand-Target Pair
TargetHistone acetyltransferase p300 [1287-1666]/Histone acetyltransferase type B catalytic subunit(Human)
Sumitomo Pharma
US Patent
Sumitomo Pharma
US Patent
Affinity DataIC50: 28nMAssay Description:The p300 HAT domain (residues 1287-1666) was expressed and purified with an N-terminal His tag from Escherichia coli cells. The expressed protein was...More data for this Ligand-Target Pair
TargetHistone acetyltransferase p300 [1287-1666]/Histone acetyltransferase type B catalytic subunit(Human)
Sumitomo Pharma
US Patent
Sumitomo Pharma
US Patent
Affinity DataIC50: 28.8nMAssay Description:The p300 HAT domain (residues 1287-1666) was expressed and purified with an N-terminal His tag from Escherichia coli cells. The expressed protein was...More data for this Ligand-Target Pair
TargetHistone acetyltransferase p300 [1287-1666]/Histone acetyltransferase type B catalytic subunit(Human)
Sumitomo Pharma
US Patent
Sumitomo Pharma
US Patent
Affinity DataIC50: 36.2nMAssay Description:The p300 HAT domain (residues 1287-1666) was expressed and purified with an N-terminal His tag from Escherichia coli cells. The expressed protein was...More data for this Ligand-Target Pair
TargetHistone acetyltransferase p300 [1287-1666]/Histone acetyltransferase type B catalytic subunit(Human)
Sumitomo Pharma
US Patent
Sumitomo Pharma
US Patent
Affinity DataIC50: 39nMAssay Description:The p300 HAT domain (residues 1287-1666) was expressed and purified with an N-terminal His tag from Escherichia coli cells. The expressed protein was...More data for this Ligand-Target Pair
TargetHistone acetyltransferase p300 [1287-1666]/Histone acetyltransferase type B catalytic subunit(Human)
Sumitomo Pharma
US Patent
Sumitomo Pharma
US Patent
Affinity DataIC50: 41nMAssay Description:The p300 HAT domain (residues 1287-1666) was expressed and purified with an N-terminal His tag from Escherichia coli cells. The expressed protein was...More data for this Ligand-Target Pair
TargetHistone acetyltransferase p300 [1287-1666]/Histone acetyltransferase type B catalytic subunit(Human)
Sumitomo Pharma
US Patent
Sumitomo Pharma
US Patent
Affinity DataIC50: 41.9nMAssay Description:The p300 HAT domain (residues 1287-1666) was expressed and purified with an N-terminal His tag from Escherichia coli cells. The expressed protein was...More data for this Ligand-Target Pair
TargetHistone acetyltransferase p300 [1287-1666]/Histone acetyltransferase type B catalytic subunit(Human)
Sumitomo Pharma
US Patent
Sumitomo Pharma
US Patent
Affinity DataIC50: 42.4nMAssay Description:The p300 HAT domain (residues 1287-1666) was expressed and purified with an N-terminal His tag from Escherichia coli cells. The expressed protein was...More data for this Ligand-Target Pair
TargetHistone acetyltransferase p300 [1287-1666]/Histone acetyltransferase type B catalytic subunit(Human)
Sumitomo Pharma
US Patent
Sumitomo Pharma
US Patent
Affinity DataIC50: 44nMAssay Description:The p300 HAT domain (residues 1287-1666) was expressed and purified with an N-terminal His tag from Escherichia coli cells. The expressed protein was...More data for this Ligand-Target Pair