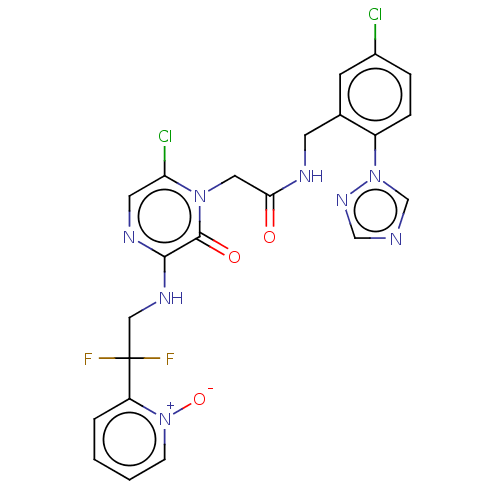
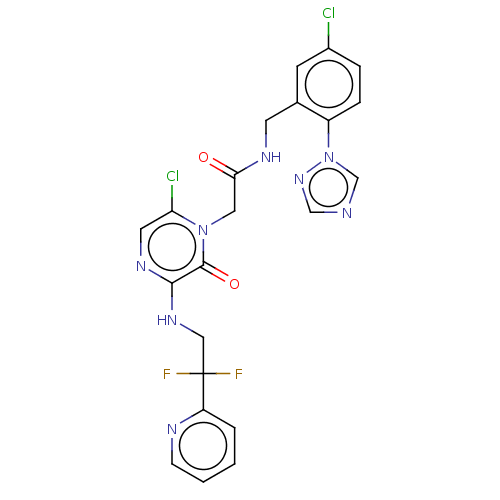
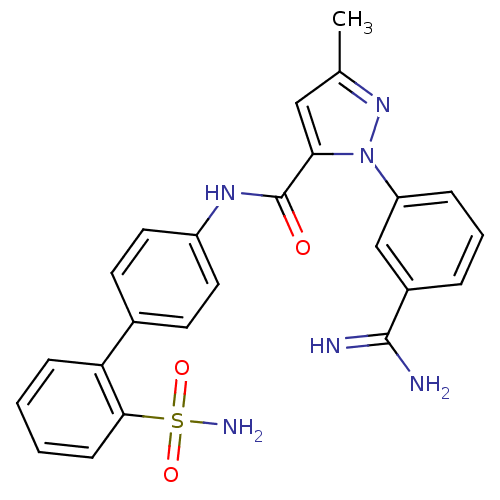
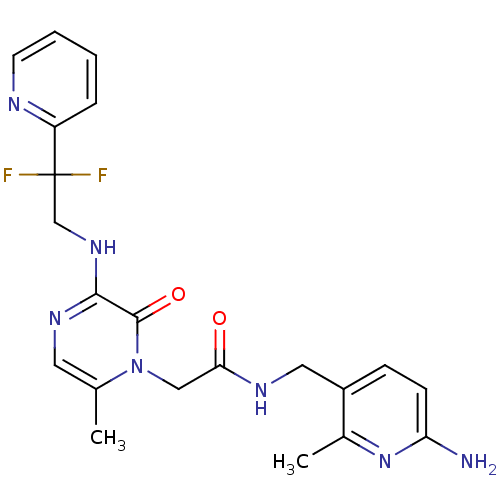
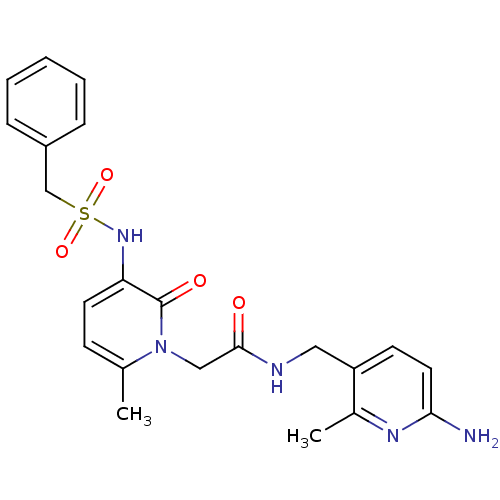
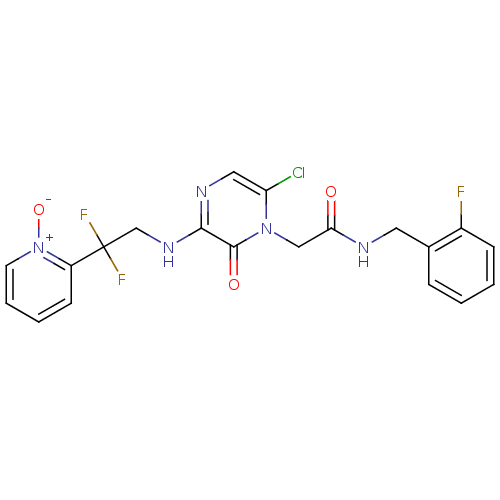
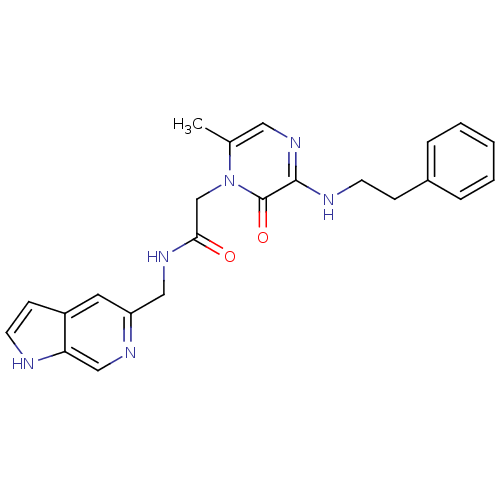
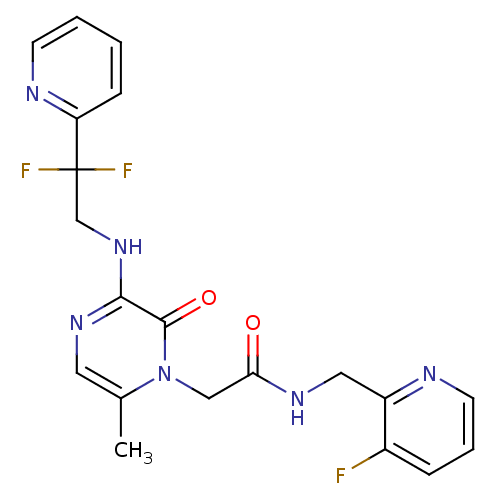
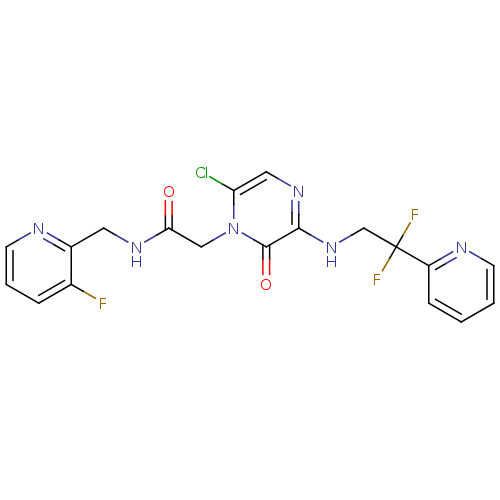
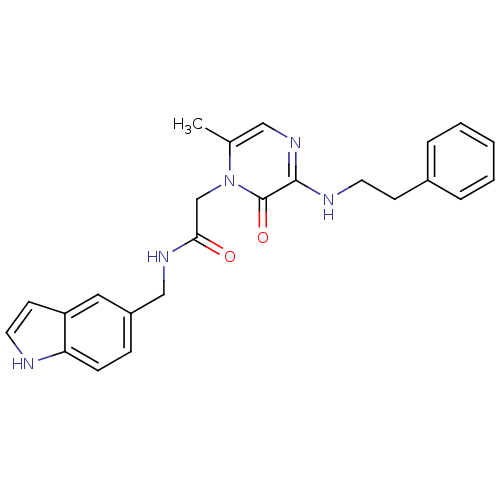
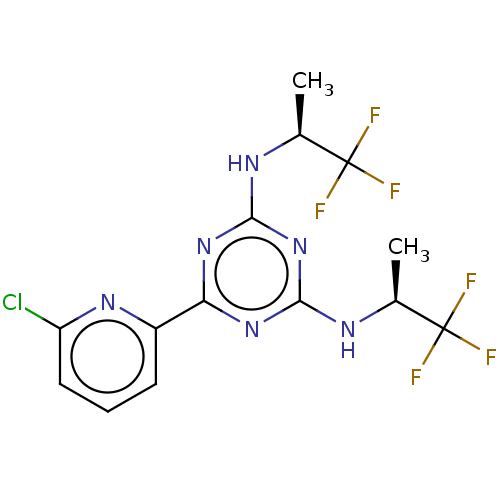
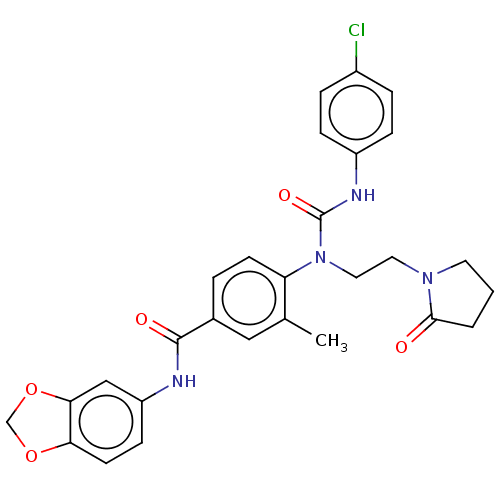
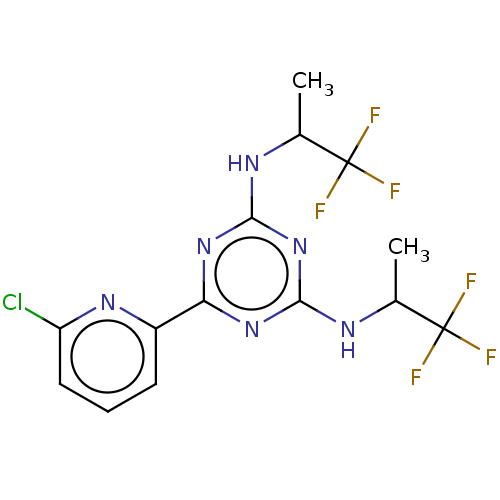
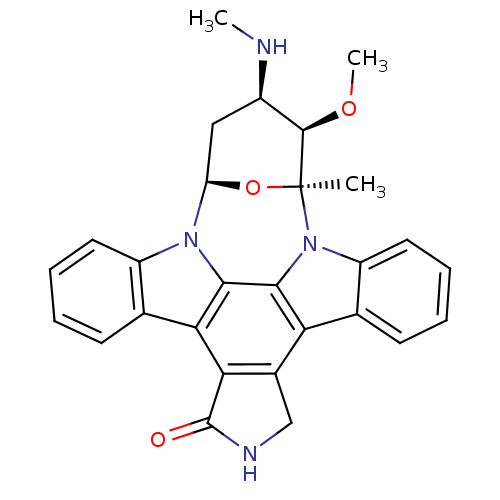
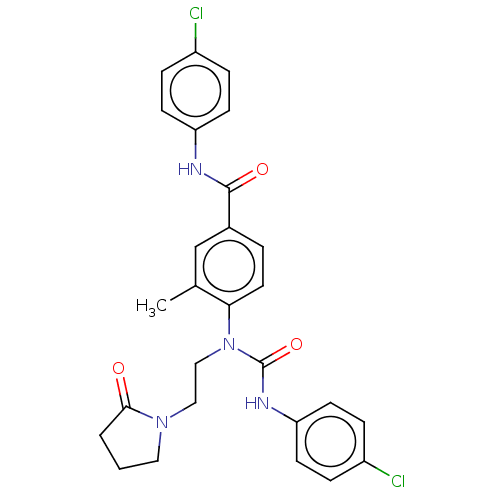
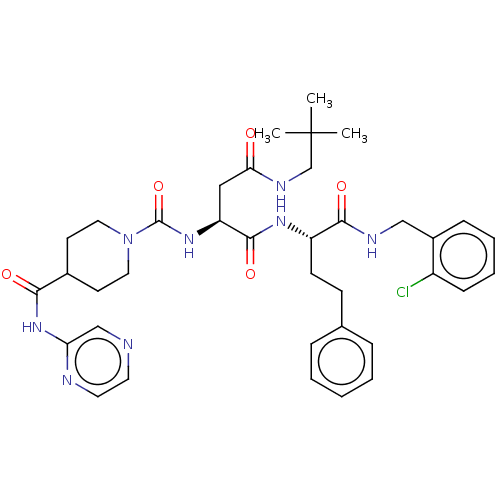
Affinity DataKi: 0.00140nMAssay Description:Inhibition of thrombin (unknown origin)More data for this Ligand-Target Pair
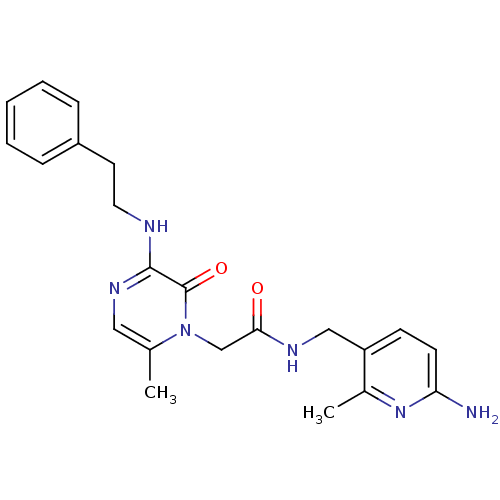
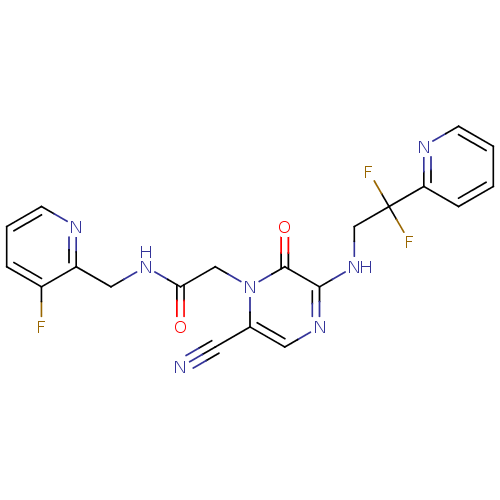
Affinity DataKi: 0.00140nMAssay Description:Binding affinity to thrombin (unknown origin)More data for this Ligand-Target Pair
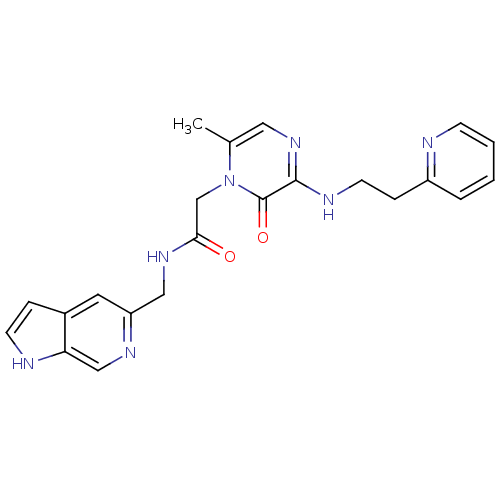
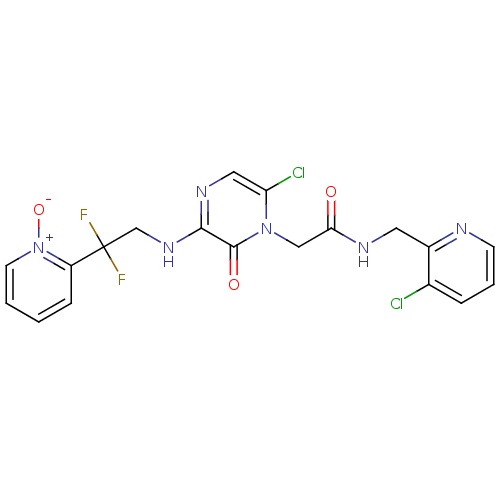
Affinity DataKi: 0.00140nMAssay Description:Binding affinity to thrombin (unknown origin)More data for this Ligand-Target Pair
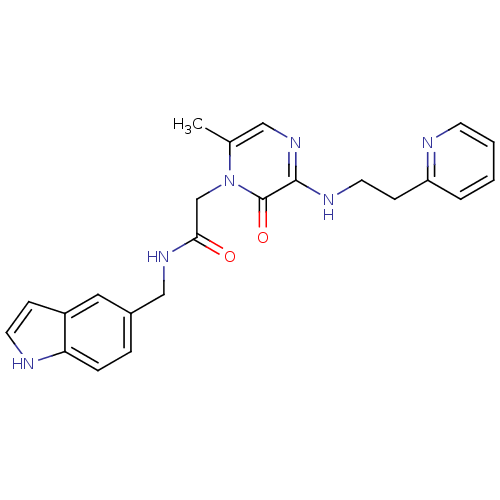
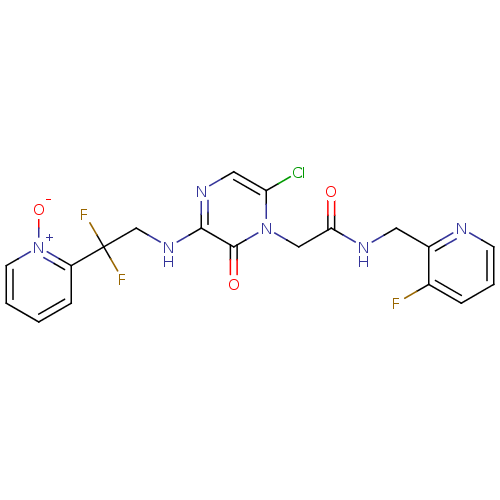
Affinity DataKi: 0.00150nMAssay Description:Inhibition of thrombin (unknown origin)More data for this Ligand-Target Pair
Affinity DataKi: <0.0100nMAssay Description:Inhibition of thrombin (unknown origin)More data for this Ligand-Target Pair
