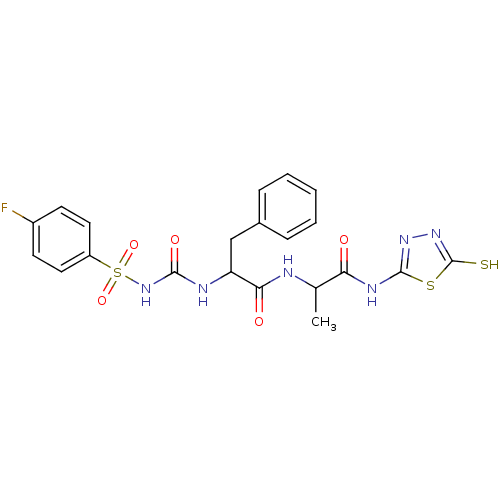
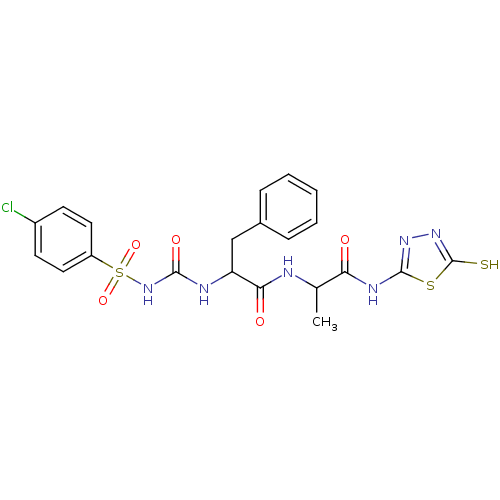
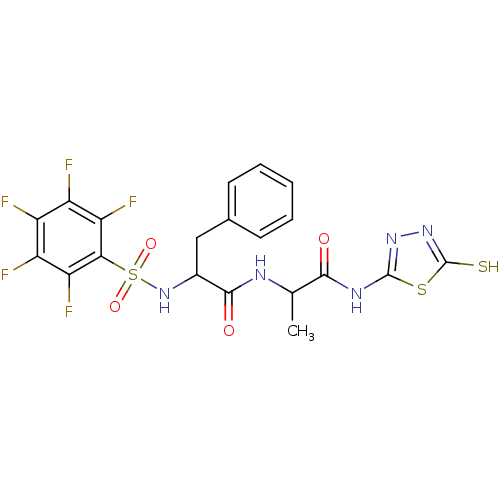
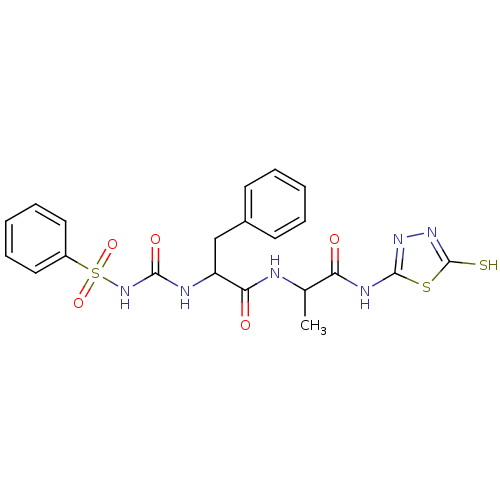
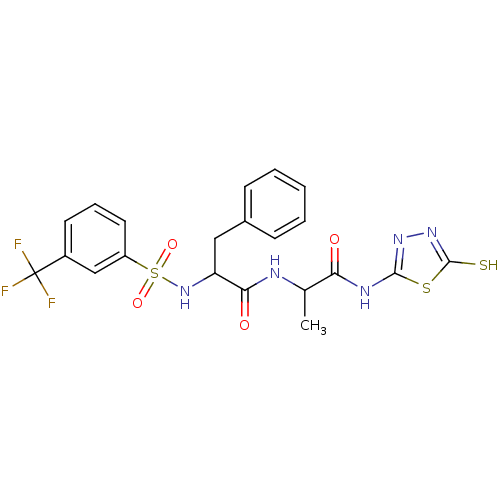
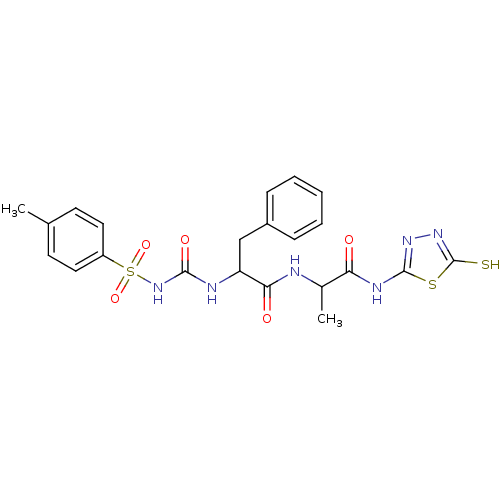
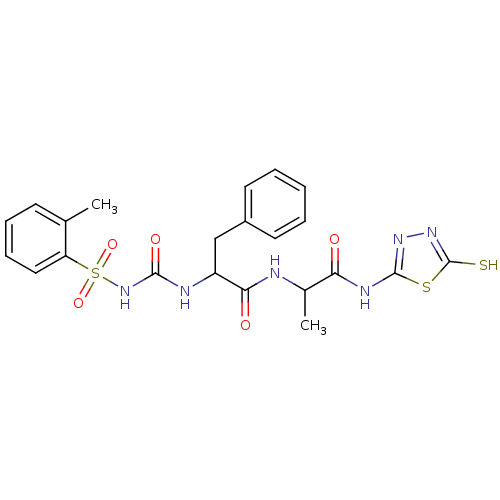
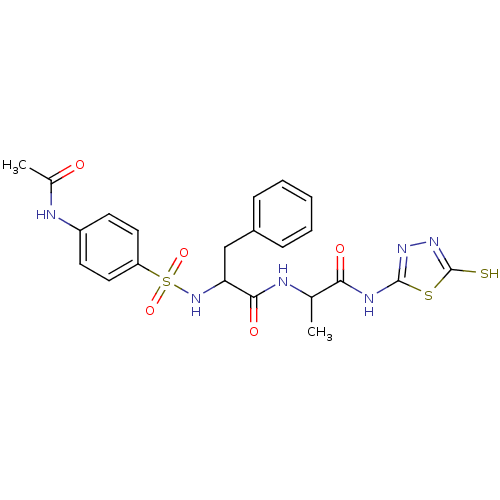
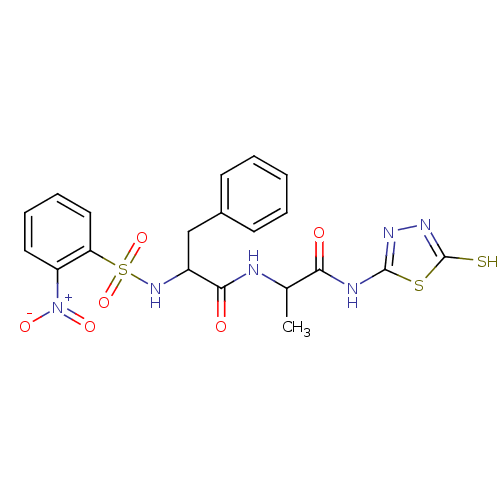
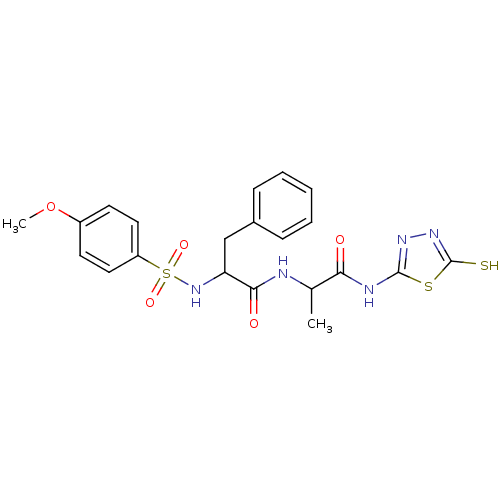
Report error Found 135 Enz. Inhib. hit(s) with all data for entry = 1507
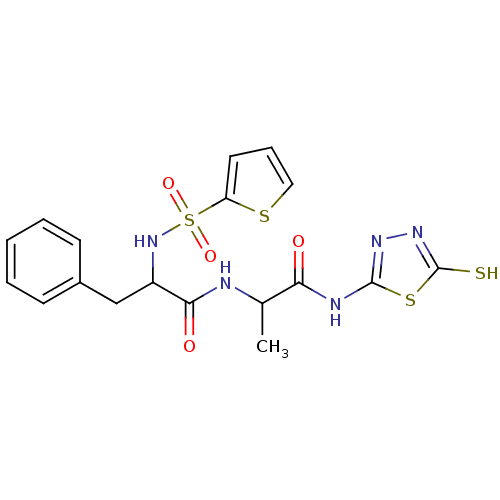
Affinity DataKi: 100nMAssay Description:Initial rates for the hydrolysis of the thioester substrate were used for assessing the catalytic activity and inhibition of the MMPs by the spectrop...More data for this Ligand-Target Pair
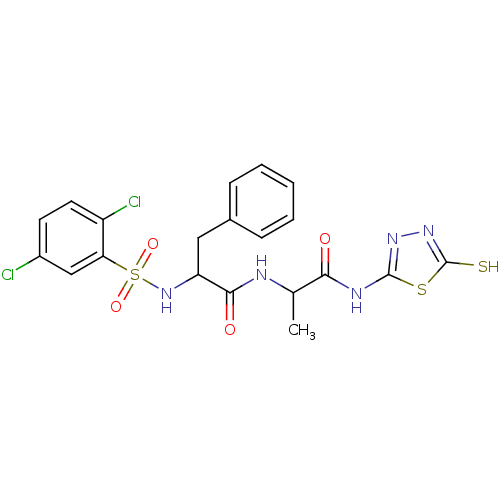
Affinity DataKi: 200nMAssay Description:Initial rates for the hydrolysis of the thioester substrate were used for assessing the catalytic activity and inhibition of the MMPs by the spectrop...More data for this Ligand-Target Pair
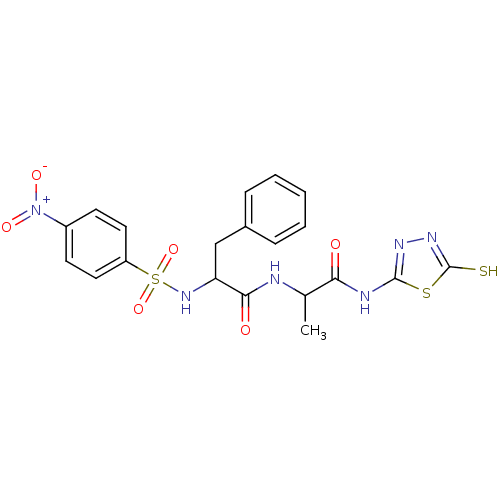
Affinity DataKi: 200nMAssay Description:Initial rates for the hydrolysis of the thioester substrate were used for assessing the catalytic activity and inhibition of the MMPs by the spectrop...More data for this Ligand-Target Pair
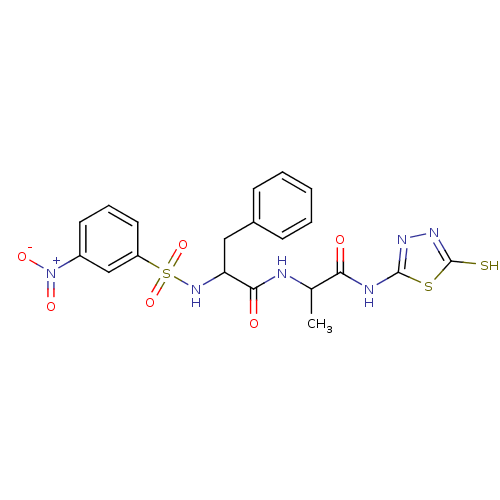
Affinity DataKi: 300nMAssay Description:Initial rates for the hydrolysis of the thioester substrate were used for assessing the catalytic activity and inhibition of the MMPs by the spectrop...More data for this Ligand-Target Pair
Affinity DataKi: 300nMAssay Description:Initial rates for the hydrolysis of the thioester substrate were used for assessing the catalytic activity and inhibition of the MMPs by the spectrop...More data for this Ligand-Target Pair
Affinity DataKi: 400nMAssay Description:Initial rates for the hydrolysis of the thioester substrate were used for assessing the catalytic activity and inhibition of the MMPs by the spectrop...More data for this Ligand-Target Pair
