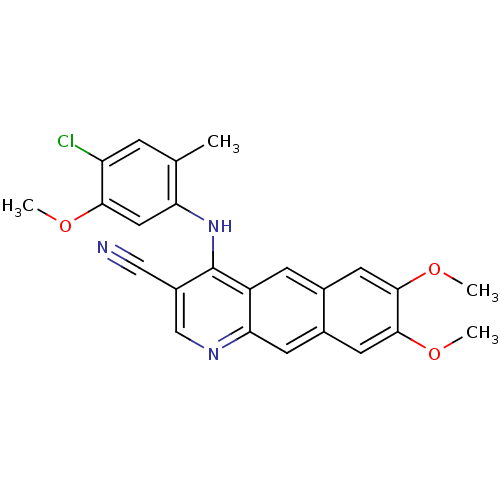
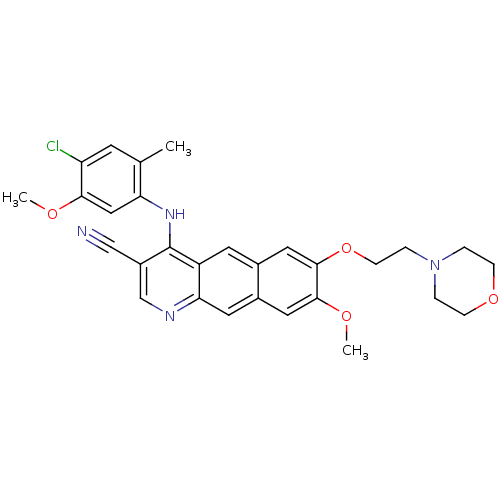
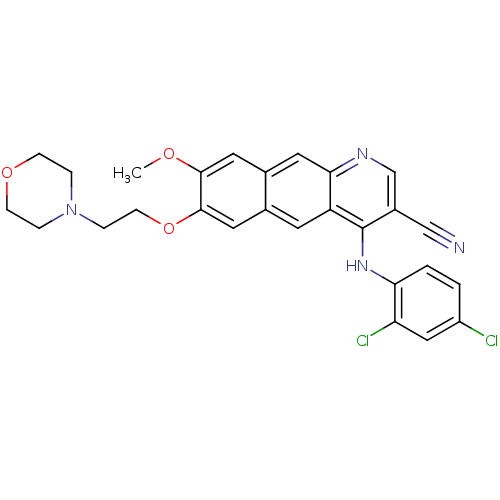
Report error Found 19 Enz. Inhib. hit(s) with all data for entry = 834
Affinity DataIC50: 0.150nMpH: 7.5 T: 2°CAssay Description:Src kinase activity was measured in an ELISA format. IC50 is the inhibitor concentration which inhibits 50% of kinase activity that catalyzes the tra...More data for this Ligand-Target Pair
Affinity DataIC50: 0.290nMpH: 7.5 T: 2°CAssay Description:Src kinase activity was measured in an ELISA format. IC50 is the inhibitor concentration which inhibits 50% of kinase activity that catalyzes the tra...More data for this Ligand-Target Pair
Affinity DataIC50: 0.310nMpH: 7.5 T: 2°CAssay Description:Src kinase activity was measured in an ELISA format. IC50 is the inhibitor concentration which inhibits 50% of kinase activity that catalyzes the tra...More data for this Ligand-Target Pair
Affinity DataIC50: 0.330nMpH: 7.5 T: 2°CAssay Description:Src kinase activity was measured in an ELISA format. IC50 is the inhibitor concentration which inhibits 50% of kinase activity that catalyzes the tra...More data for this Ligand-Target Pair
Affinity DataIC50: 0.460nMpH: 7.5 T: 2°CAssay Description:Src kinase activity was measured in an ELISA format. IC50 is the inhibitor concentration which inhibits 50% of kinase activity that catalyzes the tra...More data for this Ligand-Target Pair
Affinity DataIC50: 0.480nMpH: 7.5 T: 2°CAssay Description:Src kinase activity was measured in an ELISA format. IC50 is the inhibitor concentration which inhibits 50% of kinase activity that catalyzes the tra...More data for this Ligand-Target Pair
