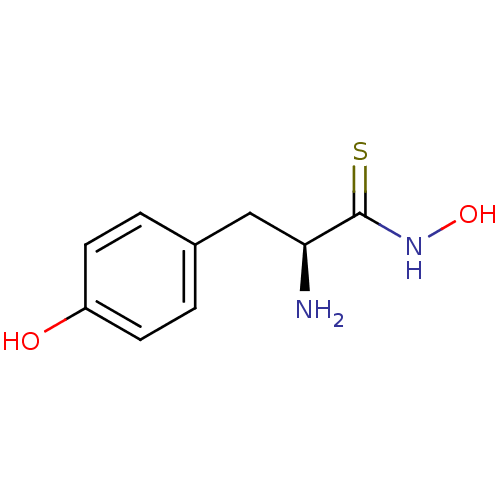
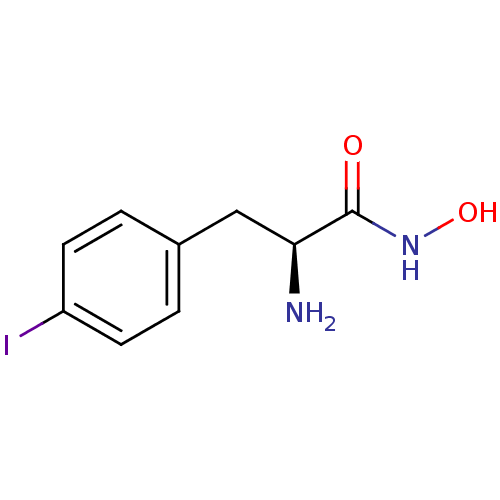
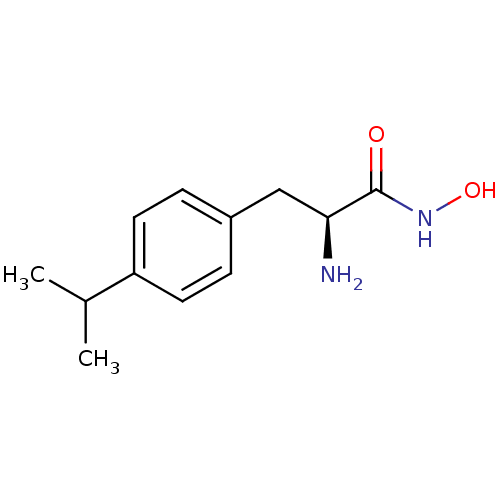
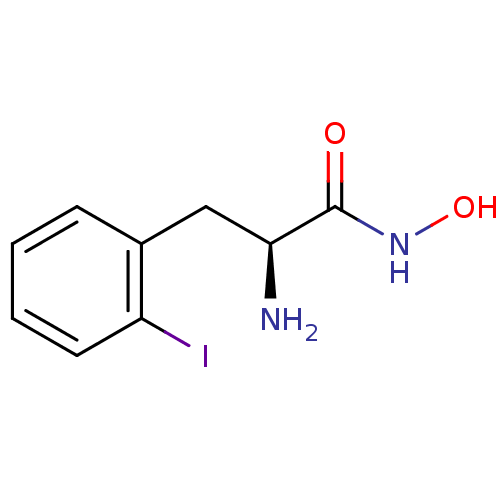
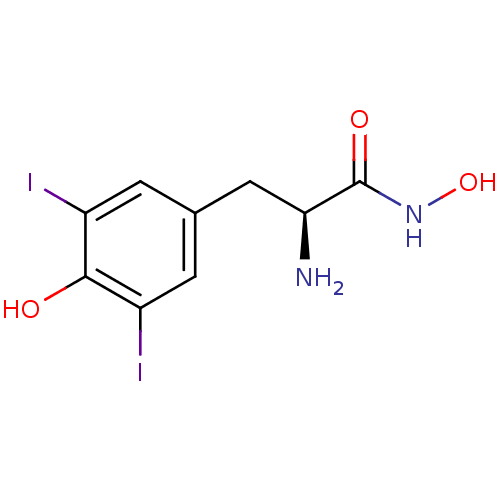
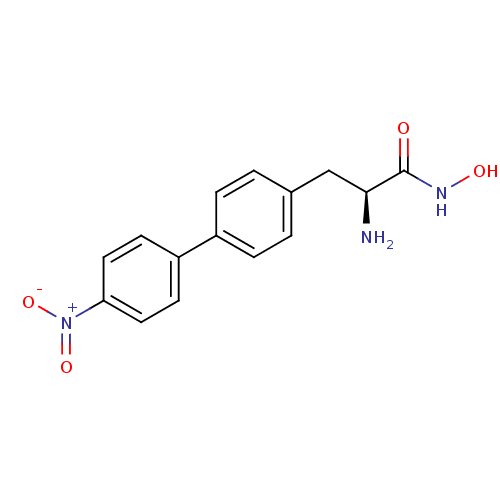
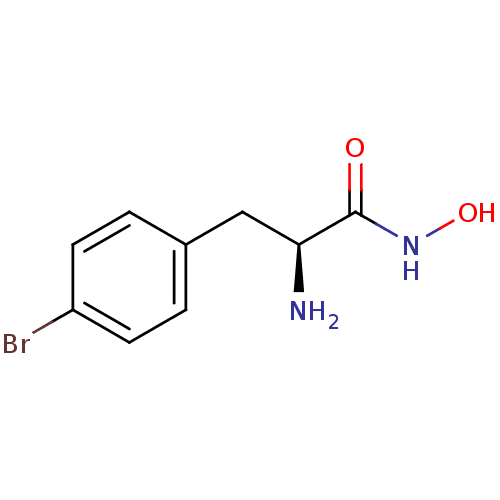
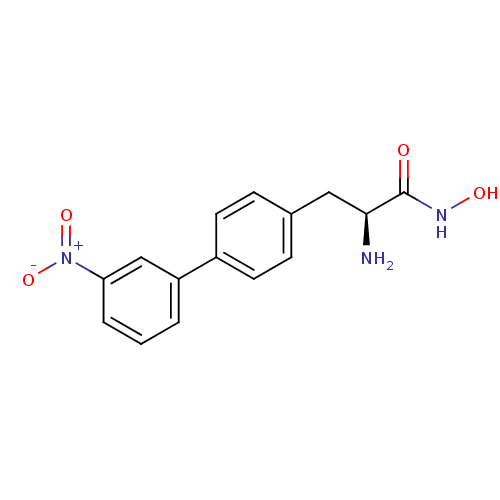
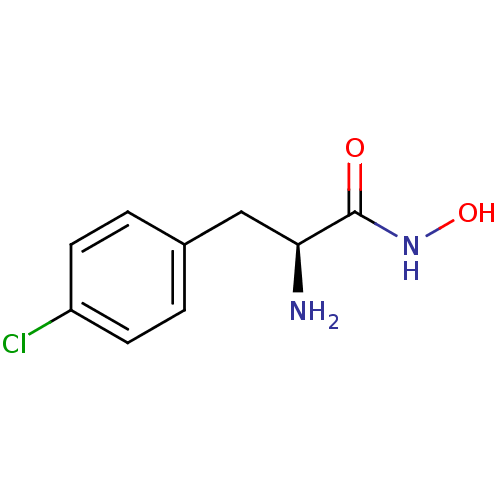
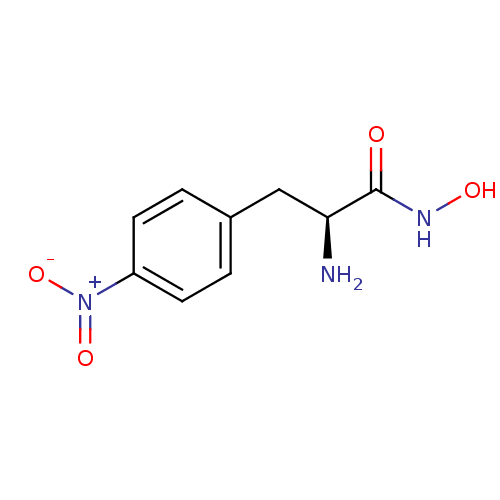
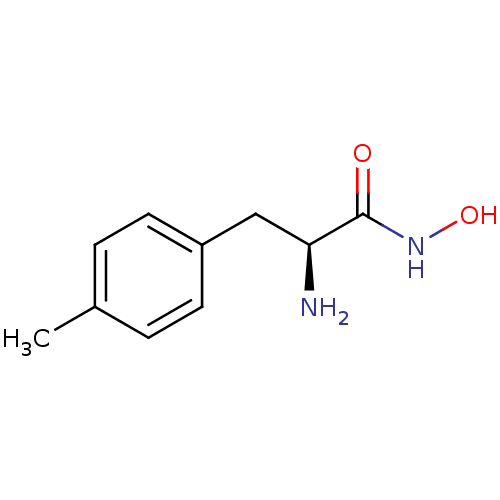
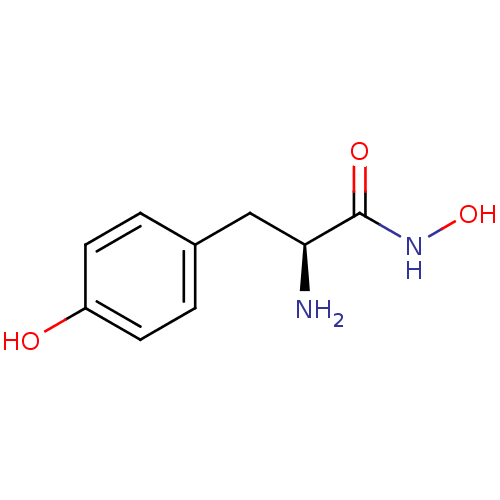
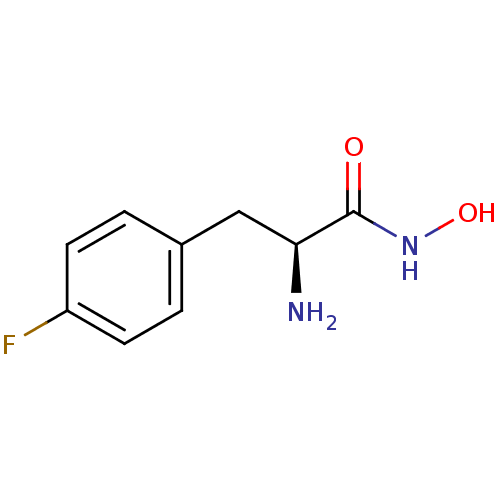
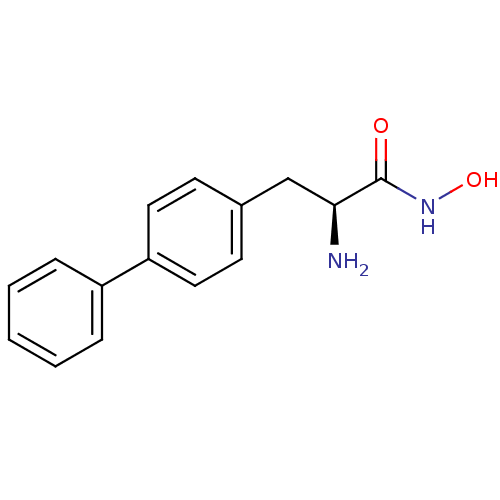
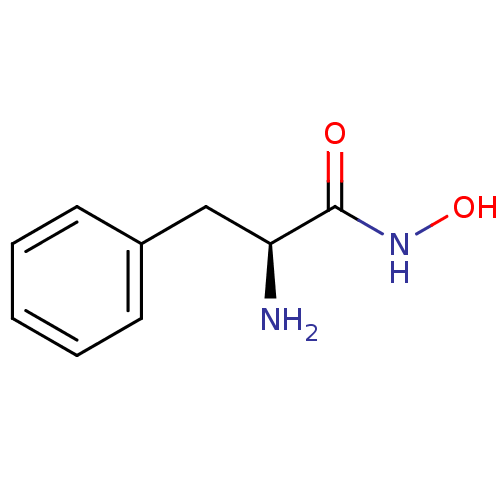
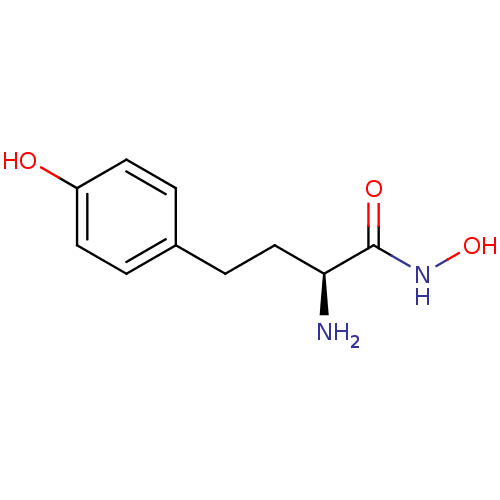
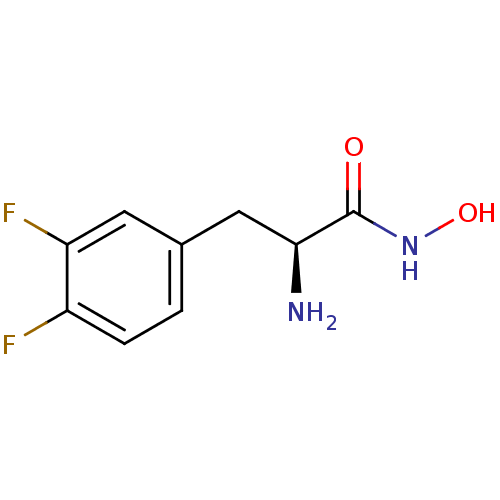
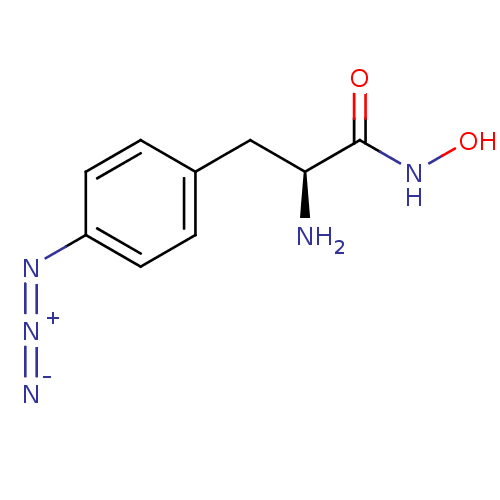
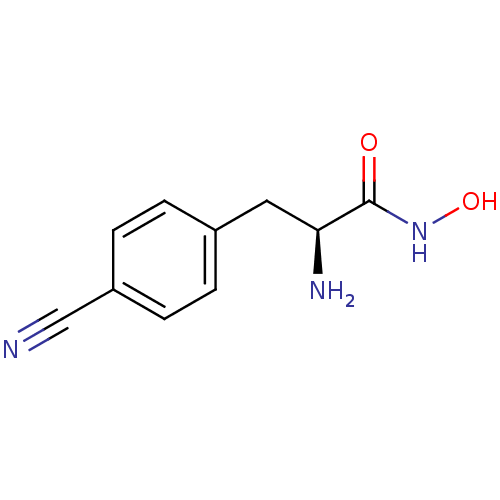
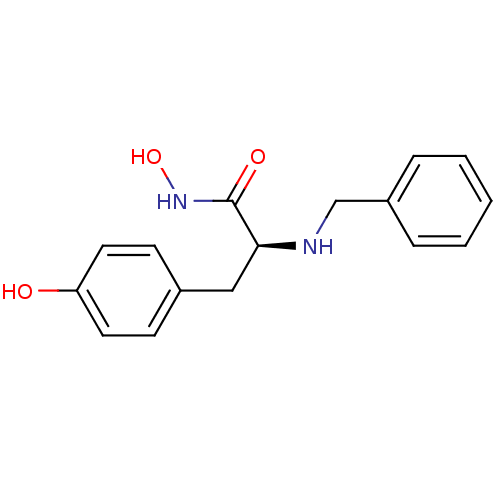
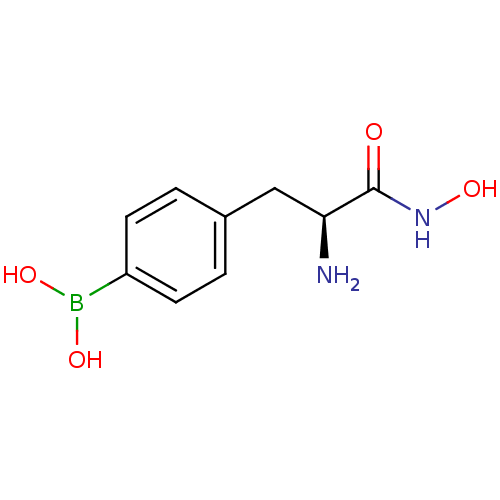
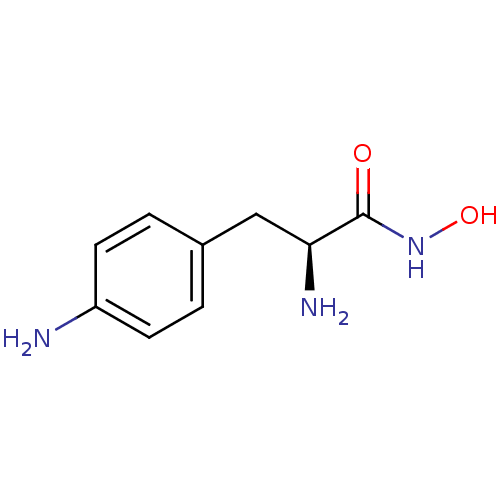