Affinity DataKi: 1.00E+3nMAssay Description:Initial rates for the hydrolysis of the thioester substrate were used for assessing the catalytic activity and inhibition of the MMPs by the spectrop...More data for this Ligand-Target Pair
Affinity DataKi: 2.00E+3nMAssay Description:Initial rates for the hydrolysis of the thioester substrate were used for assessing the catalytic activity and inhibition of the MMPs by the spectrop...More data for this Ligand-Target Pair
Affinity DataKi: 2.00E+3nMAssay Description:Initial rates for the hydrolysis of the thioester substrate were used for assessing the catalytic activity and inhibition of the MMPs by the spectrop...More data for this Ligand-Target Pair
Affinity DataKi: 3.00E+3nMAssay Description:Initial rates for the hydrolysis of the thioester substrate were used for assessing the catalytic activity and inhibition of the MMPs by the spectrop...More data for this Ligand-Target Pair
Affinity DataKi: 3.00E+3nMAssay Description:Initial rates for the hydrolysis of the thioester substrate were used for assessing the catalytic activity and inhibition of the MMPs by the spectrop...More data for this Ligand-Target Pair
Affinity DataKi: 4.00E+3nMAssay Description:Initial rates for the hydrolysis of the thioester substrate were used for assessing the catalytic activity and inhibition of the MMPs by the spectrop...More data for this Ligand-Target Pair
Affinity DataKi: 4.00E+3nMAssay Description:Initial rates for the hydrolysis of the thioester substrate were used for assessing the catalytic activity and inhibition of the MMPs by the spectrop...More data for this Ligand-Target Pair
Affinity DataKi: 4.00E+3nMAssay Description:Initial rates for the hydrolysis of the thioester substrate were used for assessing the catalytic activity and inhibition of the MMPs by the spectrop...More data for this Ligand-Target Pair
Affinity DataKi: 5.00E+3nMAssay Description:Initial rates for the hydrolysis of the thioester substrate were used for assessing the catalytic activity and inhibition of the MMPs by the spectrop...More data for this Ligand-Target Pair