Affinity DataIC50: 0.550nMpH: 7.5 T: 2°CAssay Description:Src kinase activity was measured in an ELISA format. IC50 is the inhibitor concentration which inhibits 50% of kinase activity that catalyzes the tra...More data for this Ligand-Target Pair
Affinity DataIC50: 0.720nMpH: 7.5 T: 2°CAssay Description:Src kinase activity was measured in an ELISA format. IC50 is the inhibitor concentration which inhibits 50% of kinase activity that catalyzes the tra...More data for this Ligand-Target Pair
Affinity DataIC50: 1.10nMpH: 7.5 T: 2°CAssay Description:Src kinase activity was measured in an ELISA format. IC50 is the inhibitor concentration which inhibits 50% of kinase activity that catalyzes the tra...More data for this Ligand-Target Pair
Affinity DataIC50: 1.20nMpH: 7.5 T: 2°CAssay Description:Src kinase activity was measured in an ELISA format. IC50 is the inhibitor concentration which inhibits 50% of kinase activity that catalyzes the tra...More data for this Ligand-Target Pair
Affinity DataIC50: 1.20nMpH: 7.5 T: 2°CAssay Description:Src kinase activity was measured in an ELISA format. IC50 is the inhibitor concentration which inhibits 50% of kinase activity that catalyzes the tra...More data for this Ligand-Target Pair
Affinity DataIC50: 1.40nMpH: 7.5 T: 2°CAssay Description:Src kinase activity was measured in an ELISA format. IC50 is the inhibitor concentration which inhibits 50% of kinase activity that catalyzes the tra...More data for this Ligand-Target Pair
Affinity DataIC50: 1.5nMpH: 7.5 T: 2°CAssay Description:Src kinase activity was measured in an ELISA format. IC50 is the inhibitor concentration which inhibits 50% of kinase activity that catalyzes the tra...More data for this Ligand-Target Pair