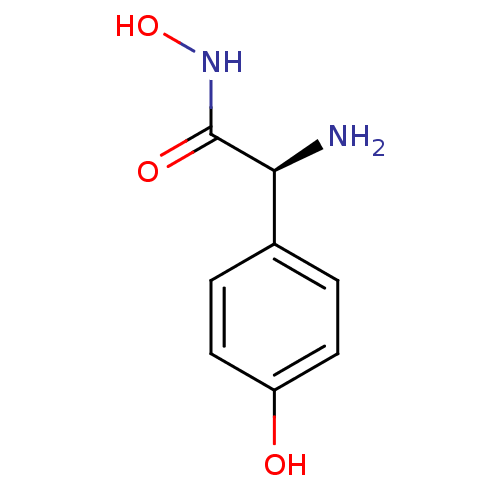
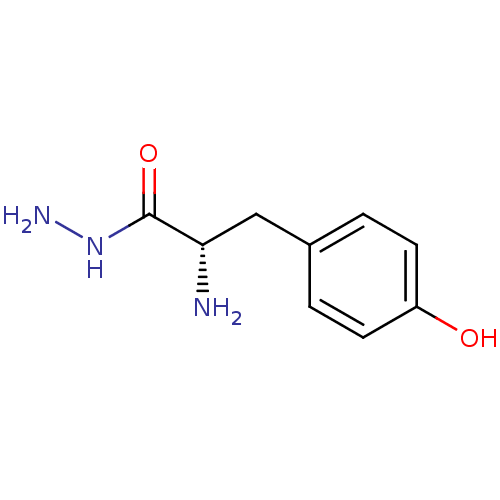
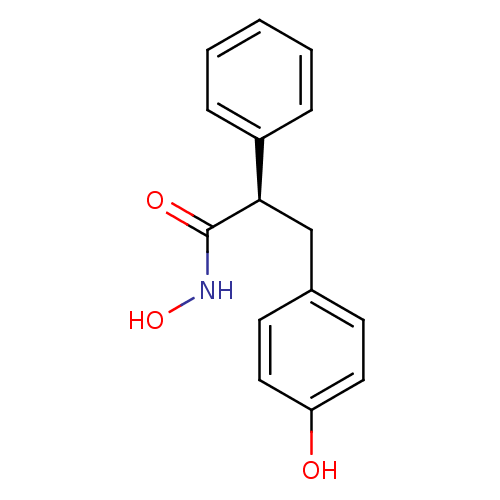
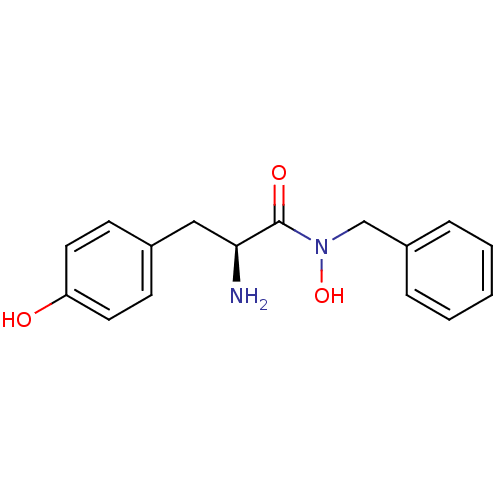
Report error Found 27 Enz. Inhib. hit(s) with all data for entry = 50018905
Affinity DataIC50: 2.50E+3nMAssay Description:Inhibition of CSK measured as poly-E4Y phosphorylation by acid precipitation assay in presence of 0.2 mM CoCl2More data for this Ligand-Target Pair
Affinity DataIC50: 3.20E+3nMAssay Description:Inhibition of CSK measured as poly-E4Y phosphorylation by acid precipitation assay in presence of 0.2 mM CoCl2More data for this Ligand-Target Pair
Affinity DataIC50: 3.30E+3nMAssay Description:Inhibition of CSK measured as poly-E4Y phosphorylation by acid precipitation assay in presence of 0.2 mM CoCl2More data for this Ligand-Target Pair
