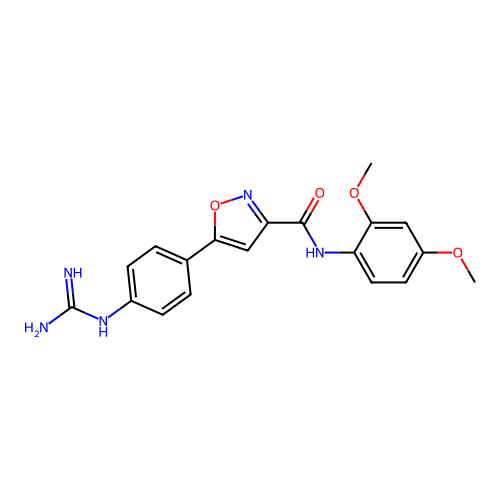
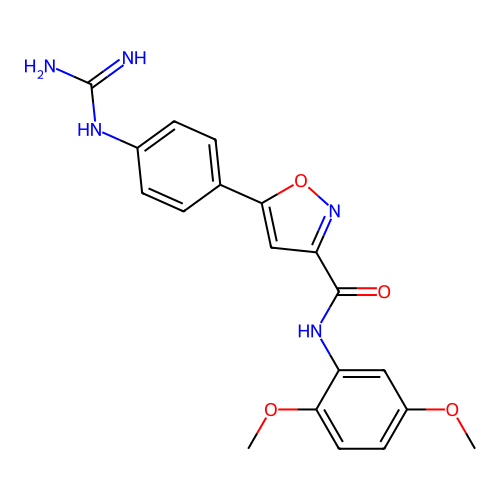
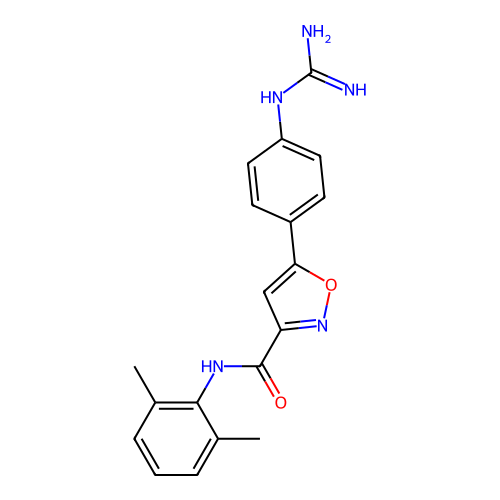
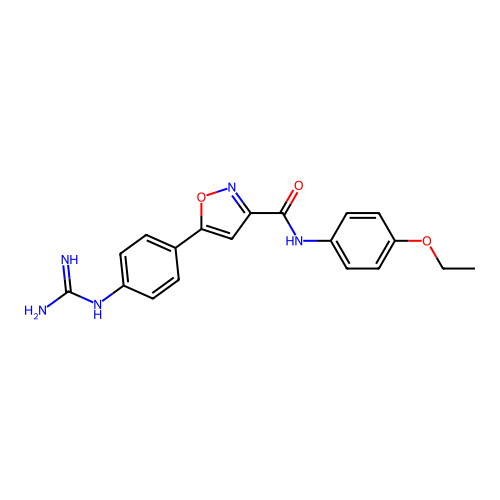
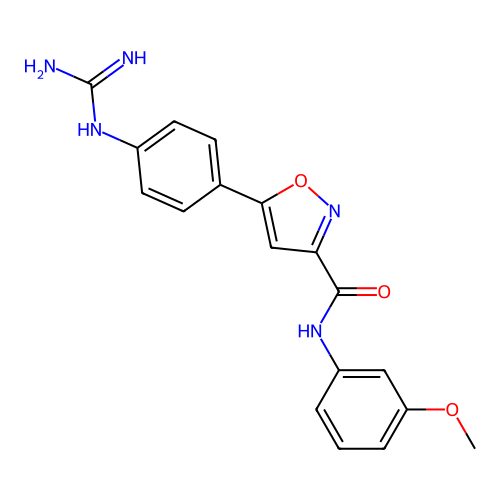
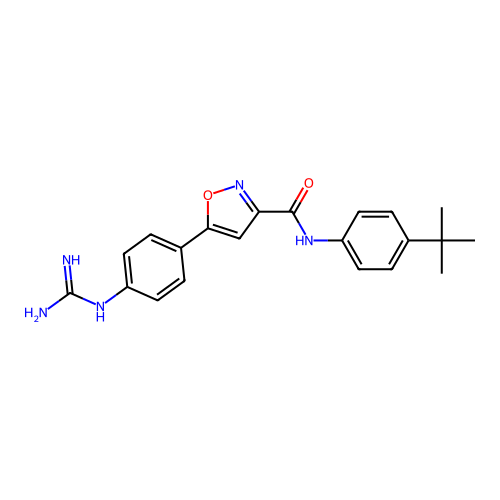
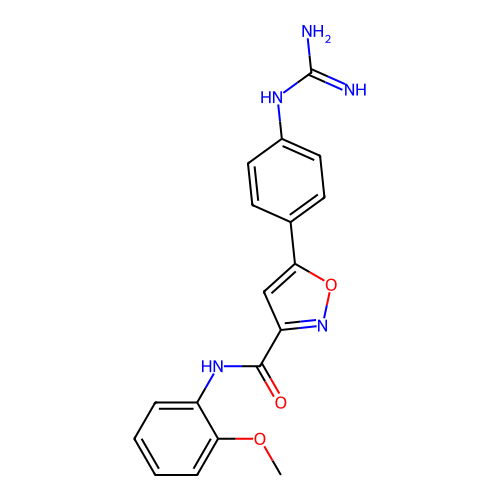
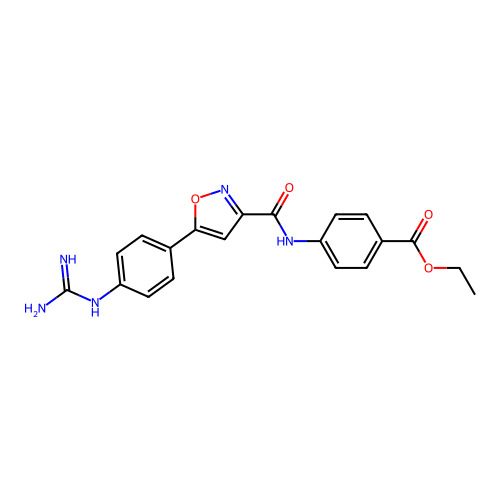
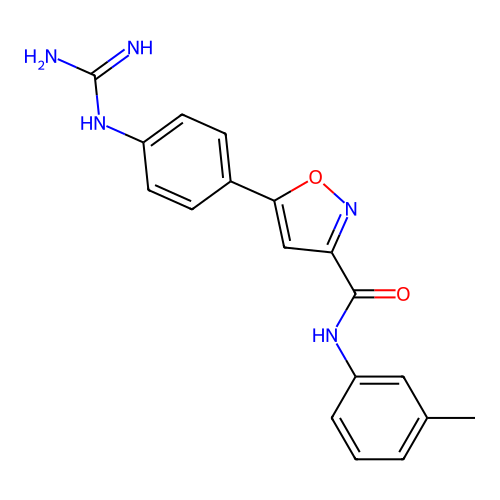
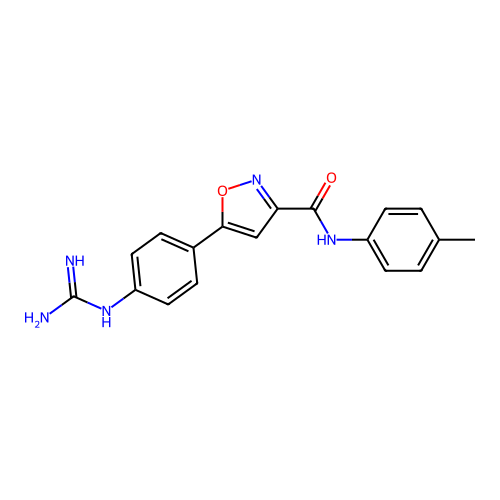
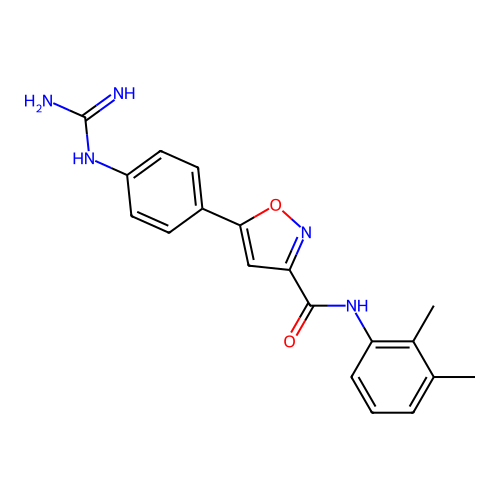
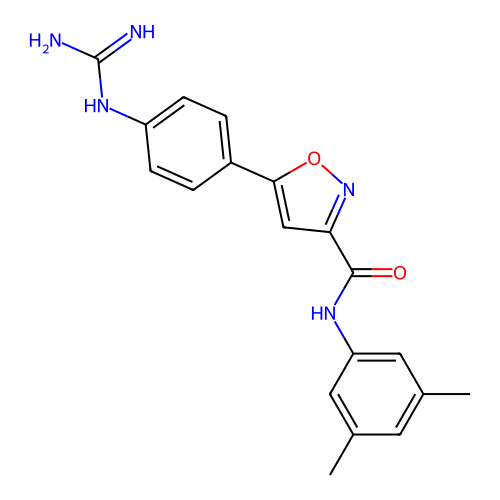
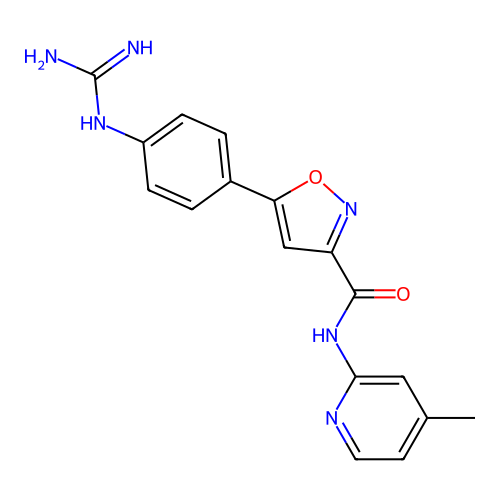
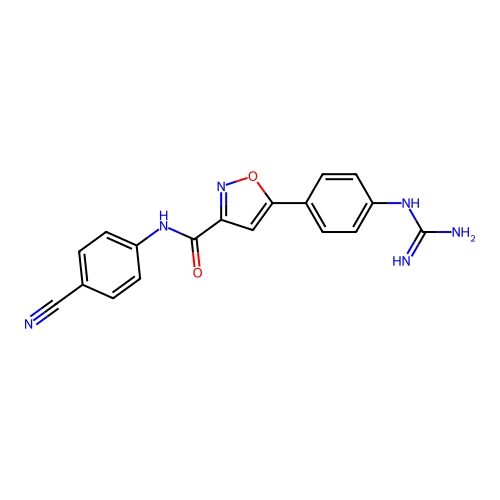
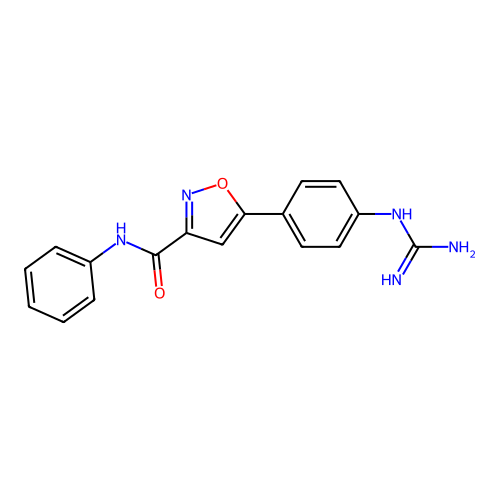
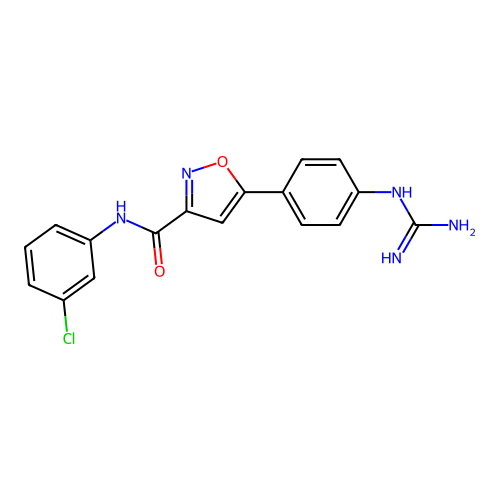
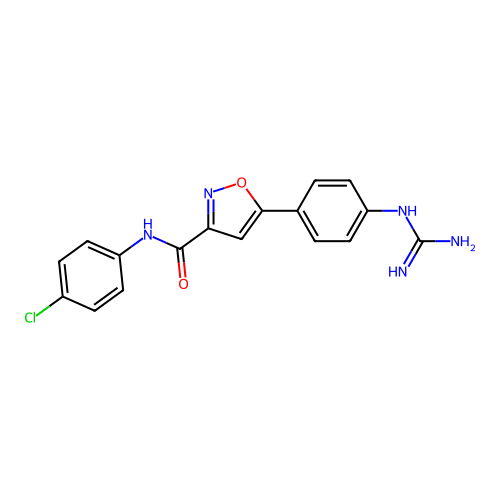
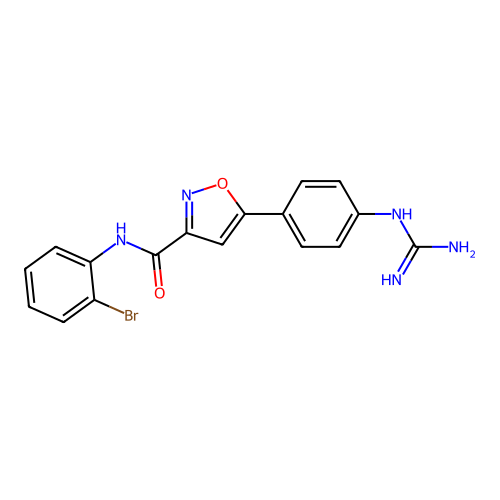
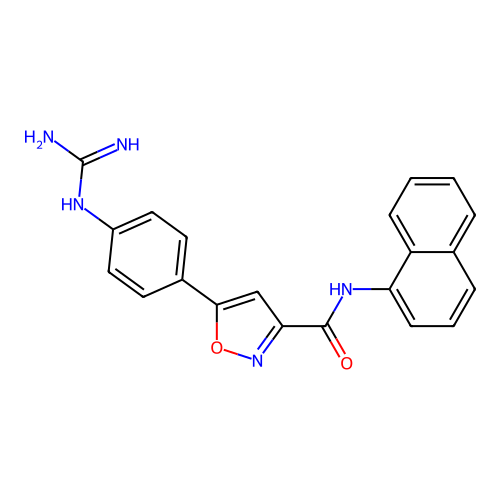