Affinity DataKi: 5.00E+3nMAssay Description:Initial rates for the hydrolysis of the thioester substrate were used for assessing the catalytic activity and inhibition of the MMPs by the spectrop...More data for this Ligand-Target Pair
Affinity DataKi: 5.00E+3nMAssay Description:Initial rates for the hydrolysis of the thioester substrate were used for assessing the catalytic activity and inhibition of the MMPs by the spectrop...More data for this Ligand-Target Pair
Affinity DataKi: 5.00E+3nMAssay Description:Initial rates for the hydrolysis of the thioester substrate were used for assessing the catalytic activity and inhibition of the MMPs by the spectrop...More data for this Ligand-Target Pair
Affinity DataKi: 5.00E+3nMAssay Description:Initial rates for the hydrolysis of the thioester substrate were used for assessing the catalytic activity and inhibition of the MMPs by the spectrop...More data for this Ligand-Target Pair
Affinity DataKi: 5.00E+3nMAssay Description:Initial rates for the hydrolysis of the thioester substrate were used for assessing the catalytic activity and inhibition of the MMPs by the spectrop...More data for this Ligand-Target Pair
Affinity DataKi: 6.00E+3nMAssay Description:Initial rates for the hydrolysis of the thioester substrate were used for assessing the catalytic activity and inhibition of the MMPs by the spectrop...More data for this Ligand-Target Pair
Affinity DataKi: 6.00E+3nMAssay Description:Initial rates for the hydrolysis of the thioester substrate were used for assessing the catalytic activity and inhibition of the MMPs by the spectrop...More data for this Ligand-Target Pair
Affinity DataKi: 7.00E+3nMAssay Description:Initial rates for the hydrolysis of the thioester substrate were used for assessing the catalytic activity and inhibition of the MMPs by the spectrop...More data for this Ligand-Target Pair
Affinity DataKi: 7.00E+3nMAssay Description:Initial rates for the hydrolysis of the thioester substrate were used for assessing the catalytic activity and inhibition of the MMPs by the spectrop...More data for this Ligand-Target Pair