Affinity DataIC50: 3.40E+3nMAssay Description:Inhibition of CSK measured as poly-E4Y phosphorylation by acid precipitation assay in presence of 0.2 mM CoCl2More data for this Ligand-Target Pair
Affinity DataIC50: 3.40E+3nMAssay Description:Inhibition of CSK measured as poly-E4Y phosphorylation by acid precipitation assay in presence of 0.2 mM CoCl2More data for this Ligand-Target Pair
Affinity DataIC50: 4.90E+3nMAssay Description:Inhibition of CSK measured as poly-E4Y phosphorylation by acid precipitation assay in presence of 0.2 mM CoCl2More data for this Ligand-Target Pair
Affinity DataIC50: 5.60E+3nMAssay Description:Inhibition of CSK measured as poly-E4Y phosphorylation by acid precipitation assay in presence of 0.2 mM CoCl2More data for this Ligand-Target Pair
Affinity DataIC50: 6.10E+3nMAssay Description:Inhibition of CSK measured as poly-E4Y phosphorylation by acid precipitation assay in presence of 0.2 mM CoCl2More data for this Ligand-Target Pair
Affinity DataIC50: 6.30E+3nMAssay Description:Inhibition of CSK measured as poly-E4Y phosphorylation by acid precipitation assay in presence of 0.2 mM CoCl2More data for this Ligand-Target Pair
Affinity DataIC50: 7.00E+3nMAssay Description:Inhibition of CSK measured as poly-E4Y phosphorylation by acid precipitation assay in presence of 0.2 mM CoCl2More data for this Ligand-Target Pair
Affinity DataIC50: 9.50E+3nMAssay Description:Inhibition of CSK measured as poly-E4Y phosphorylation by acid precipitation assay in presence of 0.2 mM CoCl2More data for this Ligand-Target Pair
Affinity DataIC50: 9.50E+3nMAssay Description:Inhibition of CSK measured as poly-E4Y phosphorylation by acid precipitation assay in presence of 0.2 mM CoCl2More data for this Ligand-Target Pair
