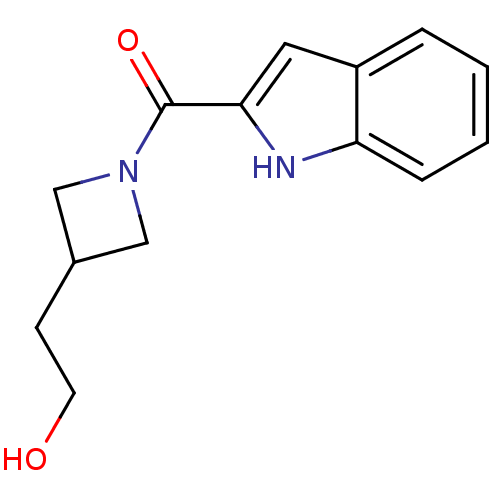
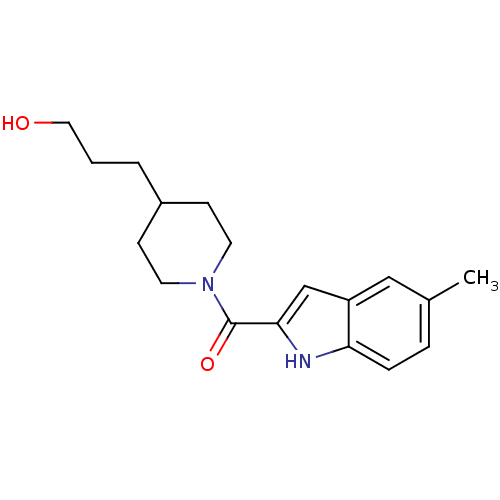
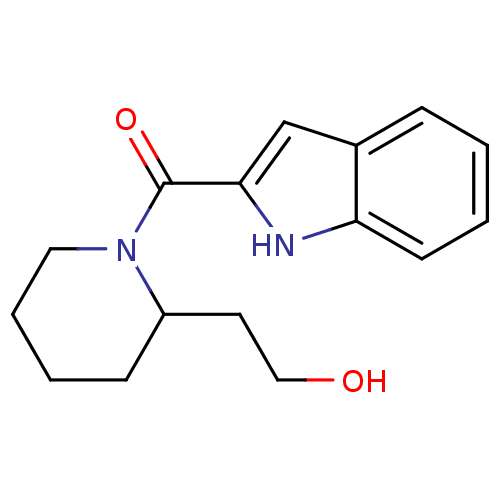
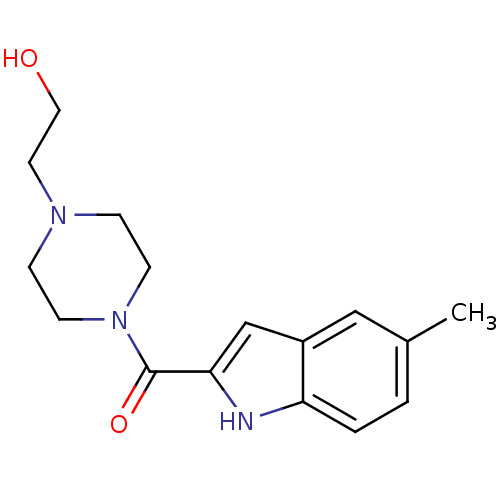
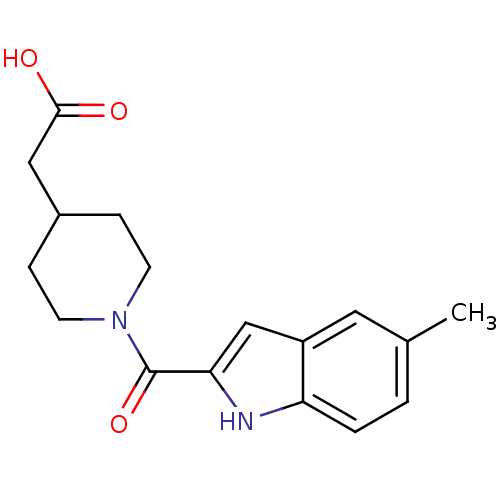
Report error Found 25 Enz. Inhib. hit(s) with all data for entry = 50043278
Affinity DataIC50: 0.340nMAssay Description:Inhibition of human 17beta-HSD5 expressed in HEK293 cells using androstenedione as substrate assessed as testosterone synthesis after 4 hrsMore data for this Ligand-Target Pair
Affinity DataIC50: 1.90nMAssay Description:Inhibition of human 17beta-HSD5 expressed in human CWR22R cells using androstenedione as substrate assessed as testosterone synthesis after 4 hrsMore data for this Ligand-Target Pair
Affinity DataIC50: 2.10nMAssay Description:Inhibition of human 17beta-HSD5 expressed in HEK293 cells using androstenedione as substrate assessed as testosterone synthesis after 4 hrsMore data for this Ligand-Target Pair
Affinity DataIC50: 4.5nMAssay Description:Inhibition of human 17beta-HSD5 expressed in HEK293 cells using androstenedione as substrate assessed as testosterone synthesis after 4 hrsMore data for this Ligand-Target Pair
Affinity DataIC50: 12nMAssay Description:Inhibition of human 17beta-HSD5 expressed in HEK293 cells using androstenedione as substrate assessed as testosterone synthesis after 4 hrsMore data for this Ligand-Target Pair
Affinity DataIC50: 13nMAssay Description:Inhibition of human 17beta-HSD5 expressed in HEK293 cells using androstenedione as substrate assessed as testosterone synthesis after 4 hrsMore data for this Ligand-Target Pair
Affinity DataIC50: 24nMAssay Description:Inhibition of human AKR1C3-mediated 9,10-phenanthrenequinone reduction after 10 to 20 mins by spectrophotometry in presence of NADPHMore data for this Ligand-Target Pair
Affinity DataIC50: 33nMAssay Description:Inhibition of human 17beta-HSD5 expressed in HEK293 cells using androstenedione as substrate assessed as testosterone synthesis after 4 hrsMore data for this Ligand-Target Pair
Affinity DataIC50: 37nMAssay Description:Inhibition of human 17beta-HSD5 expressed in HEK293 cells using androstenedione as substrate assessed as testosterone synthesis after 4 hrsMore data for this Ligand-Target Pair
Affinity DataIC50: 58nMAssay Description:Inhibition of human 17beta-HSD5 expressed in human CWR22R cells using androstenedione as substrate assessed as testosterone synthesis after 4 hrsMore data for this Ligand-Target Pair
Affinity DataIC50: 140nMAssay Description:Inhibition of human 17beta-HSD5 expressed in HEK293 cells using androstenedione as substrate assessed as testosterone synthesis after 4 hrsMore data for this Ligand-Target Pair