Affinity DataIC50: 1.80nMpH: 7.5 T: 2°CAssay Description:Src kinase activity was measured in an ELISA format. IC50 is the inhibitor concentration which inhibits 50% of kinase activity that catalyzes the tra...More data for this Ligand-Target Pair
Affinity DataIC50: 2.10nMpH: 7.5 T: 2°CAssay Description:Src kinase activity was measured in an ELISA format. IC50 is the inhibitor concentration which inhibits 50% of kinase activity that catalyzes the tra...More data for this Ligand-Target Pair
Affinity DataIC50: 2.10nMpH: 7.5 T: 2°CAssay Description:Src kinase activity was measured in an ELISA format. IC50 is the inhibitor concentration which inhibits 50% of kinase activity that catalyzes the tra...More data for this Ligand-Target Pair
Affinity DataIC50: 2.5nMpH: 7.5 T: 2°CAssay Description:Src kinase activity was measured in an ELISA format. IC50 is the inhibitor concentration which inhibits 50% of kinase activity that catalyzes the tra...More data for this Ligand-Target Pair
Affinity DataIC50: 2.60nMpH: 7.5 T: 2°CAssay Description:Src kinase activity was measured in an ELISA format. IC50 is the inhibitor concentration which inhibits 50% of kinase activity that catalyzes the tra...More data for this Ligand-Target Pair
Affinity DataIC50: 13nMpH: 7.5 T: 2°CAssay Description:Src kinase activity was measured in an ELISA format. IC50 is the inhibitor concentration which inhibits 50% of kinase activity that catalyzes the tra...More data for this Ligand-Target Pair


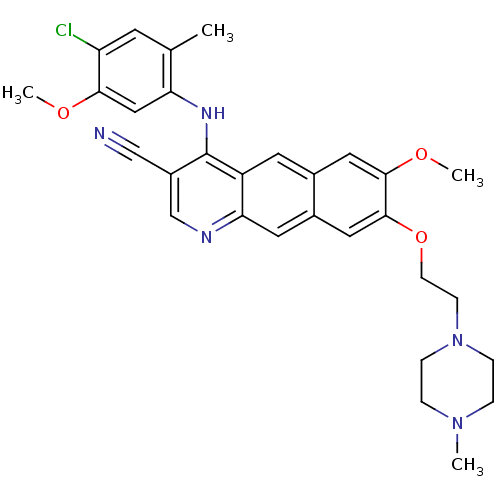

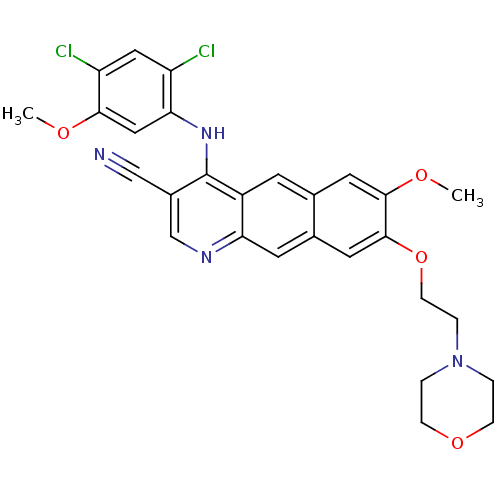

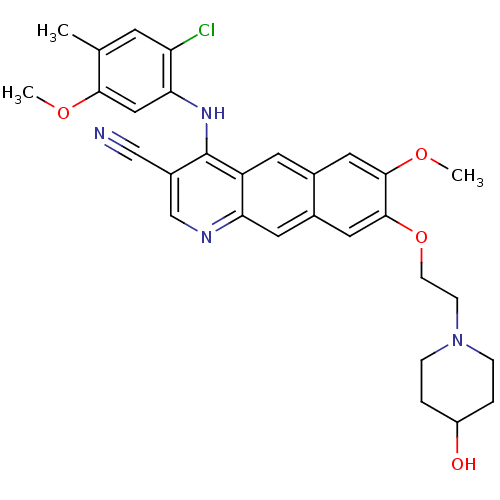
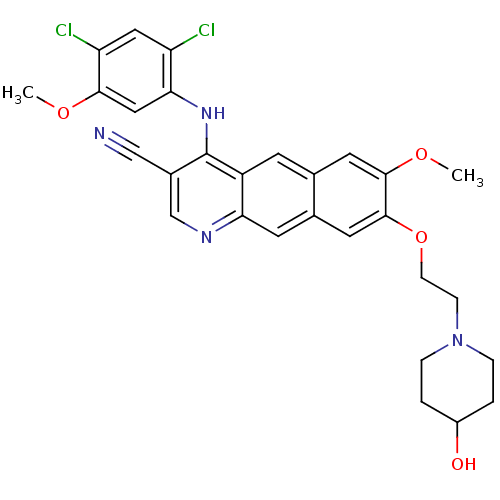

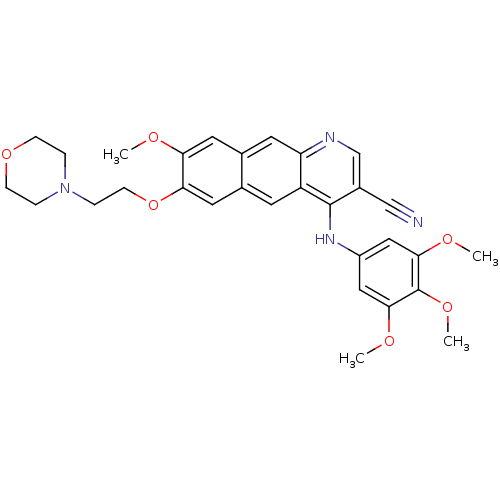
 3D Structure (crystal)
3D Structure (crystal)