Affinity DataKi: 7.00E+3nMAssay Description:Initial rates for the hydrolysis of the thioester substrate were used for assessing the catalytic activity and inhibition of the MMPs by the spectrop...More data for this Ligand-Target Pair
Affinity DataKi: 8.00E+3nMAssay Description:Initial rates for the hydrolysis of the thioester substrate were used for assessing the catalytic activity and inhibition of the MMPs by the spectrop...More data for this Ligand-Target Pair
Affinity DataKi: 8.00E+3nMAssay Description:Initial rates for the hydrolysis of the thioester substrate were used for assessing the catalytic activity and inhibition of the MMPs by the spectrop...More data for this Ligand-Target Pair
Affinity DataKi: 9.00E+3nMAssay Description:Initial rates for the hydrolysis of the thioester substrate were used for assessing the catalytic activity and inhibition of the MMPs by the spectrop...More data for this Ligand-Target Pair
Affinity DataKi: 9.00E+3nM ΔG°: -28.5kJ/molepH: 6.0 T: 2°CAssay Description:Initial rates for the hydrolysis of the thioester substrate were used for assessing the catalytic activity and inhibition of the MMPs by the spectrop...More data for this Ligand-Target Pair
Affinity DataKi: 9.00E+3nMAssay Description:Initial rates for the hydrolysis of the thioester substrate were used for assessing the catalytic activity and inhibition of the MMPs by the spectrop...More data for this Ligand-Target Pair
Affinity DataKi: 9.00E+3nMAssay Description:Initial rates for the hydrolysis of the thioester substrate were used for assessing the catalytic activity and inhibition of the MMPs by the spectrop...More data for this Ligand-Target Pair
Affinity DataKi: 9.00E+3nMAssay Description:Initial rates for the hydrolysis of the thioester substrate were used for assessing the catalytic activity and inhibition of the MMPs by the spectrop...More data for this Ligand-Target Pair
Affinity DataKi: 1.00E+4nMAssay Description:Initial rates for the hydrolysis of the thioester substrate were used for assessing the catalytic activity and inhibition of the MMPs by the spectrop...More data for this Ligand-Target Pair