Affinity DataKi: <0.0100nMAssay Description:Binding affinity to thrombin (unknown origin)More data for this Ligand-Target Pair
Affinity DataKi: <0.0100nMAssay Description:Binding affinity to thrombin (unknown origin)More data for this Ligand-Target Pair
Affinity DataKi: <0.0100nMAssay Description:Inhibition of thrombin (unknown origin)More data for this Ligand-Target Pair
Affinity DataKi: 0.0130nMAssay Description:Inhibition of human factor 10a using S-2765 as substrate after 30 mins by spectrophotometric methodMore data for this Ligand-Target Pair
Affinity DataKi: 0.0420nMAssay Description:Inhibition of thrombin (unknown origin)More data for this Ligand-Target Pair
Affinity DataKi: 0.5nMAssay Description:Inhibition of human thrombin assessed as release of p-nitroanilide from chromogenic substrateMore data for this Ligand-Target Pair
Affinity DataKi: 0.800nMAssay Description:Inhibition of human thrombin assessed as release of p-nitroanilide from chromogenic substrateMore data for this Ligand-Target Pair
Affinity DataKi: 1.20nMAssay Description:Inhibition of human thrombin assessed as release of p-nitroanilide from chromogenic substrateMore data for this Ligand-Target Pair
Affinity DataKi: 1.60nMAssay Description:Inhibition of human thrombin assessed as release of p-nitroanilide from chromogenic substrateMore data for this Ligand-Target Pair
Affinity DataKi: 2.30nMAssay Description:Inhibition of thrombin (unknown origin)More data for this Ligand-Target Pair
Affinity DataKi: 2.30nMAssay Description:Inhibition of thrombin (unknown origin)More data for this Ligand-Target Pair
