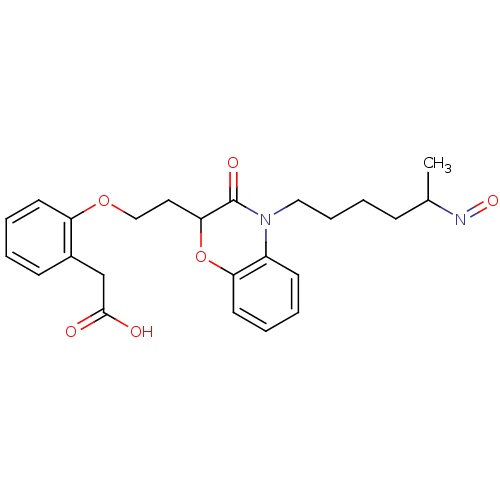
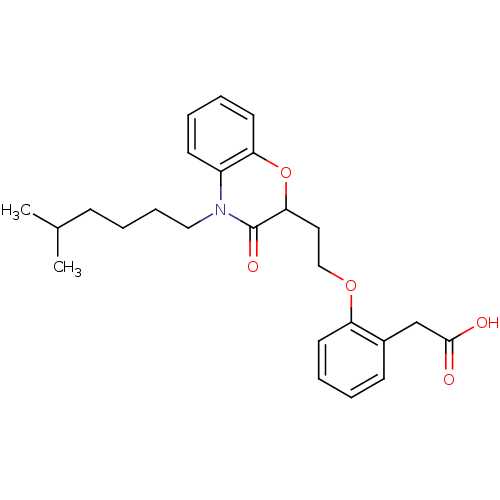
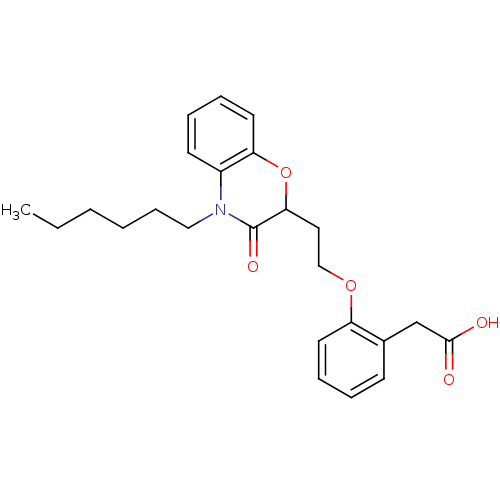
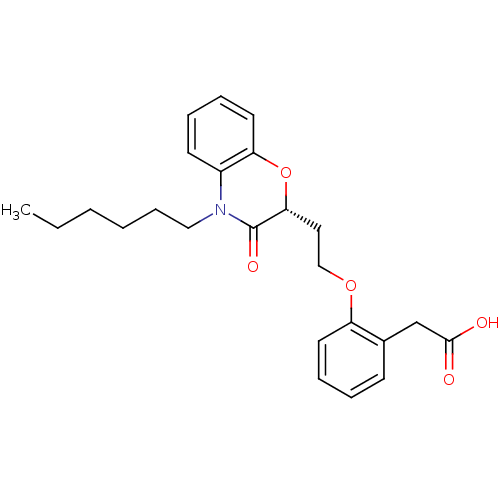
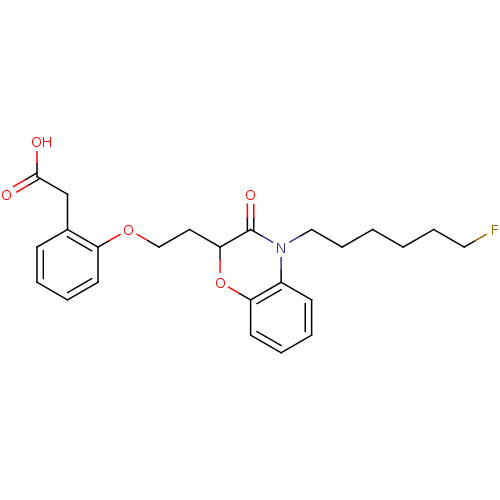
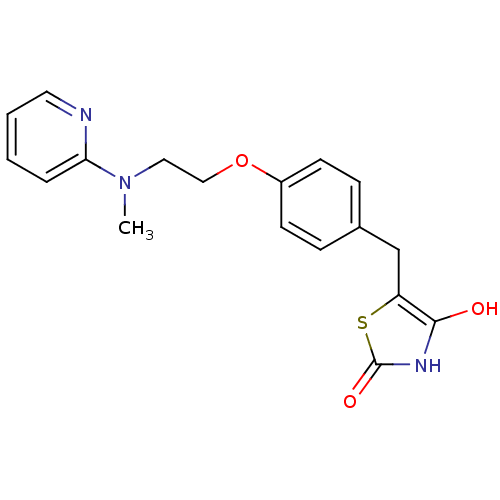
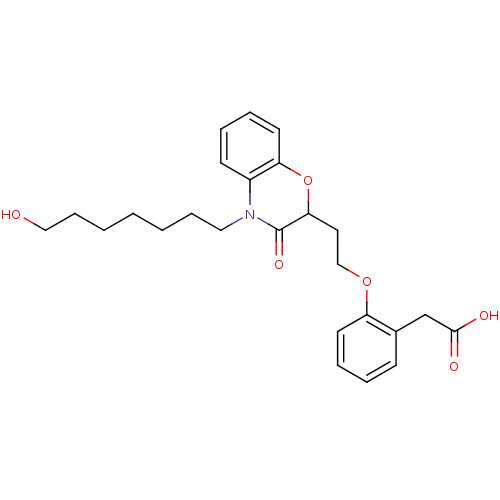
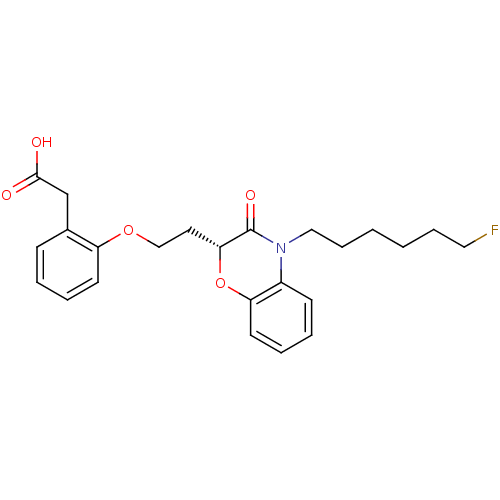
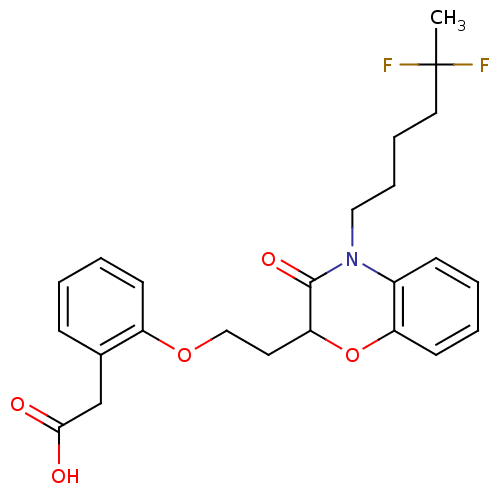
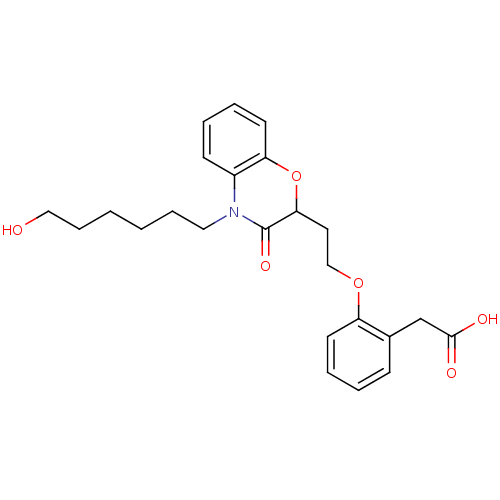
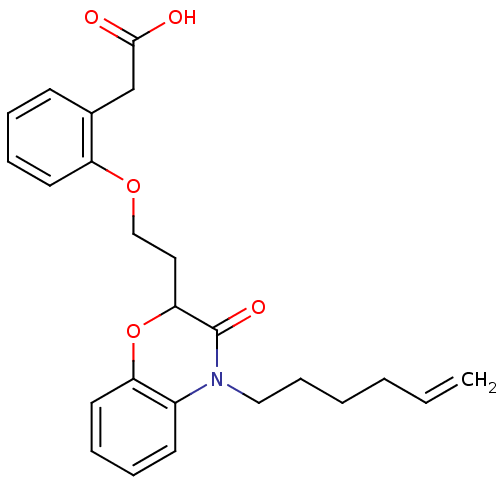
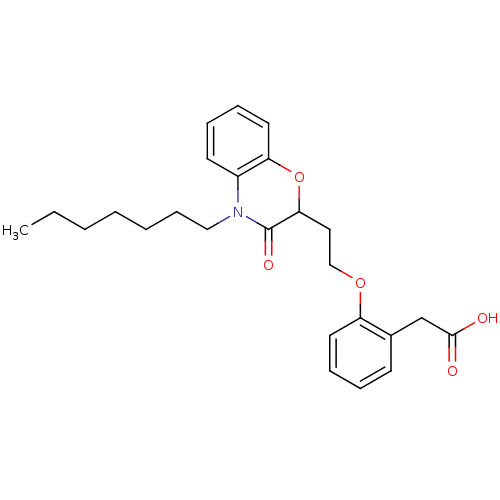
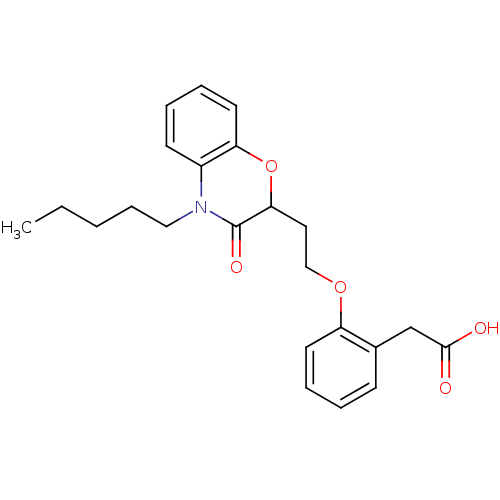
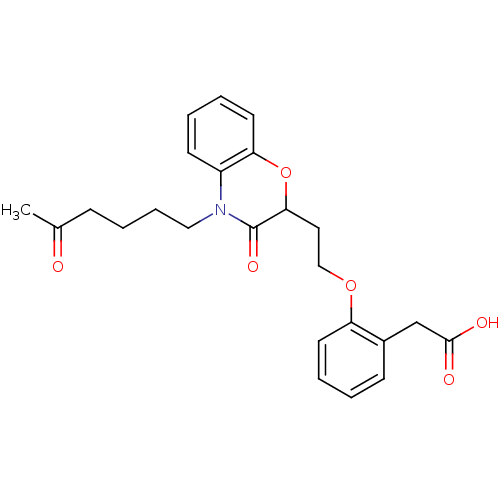
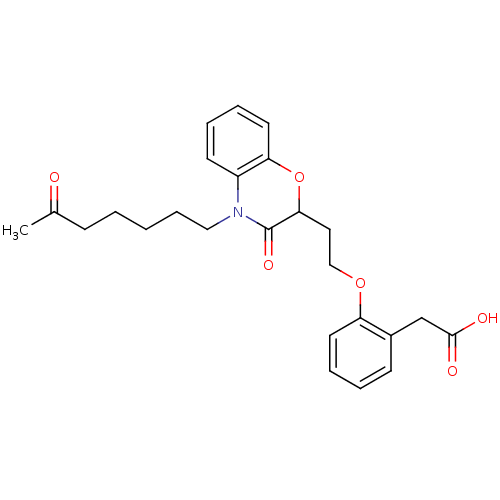
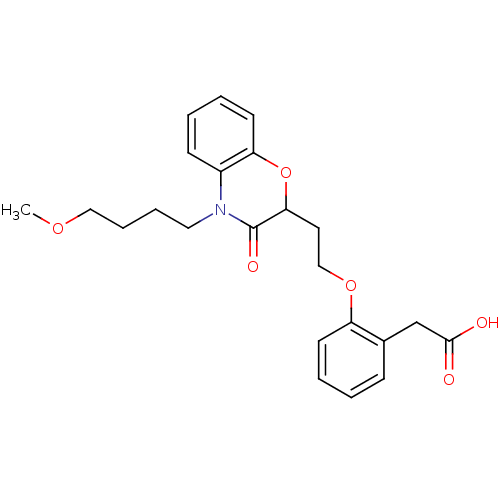
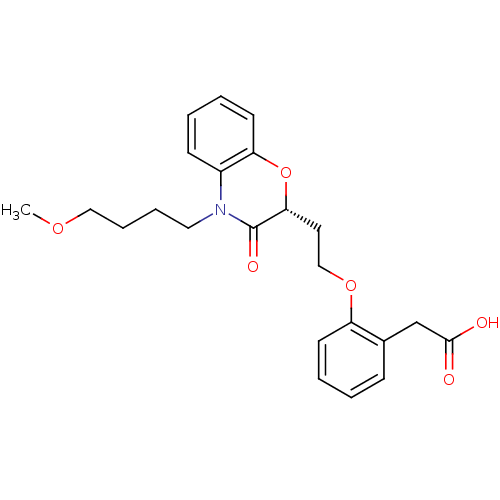
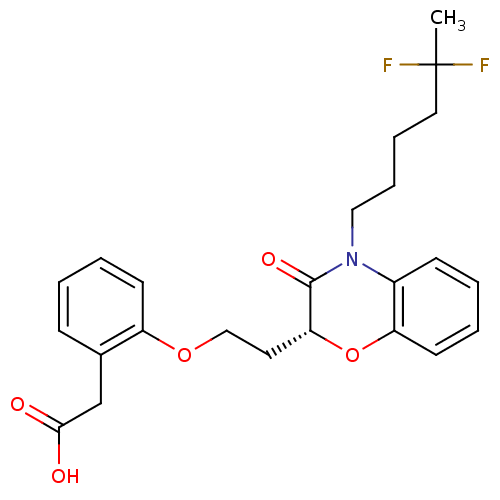
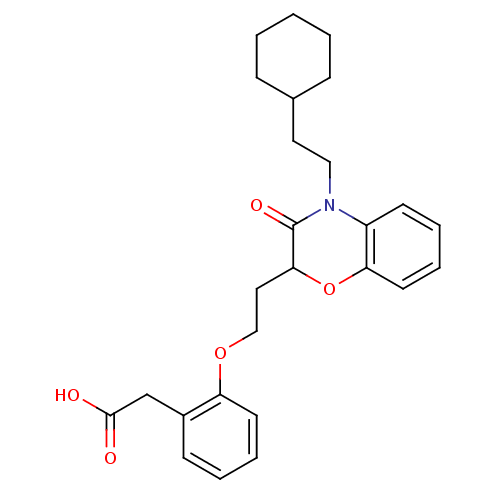
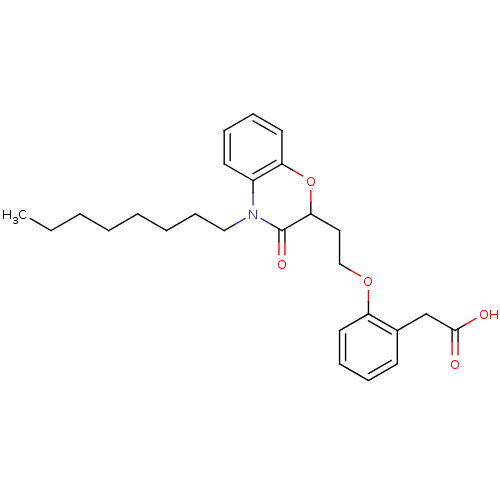
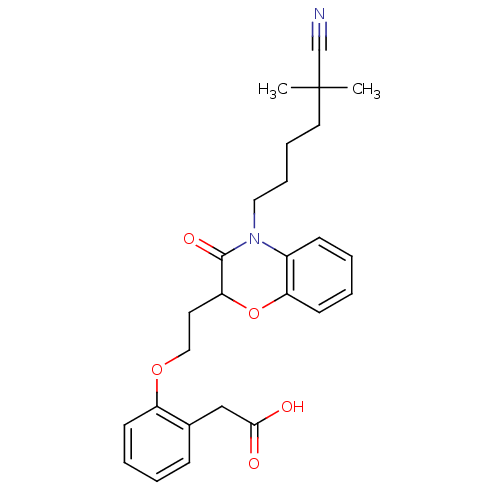
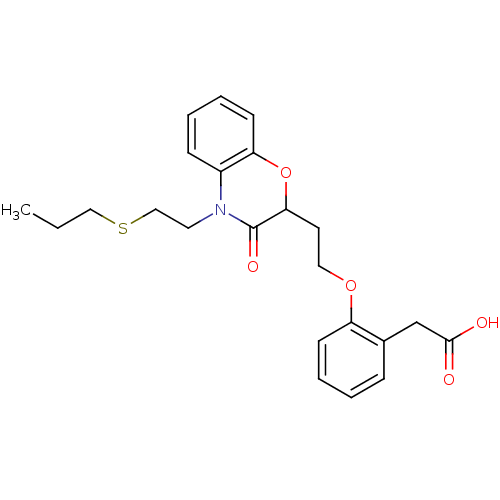
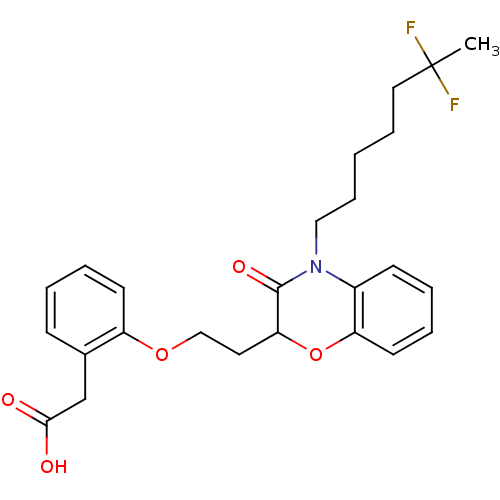
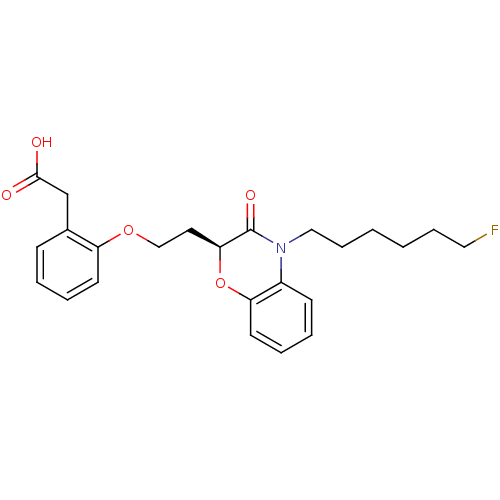
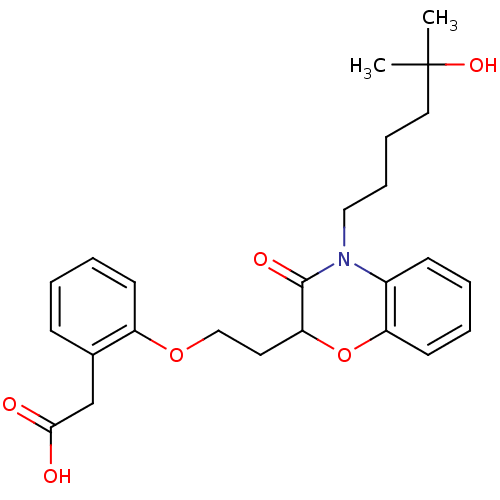
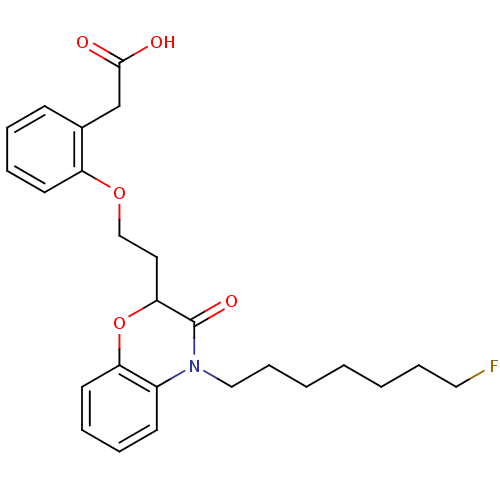
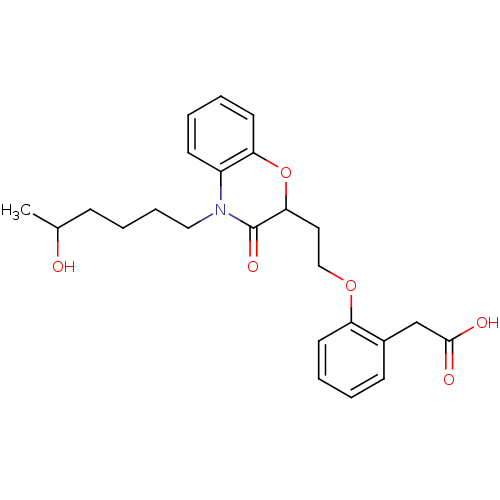
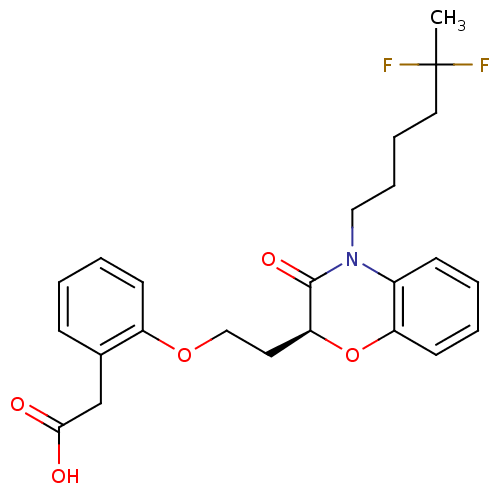
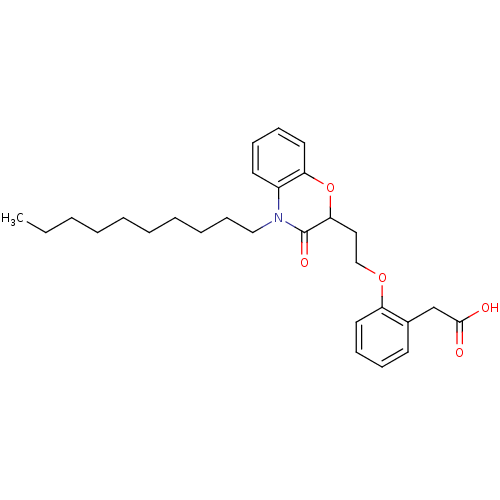
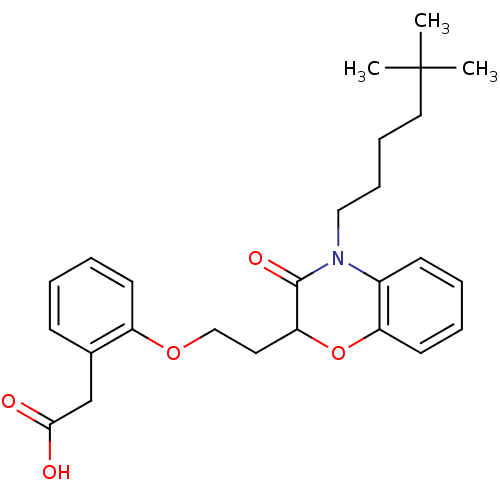
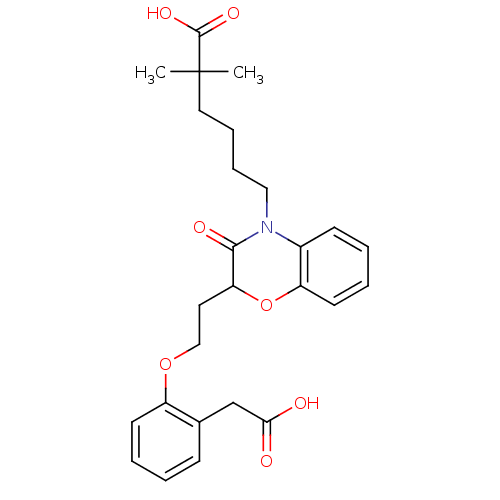
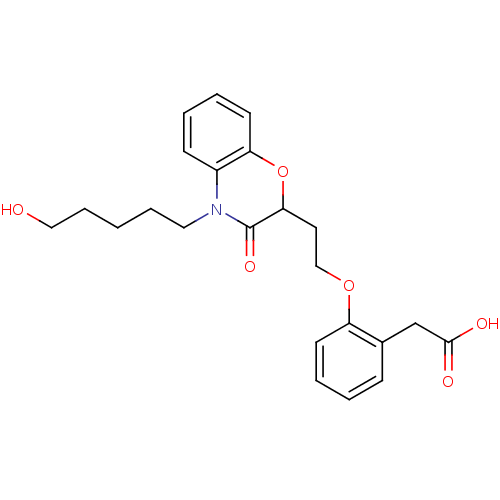
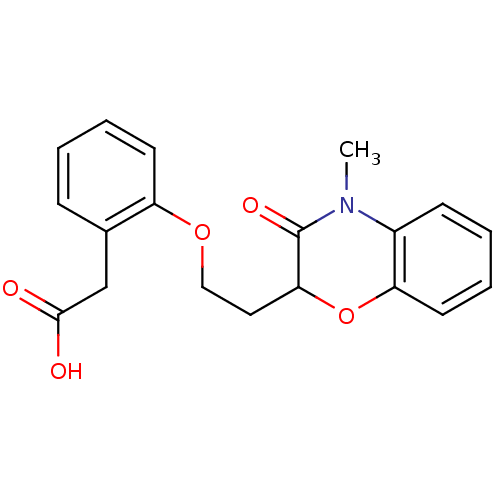
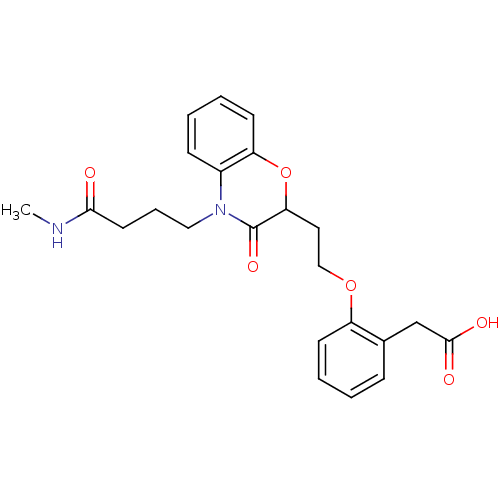
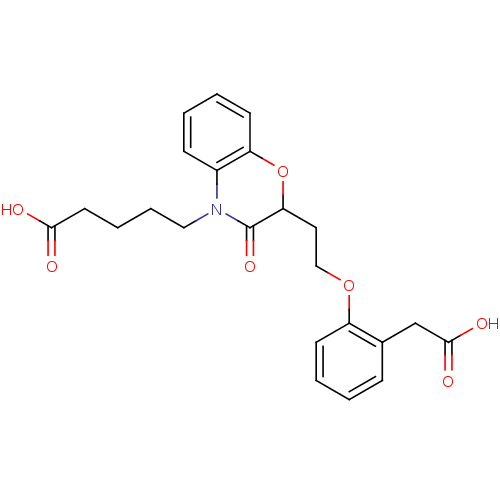
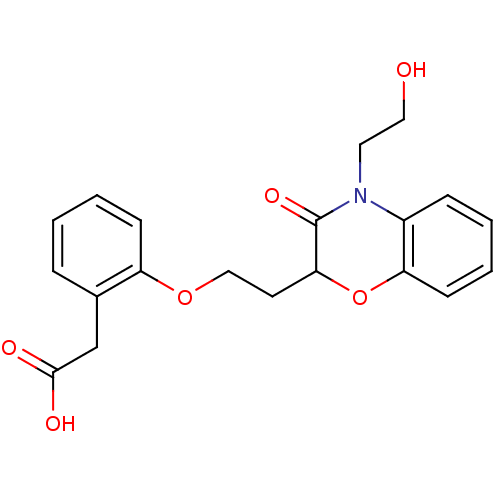
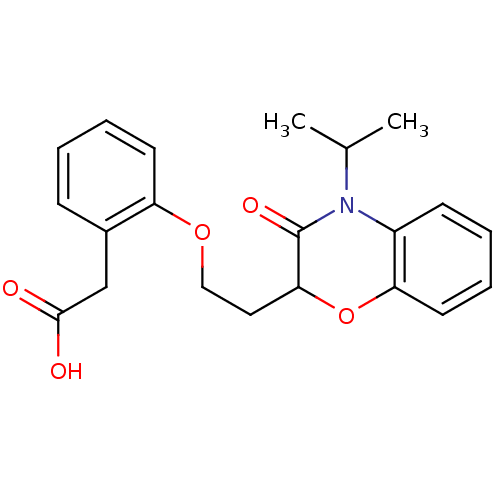
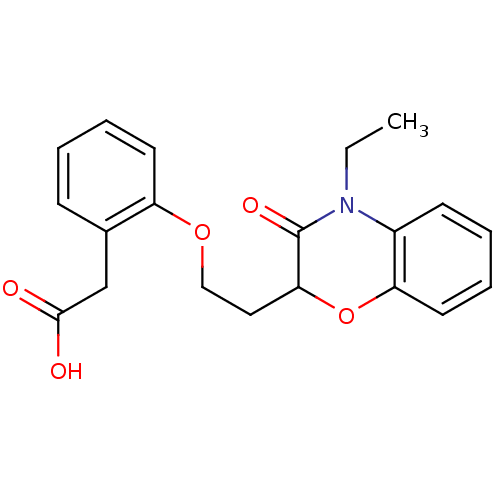
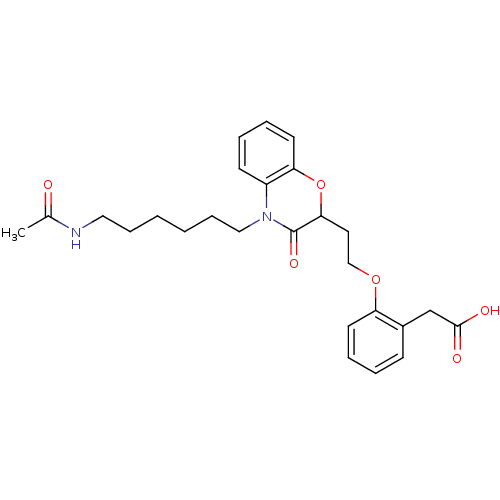
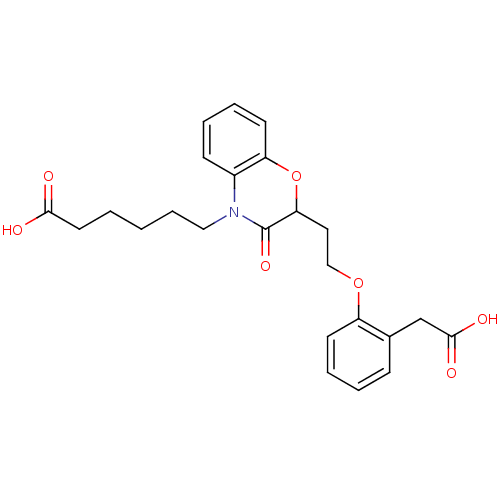
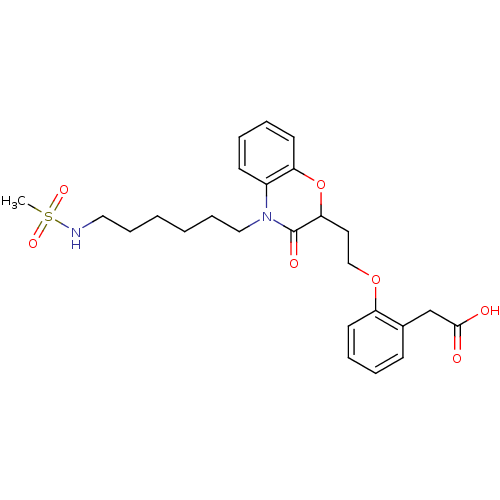
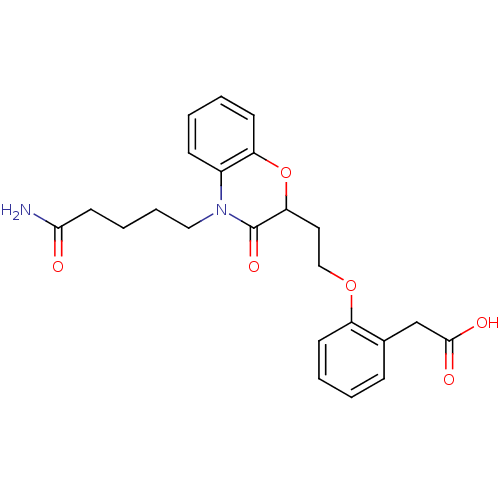
Report error Found 46 Enz. Inhib. hit(s) with all data for entry = 50014082