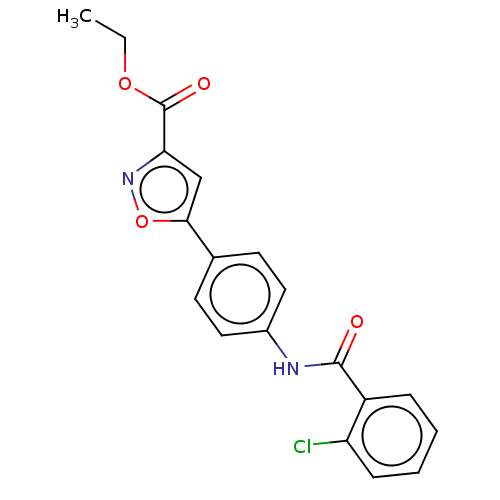
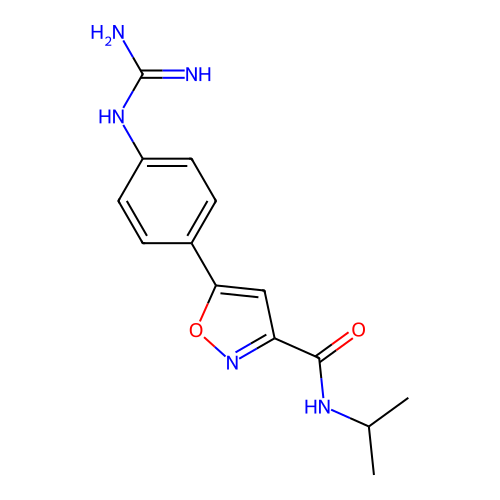
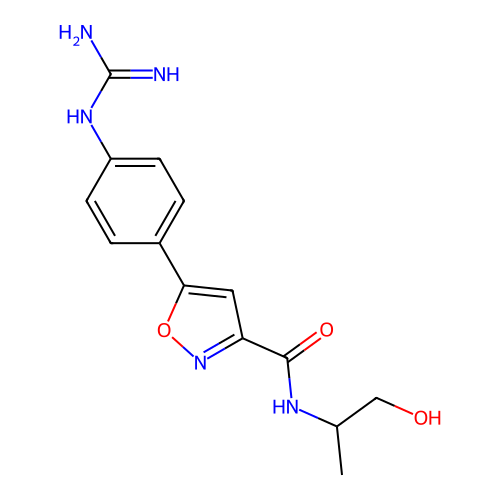
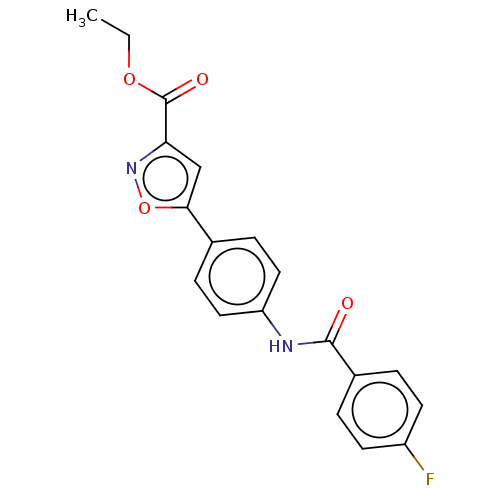
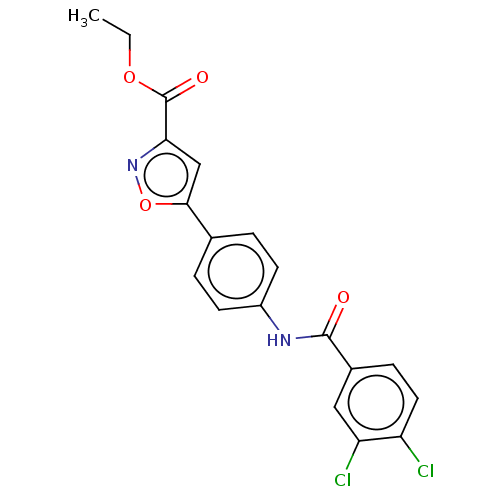
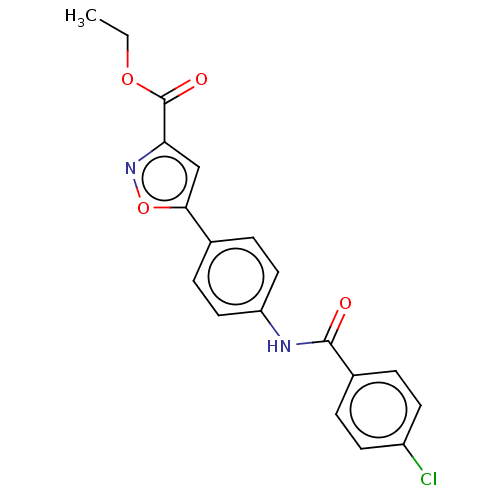
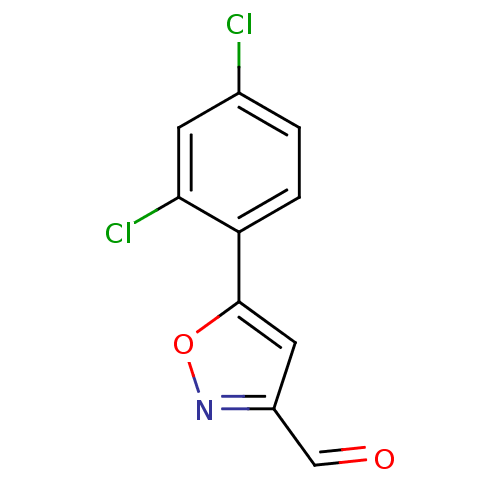
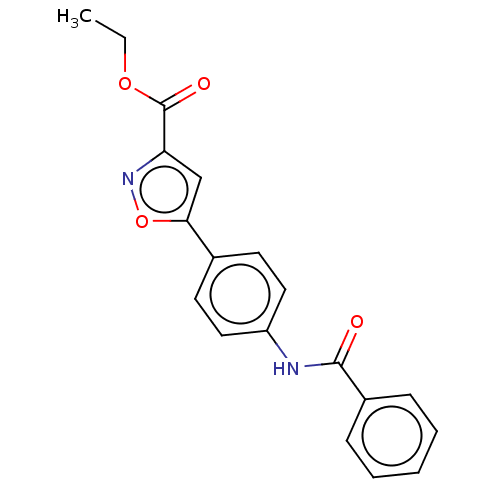
Report error Found 31 Enz. Inhib. hit(s) with all data for entry = 50044489
Affinity DataIC50: 1.40E+3nMAssay Description:Inhibition of human acrosin using N-alpha-benzoyl-L-arginine p-nitroanilide as substrate after 3 hrs by spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 2.30E+3nMAssay Description:Inhibition of human acrosin using N-alpha-benzoyl-L-arginine p-nitroanilide as substrate after 3 hrs by spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 