Affinity DataKi: 2.90nMAssay Description:Inhibition of thrombin (unknown origin)More data for this Ligand-Target Pair
Affinity DataKi: 3.10nMAssay Description:Inhibition of thrombin (unknown origin)More data for this Ligand-Target Pair
Affinity DataKi: 3.70nMAssay Description:Inhibition of human thrombin assessed as release of p-nitroanilide from chromogenic substrateMore data for this Ligand-Target Pair
Affinity DataKi: 4.20nMAssay Description:Inhibition of thrombin (unknown origin)More data for this Ligand-Target Pair
Affinity DataKi: 5.20nMAssay Description:Inhibition of thrombin (unknown origin)More data for this Ligand-Target Pair
Affinity DataKi: 16nMAssay Description:Inhibition of human trypsin using S-2222 as substrate after 30 mins by spectrophotometric methodMore data for this Ligand-Target Pair
Affinity DataKi: 18nMAssay Description:Inhibition of human thrombin assessed as release of p-nitroanilide from chromogenic substrateMore data for this Ligand-Target Pair
Affinity DataKi: 300nMAssay Description:Inhibition of human thrombin using S-2366 as substrate after 30 mins by spectrophotometric methodMore data for this Ligand-Target Pair
Affinity DataKi: 800nMAssay Description:Inhibition of trypsin (unknown origin)More data for this Ligand-Target Pair
Affinity DataKi: 1.80E+3nMAssay Description:Inhibition of human trypsin assessed as release of p-nitroanilide from chromogenic substrateMore data for this Ligand-Target Pair