TargetPeroxisome proliferator-activated receptor gamma(Human)
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
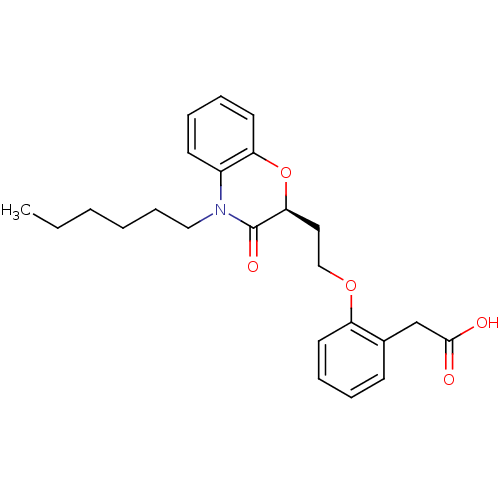
Affinity DataEC50: 10nMAssay Description:Agonistic activity against PPAR (peroxisome proliferator activated receptor gamma) in Suarus chinesisMore data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor gamma(Human)
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
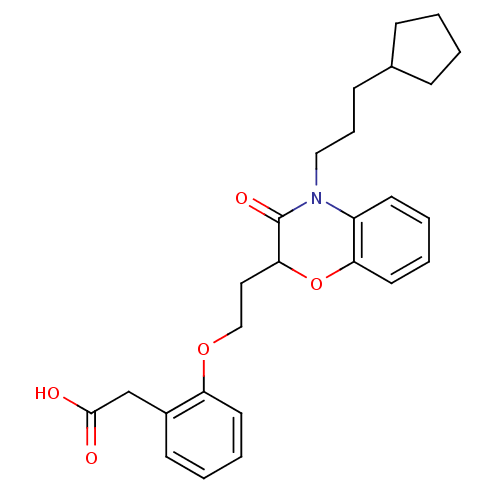
Affinity DataEC50: 79nMAssay Description:Agonistic activity against PPAR (peroxisome proliferator activated receptor gamma) in Suarus chinesisMore data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor gamma(Human)
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
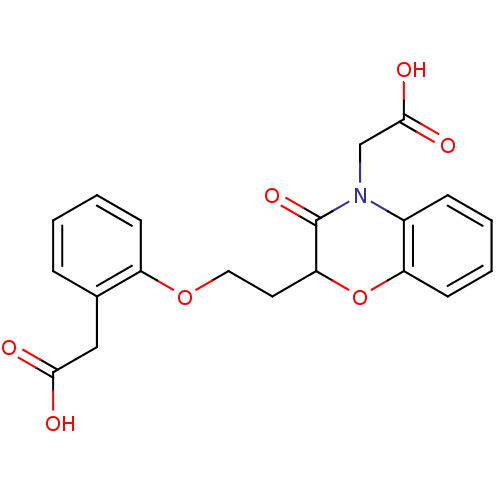
Affinity DataEC50: 100nMAssay Description:Agonistic activity against PPAR (peroxisome proliferator activated receptor gamma) in Suarus chinesisMore data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor gamma(Human)
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
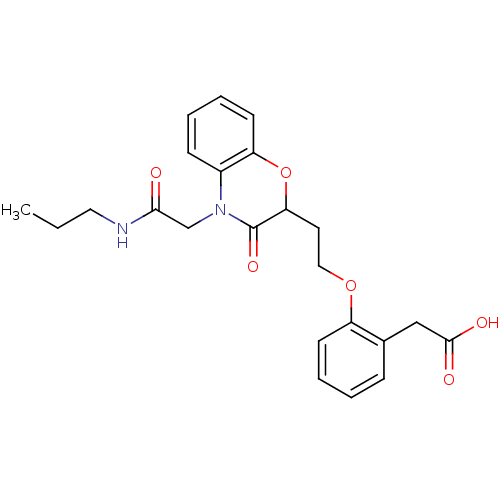
Affinity DataEC50: 100nMAssay Description:Agonistic activity against PPAR (peroxisome proliferator activated receptor gamma) in Suarus chinesisMore data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor gamma(Human)
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataEC50: 117nMAssay Description:Agonistic activity against PPAR (peroxisome proliferator activated receptor gamma) in Suarus chinesisMore data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor gamma(Human)
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataEC50: 120nMAssay Description:Agonistic activity against PPAR (peroxisome proliferator activated receptor gamma) in Suarus chinesisMore data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor gamma(Human)