5.70E+3nMAssay Description:Inhibition of human acrosin using N-alpha-benzoyl-L-arginine p-nitroanilide as substrate after 3 hrs by spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 6.40E+3nMAssay Description:Inhibition of human acrosin using N-alpha-benzoyl-L-arginine p-nitroanilide as substrate after 3 hrs by spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 6.60E+3nMAssay Description:Inhibition of human acrosin using N-alpha-benzoyl-L-arginine p-nitroanilide as substrate after 3 hrs by spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 7.80E+3nMAssay Description:Inhibition of human acrosin using N-alpha-benzoyl-L-arginine p-nitroanilide as substrate after 3 hrs by spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 8.40E+3nMAssay Description:Inhibition of human acrosin using N-alpha-benzoyl-L-arginine p-nitroanilide as substrate after 3 hrs by spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 9.30E+3nMAssay Description:Inhibition of human acrosin using N-alpha-benzoyl-L-arginine p-nitroanilide as substrate after 3 hrs by spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 9.40E+3nMAssay Description:Inhibition of human acrosin using N-alpha-benzoyl-L-arginine p-nitroanilide as substrate after 3 hrs by spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 1.01E+4nMAssay Description:Inhibition of human acrosin using N-alpha-benzoyl-L-arginine p-nitroanilide as substrate after 3 hrs by spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 1.15E+4nMAssay Description:Inhibition