Affinity DataKi: 3.20E+3nMAssay Description:Inhibition of human trypsin assessed as release of p-nitroanilide from chromogenic substrateMore data for this Ligand-Target Pair
Affinity DataKi: 9.50E+4nMAssay Description:Inhibition of human trypsin assessed as release of p-nitroanilide from chromogenic substrateMore data for this Ligand-Target Pair
Affinity DataKi: >1.00E+5nMAssay Description:Inhibition of human trypsin assessed as release of p-nitroanilide from chromogenic substrateMore data for this Ligand-Target Pair
Affinity DataKi: 2.00E+5nMAssay Description:Inhibition of human trypsin assessed as release of p-nitroanilide from chromogenic substrateMore data for this Ligand-Target Pair
Affinity DataKi: 2.02E+5nMAssay Description:Inhibition of trypsin (unknown origin)More data for this Ligand-Target Pair
Affinity DataKi: 2.13E+5nMAssay Description:Inhibition of trypsin (unknown origin)More data for this Ligand-Target Pair
Affinity DataKi: 2.52E+5nMAssay Description:Inhibition of trypsin (unknown origin)More data for this Ligand-Target Pair
Affinity DataKi: 2.90E+5nMAssay Description:Inhibition of human trypsin assessed as release of p-nitroanilide from chromogenic substrateMore data for this Ligand-Target Pair
TargetEctonucleotide pyrophosphatase/phosphodiesterase family member 2(Homo sapiens (Human))
Suzhou Ark Biopharmaceutical
US Patent
Suzhou Ark Biopharmaceutical
US Patent
Affinity DataIC50: 0nMAssay Description:The tested compounds were dissolved with dimethyl sulfoxide (DMSO) to prepare 20 mM of stock solutions, the stock solutions were subjected to 4-fold ...More data for this Ligand-Target Pair