Affinity DataKi: 1.00E+4nMAssay Description:Initial rates for the hydrolysis of the thioester substrate were used for assessing the catalytic activity and inhibition of the MMPs by the spectrop...More data for this Ligand-Target Pair
Affinity DataKi: 1.00E+4nMAssay Description:Initial rates for the hydrolysis of the thioester substrate were used for assessing the catalytic activity and inhibition of the MMPs by the spectrop...More data for this Ligand-Target Pair
Affinity DataKi: 1.00E+4nMAssay Description:Initial rates for the hydrolysis of the thioester substrate were used for assessing the catalytic activity and inhibition of the MMPs by the spectrop...More data for this Ligand-Target Pair
Affinity DataKi: 1.00E+4nMAssay Description:Initial rates for the hydrolysis of the thioester substrate were used for assessing the catalytic activity and inhibition of the MMPs by the spectrop...More data for this Ligand-Target Pair
Affinity DataKi: 1.10E+4nMAssay Description:Initial rates for the hydrolysis of the thioester substrate were used for assessing the catalytic activity and inhibition of the MMPs by the spectrop...More data for this Ligand-Target Pair
Affinity DataKi: 1.20E+4nMAssay Description:Initial rates for the hydrolysis of the thioester substrate were used for assessing the catalytic activity and inhibition of the MMPs by the spectrop...More data for this Ligand-Target Pair
Affinity DataKi: 1.20E+4nMAssay Description:Initial rates for the hydrolysis of the thioester substrate were used for assessing the catalytic activity and inhibition of the MMPs by the spectrop...More data for this Ligand-Target Pair
Affinity DataKi: 1.20E+4nMAssay Description:Initial rates for the hydrolysis of the thioester substrate were used for assessing the catalytic activity and inhibition of the MMPs by the spectrop...More data for this Ligand-Target Pair