Affinity DataIC50: 160nMAssay Description:Inhibition of human 17beta-HSD5 expressed in HEK293 cells using androstenedione as substrate assessed as testosterone synthesis after 4 hrsMore data for this Ligand-Target Pair
Affinity DataIC50: 230nMAssay Description:Inhibition of human 17beta-HSD5 expressed in HEK293 cells using androstenedione as substrate assessed as testosterone synthesis after 4 hrsMore data for this Ligand-Target Pair
Affinity DataIC50: 770nMAssay Description:Inhibition of human 17beta-HSD5 expressed in HEK293 cells using androstenedione as substrate assessed as testosterone synthesis after 4 hrsMore data for this Ligand-Target Pair
Affinity DataIC50: 790nMAssay Description:Inhibition of human 17beta-HSD5 expressed in HEK293 cells using androstenedione as substrate assessed as testosterone synthesis after 4 hrsMore data for this Ligand-Target Pair
Affinity DataIC50: 1.10E+3nMAssay Description:Inhibition of human 17beta-HSD5 expressed in HEK293 cells using androstenedione as substrate assessed as testosterone synthesis after 4 hrsMore data for this Ligand-Target Pair
Affinity DataIC50: 1.20E+3nMAssay Description:Inhibition of human 17beta-HSD5 expressed in HEK293 cells using androstenedione as substrate assessed as testosterone synthesis after 4 hrsMore data for this Ligand-Target Pair
Affinity DataIC50: 2.10E+3nMAssay Description:Inhibition of human 17beta-HSD5 expressed in HEK293 cells using androstenedione as substrate assessed as testosterone synthesis after 4 hrsMore data for this Ligand-Target Pair
Affinity DataIC50: 2.80E+3nMAssay Description:Inhibition of human 17beta-HSD5 expressed in HEK293 cells using androstenedione as substrate assessed as testosterone synthesis after 4 hrsMore data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMAssay Description:Inhibition of human 17beta-HSD5 expressed in HEK293 cells using androstenedione as substrate assessed as testosterone synthesis after 4 hrsMore data for this Ligand-Target Pair
Affinity DataIC50: 2.00E+4nMAssay Description:Inhibition of human AKR1C2-mediated 9,10-phenanthrenequinone reduction after 10 to 20 mins by spectrophotometry in presence of NADPHMore data for this Ligand-Target Pair
Affinity DataIC50: 2.50E+4nMAssay Description:Inhibition of CYP2C9 (unknown origin)More data for this Ligand-Target Pair
Affinity DataIC50: 2.50E+4nMAssay Description:Inhibition of CYP2C19 (unknown origin)More data for this Ligand-Target Pair
Affinity DataIC50: 2.50E+4nMAssay Description:Inhibition of CYP2D6 (unknown origin)More data for this Ligand-Target Pair
Affinity DataIC50: 2.50E+4nMAssay Description:Inhibition of CYP1A2 (unknown origin)More data for this Ligand-Target Pair


 3D Structure (crystal)
3D Structure (crystal)