TargetBifunctional epoxide hydrolase 2(Homo sapiens (Human))
Shenyang Pharmaceutical University
Curated by ChEMBL
Shenyang Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 0.0300nMAssay Description:Inhibition of sEH (unknown origin)More data for this Ligand-Target Pair
TargetVascular endothelial growth factor receptor 2(Homo sapiens (Human))
Hangzhou Xixi Hospital
Curated by ChEMBL
Hangzhou Xixi Hospital
Curated by ChEMBL
Affinity DataIC50: 0.0350nMAssay Description:Inhibition of VEGFR-2 (unknown origin)More data for this Ligand-Target Pair
TargetVascular endothelial growth factor receptor 2(Homo sapiens (Human))
Hangzhou Xixi Hospital
Curated by ChEMBL
Hangzhou Xixi Hospital
Curated by ChEMBL
Affinity DataIC50: 0.0860nMAssay Description:Inhibition of VEGFR-2 (unknown origin) by HTRF methodMore data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit beta isoform(Homo sapiens (Human))
Jiaxing University
Curated by ChEMBL
Jiaxing University
Curated by ChEMBL
Affinity DataIC50: 0.190nMAssay Description:Inhibition of PI3Kbeta (unknown origin) using PIP2 as substrate measured after 1 hr by ADP-glo assayMore data for this Ligand-Target Pair
TargetIsocitrate dehydrogenase [NADP] cytoplasmic(Homo sapiens (Human))
Agios Pharmaceuticals
Curated by ChEMBL
Agios Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 0.25nMAssay Description:Inhibition of IDH1 R132H mutant in glioma patient-derived human TS603 neurosphere cells assessed as reduction in 2-HG content using alpha-ketoglutara...More data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform(Homo sapiens (Human))