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataEC50: 149nMAssay Description:Agonistic activity against PPAR (peroxisome proliferator activated receptor gamma) in Suarus chinesisMore data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor gamma(Human)
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataEC50: 152nMAssay Description:Agonistic activity against PPAR (peroxisome proliferator activated receptor gamma) in Suarus chinesisMore data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor gamma(Human)
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataEC50: 179nMAssay Description:Agonistic activity against PPAR (peroxisome proliferator activated receptor gamma) in Suarus chinesisMore data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor gamma(Human)
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataEC50: 200nMAssay Description:Agonistic activity against PPAR (peroxisome proliferator activated receptor gamma) in Suarus chinesisMore data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor gamma(Human)
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataEC50: 208nMAssay Description:Agonistic activity against PPAR (peroxisome proliferator activated receptor gamma) in Suarus chinesisMore data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor gamma(Human)
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataEC50: 234nMAssay Description:Agonistic activity against PPAR (peroxisome proliferator activated receptor gamma) in Suarus chinesisMore data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor gamma(Human)
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataEC50: 243nMAssay Description:Agonistic activity against PPAR (peroxisome proliferator activated receptor gamma) in Suarus chinesisMore data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor gamma(Human)
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataEC50: 260nMAssay Description:Agonistic activity against PPAR (peroxisome proliferator activated receptor gamma) in Suarus chinesisMore data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor gamma(Human)
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataEC50: 264nMAssay Description:Agonistic activity against PPAR (peroxisome proliferator activated receptor gamma) in Suarus chinesisMore data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor gamma(Human)
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataEC50: 274nMAssay Description:Agonistic activity against PPAR (peroxisome proliferator activated receptor gamma) in Suarus chinesisMore data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor gamma(Human)
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataEC50: 274nMAssay Description:Agonistic activity against PPAR (peroxisome proliferator activated receptor gamma) in Suarus chinesisMore data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor gamma(Human)
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataEC50: 295nMAssay Description:Agonistic activity against PPAR (peroxisome proliferator activated receptor gamma) in Suarus chinesisMore data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor gamma(Human)
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataEC50: 300nMAssay Description:Agonistic activity against PPAR (peroxisome proliferator activated receptor gamma) in Suarus chinesisMore data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor gamma(Human)