Affinity DataIC50: 1.10E+4nMAssay Description:Inhibition of CSK measured as poly-E4Y phosphorylation by acid precipitation assay in presence of 0.2 mM CoCl2More data for this Ligand-Target Pair
Affinity DataIC50: 1.20E+4nMAssay Description:Inhibition of CSK measured as poly-E4Y phosphorylation by acid precipitation assay in presence of 0.2 mM CoCl2More data for this Ligand-Target Pair
Affinity DataIC50: 1.55E+4nMAssay Description:Inhibition of CSK measured as poly-E4Y phosphorylation by acid precipitation assay in presence of 0.2 mM CoCl2More data for this Ligand-Target Pair
Affinity DataIC50: 1.80E+4nMAssay Description:Inhibition of CSK measured as poly-E4Y phosphorylation by acid precipitation assay in presence of 0.2 mM CoCl2More data for this Ligand-Target Pair
Affinity DataIC50: 1.80E+4nMAssay Description:Inhibition of CSK measured as poly-E4Y phosphorylation by acid precipitation assay in presence of 0.2 mM CoCl2More data for this Ligand-Target Pair
Affinity DataIC50: 2.10E+4nMAssay Description:Inhibition of CSK measured as poly-E4Y phosphorylation by acid precipitation assay in presence of 0.2 mM CoCl2More data for this Ligand-Target Pair
Affinity DataIC50: 2.80E+4nMAssay Description:Inhibition of CSK measured as poly-E4Y phosphorylation by acid precipitation assay in presence of 0.2 mM CoCl2More data for this Ligand-Target Pair
Affinity DataIC50: 2.90E+4nMAssay Description:Inhibition of CSK measured as poly-E4Y phosphorylation by acid precipitation assay in presence of 0.2 mM CoCl2More data for this Ligand-Target Pair
Affinity DataIC50: 3.40E+4nMAssay Description:Inhibition of CSK measured as poly-E4Y phosphorylation by acid precipitation assay in presence of 0.2 mM CoCl2More data for this Ligand-Target Pair
Affinity DataIC50: 4.70E+4nMAssay Description:Inhibition of CSK measured as poly-E4Y phosphorylation by acid precipitation assay in presence of 0.2 mM CoCl2More data for this Ligand-Target Pair
Affinity DataIC50: 5.70E+4nMAssay Description:Inhibition of CSK measured as poly-E4Y phosphorylation by acid precipitation assay in presence of 0.2 mM CoCl2More data for this Ligand-Target Pair
Affinity DataIC50: 7.50E+4nMAssay Description:Inhibition of CSK measured as poly-E4Y phosphorylation by acid precipitation assay in presence of 0.2 mM CoCl2More data for this Ligand-Target Pair
Affinity DataIC50: 2.90E+5nMAssay Description:Inhibition of CSK measured as poly-E4Y phosphorylation by acid precipitation assay in presence of 0.2 mM CoCl2More data for this Ligand-Target Pair
Affinity DataIC50: 2.91E+5nMAssay Description:Inhibition of CSK measured as poly-E4Y phosphorylation by acid precipitation assay in presence of 0.2 mM CoCl2More data for this Ligand-Target Pair