Affinity DataKi: 1.20E+4nMAssay Description:Initial rates for the hydrolysis of the thioester substrate were used for assessing the catalytic activity and inhibition of the MMPs by the spectrop...More data for this Ligand-Target Pair
Affinity DataKi: 1.20E+4nMAssay Description:Initial rates for the hydrolysis of the thioester substrate were used for assessing the catalytic activity and inhibition of the MMPs by the spectrop...More data for this Ligand-Target Pair
Affinity DataKi: 1.30E+4nM ΔG°: -27.6kJ/molepH: 6.0 T: 2°CAssay Description:Initial rates for the hydrolysis of the thioester substrate were used for assessing the catalytic activity and inhibition of the MMPs by the spectrop...More data for this Ligand-Target Pair
Affinity DataKi: 1.30E+4nMAssay Description:Initial rates for the hydrolysis of the thioester substrate were used for assessing the catalytic activity and inhibition of the MMPs by the spectrop...More data for this Ligand-Target Pair
Affinity DataKi: 1.30E+4nMAssay Description:Initial rates for the hydrolysis of the thioester substrate were used for assessing the catalytic activity and inhibition of the MMPs by the spectrop...More data for this Ligand-Target Pair
Affinity DataKi: 1.30E+4nMAssay Description:Initial rates for the hydrolysis of the thioester substrate were used for assessing the catalytic activity and inhibition of the MMPs by the spectrop...More data for this Ligand-Target Pair
Affinity DataKi: 1.40E+4nMAssay Description:Initial rates for the hydrolysis of the thioester substrate were used for assessing the catalytic activity and inhibition of the MMPs by the spectrop...More data for this Ligand-Target Pair
Affinity DataKi: 1.40E+4nMAssay Description:Initial rates for the hydrolysis of the thioester substrate were used for assessing the catalytic activity and inhibition of the MMPs by the spectrop...More data for this Ligand-Target Pair
Affinity DataKi: 1.40E+4nM ΔG°: -27.4kJ/molepH: 6.0 T: 2°CAssay Description:Initial rates for the hydrolysis of the thioester substrate were used for assessing the catalytic activity and inhibition of the MMPs by the spectrop...More data for this Ligand-Target Pair