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataEC50: 300nMAssay Description:Agonistic activity against PPAR (peroxisome proliferator activated receptor gamma) in Suarus chinesisMore data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor gamma(Human)
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataEC50: 359nMAssay Description:Agonistic activity against PPAR (peroxisome proliferator activated receptor gamma) in Suarus chinesisMore data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor gamma(Human)
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataEC50: 380nMAssay Description:Agonistic activity against PPAR (peroxisome proliferator activated receptor gamma) in Suarus chinesisMore data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor gamma(Human)
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataEC50: 534nMAssay Description:Agonistic activity against PPAR (peroxisome proliferator activated receptor gamma) in Suarus chinesisMore data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor gamma(Human)
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataEC50: 570nMAssay Description:Agonistic activity against PPAR (peroxisome proliferator activated receptor gamma) in Suarus chinesisMore data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor gamma(Human)
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataEC50: 644nMAssay Description:Agonistic activity against PPAR (peroxisome proliferator activated receptor gamma) in Suarus chinesisMore data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor gamma(Human)
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataEC50: 718nMAssay Description:Agonistic activity against PPAR (peroxisome proliferator activated receptor gamma) in Suarus chinesisMore data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor gamma(Human)
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataEC50: 1.00E+3nMAssay Description:Agonistic activity against PPAR (peroxisome proliferator activated receptor gamma) in Suarus chinesisMore data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor gamma(Human)
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataEC50: 1.00E+3nMAssay Description:Agonistic activity against PPAR (peroxisome proliferator activated receptor gamma) in Suarus chinesisMore data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor gamma(Human)
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataEC50: 1.00E+3nMAssay Description:Agonistic activity against PPAR (peroxisome proliferator activated receptor gamma) in Suarus chinesisMore data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor gamma(Human)
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataEC50: 1.00E+3nMAssay Description:Agonistic activity against PPAR (peroxisome proliferator activated receptor gamma) in Suarus chinesisMore data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor gamma(Human)
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataEC50: 1.00E+3nMAssay Description:Agonistic activity against PPAR (peroxisome proliferator activated receptor gamma) in Suarus chinesisMore data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor gamma(Human)
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataEC50: 1.00E+3nMAssay Description:Agonistic activity against PPAR (peroxisome proliferator activated receptor gamma) in Suarus chinesisMore data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor gamma(Human)
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataEC50: 1.20E+3nMAssay Description:Agonistic activity against PPAR (peroxisome proliferator activated receptor gamma) in Suarus chinesisMore data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor gamma(Human)