Jiaxing University
Curated by ChEMBL
Jiaxing University
Curated by ChEMBL
Affinity DataIC50: 0.300nMAssay Description:Inhibition of recombinant human PI3Kalpha using PIP2 as substrate incubated for 1 hr by kinase-glo luminescent assayMore data for this Ligand-Target Pair
TargetBifunctional epoxide hydrolase 2(Homo sapiens (Human))
Shenyang Pharmaceutical University
Curated by ChEMBL
Shenyang Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 0.300nMAssay Description:Inhibition of sEH (unknown origin)More data for this Ligand-Target Pair
TargetBifunctional epoxide hydrolase 2(Homo sapiens (Human))
Shenyang Pharmaceutical University
Curated by ChEMBL
Shenyang Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 0.300nMAssay Description:Inhibition of sEH (unknown origin)More data for this Ligand-Target Pair
TargetIsocitrate dehydrogenase [NADP] cytoplasmic(Homo sapiens (Human))
Agios Pharmaceuticals
Curated by ChEMBL
Agios Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 0.300nMAssay Description:Inhibition of IDH1 R132H mutant in glioma patient-derived human TS603 neurosphere cells assessed as reduction in 2-HG content using alpha-ketoglutara...More data for this Ligand-Target Pair
Affinity DataIC50: 0.340nMAssay Description:Inhibition of recombinant human N-terminal FLAG-tagged mTOR (1362 to end amino acids) using ULight-4E-BP1 as substrate incubated for 1 hr by LANCE Ul...More data for this Ligand-Target Pair