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataEC50: 2.70E+3nMAssay Description:Agonistic activity against PPAR (peroxisome proliferator activated receptor gamma) in Suarus chinesisMore data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor gamma(Human)
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataEC50: >5.00E+3nMAssay Description:Agonistic activity against PPAR (peroxisome proliferator activated receptor gamma) in Suarus chinesisMore data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor gamma(Human)
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataEC50: >5.00E+3nMAssay Description:Agonistic activity against PPAR (peroxisome proliferator activated receptor gamma) in Suarus chinesisMore data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor gamma(Human)
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataEC50: >5.00E+3nMAssay Description:Agonistic activity against PPAR (peroxisome proliferator activated receptor gamma) in Suarus chinesisMore data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor gamma(Human)
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataEC50: >5.00E+3nMAssay Description:Agonistic activity against PPAR (peroxisome proliferator activated receptor gamma) in Suarus chinesisMore data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor gamma(Human)
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataEC50: >5.00E+3nMAssay Description:Agonistic activity against PPAR (peroxisome proliferator activated receptor gamma) in Suarus chinesisMore data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor gamma(Human)
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataEC50: >5.00E+3nMAssay Description:Agonistic activity against PPAR (peroxisome proliferator activated receptor gamma) in Suarus chinesisMore data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor gamma(Human)
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataEC50: >5.00E+3nMAssay Description:Agonistic activity against PPAR (peroxisome proliferator activated receptor gamma) in Suarus chinesisMore data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor gamma(Human)
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataEC50: >5.00E+3nMAssay Description:Agonistic activity against PPAR (peroxisome proliferator activated receptor gamma) in Suarus chinesisMore data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor gamma(Human)
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataEC50: >5.00E+3nMAssay Description:Agonistic activity against PPAR (peroxisome proliferator activated receptor gamma) in Suarus chinesisMore data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor gamma(Human)
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataEC50: >5.00E+3nMAssay Description:Agonistic activity against PPAR (peroxisome proliferator activated receptor gamma) in Suarus chinesisMore data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor gamma(Human)
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataEC50: >5.00E+3nMAssay Description:Agonistic activity against PPAR (peroxisome proliferator activated receptor gamma) in Suarus chinesisMore data for this Ligand-Target Pair
TargetPeroxisome proliferator-activated receptor gamma(Human)
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Johnson and Johnson Pharmaceutical Research and Development
Curated by ChEMBL
Affinity DataEC50: >5.00E+3nMAssay Description:Agonistic activity against PPAR (peroxisome proliferator activated receptor gamma) in Suarus chinesisMore data for this Ligand-Target Pair