TargetPlatelet-derived growth factor receptor alpha(Homo sapiens (Human))
Hangzhou Xixi Hospital
Curated by ChEMBL
Hangzhou Xixi Hospital
Curated by ChEMBL
Affinity DataIC50: 0.430nMAssay Description:Inhibition of PDGFRalpha (unknown origin) by HTRF methodMore data for this Ligand-Target Pair
TargetPhosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform(Homo sapiens (Human))
Jiaxing University
Curated by ChEMBL
Jiaxing University
Curated by ChEMBL
Affinity DataIC50: 0.440nMAssay Description:Inhibition of recombinant human full-length His-tagged PI3Kgamma expressed in baculovirus expression system using PIP2 as substrate measured after 1 ...More data for this Ligand-Target Pair
TargetBifunctional epoxide hydrolase 2(Homo sapiens (Human))
Shenyang Pharmaceutical University
Curated by ChEMBL
Shenyang Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 0.5nMAssay Description:Inhibition of sEH (unknown origin)More data for this Ligand-Target Pair
TargetReceptor-type tyrosine-protein kinase FLT3(Homo sapiens (Human))
Hangzhou Xixi Hospital
Curated by ChEMBL
Hangzhou Xixi Hospital
Curated by ChEMBL
Affinity DataIC50: 0.520nMAssay Description:Inhibition of FLT3 (unknown origin) by HTRF methodMore data for this Ligand-Target Pair
Affinity DataIC50: 0.600nMAssay Description:Inhibition of chymotrypsin-like activity of human 20S proteasome pretreated for 15 mins followed by Suc-Leu-Leu-Val-Tyr-AMC substrate addition by flu...More data for this Ligand-Target Pair

 3D Structure (crystal)
3D Structure (crystal)