of human acrosin using N-alpha-benzoyl-L-arginine p-nitroanilide as substrate after 3 hrs by spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 1.21E+4nMAssay Description:Inhibition of human acrosin using N-alpha-benzoyl-L-arginine p-nitroanilide as substrate after 3 hrs by spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 1.44E+4nMAssay Description:Inhibition of human acrosin using N-alpha-benzoyl-L-arginine p-nitroanilide as substrate after 3 hrs by spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 1.44E+4nMAssay Description:Inhibition of human acrosin using N-alpha-benzoyl-L-arginine p-nitroanilide as substrate after 3 hrs by spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 2.03E+4nMAssay Description:Inhibition of human acrosin using N-alpha-benzoyl-L-arginine p-nitroanilide as substrate after 3 hrs by spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 2.05E+4nMAssay Description:Inhibition of human acrosin using N-alpha-benzoyl-L-arginine p-nitroanilide as substrate after 3 hrs by spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 2.14E+4nMAssay Description:Inhibition of human acrosin using N-alpha-benzoyl-L-arginine p-nitroanilide as substrate after 3 hrs by spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 2.39E+4nMAssay Description:Inhibition of human acrosin using N-alpha-benzoyl-L-arginine p-nitroanilide as substrate after 3 hrs by spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 2.46E+4nMAssay Description:Inhibition of human acrosin using N-alpha-benzoyl-L-arginine p-nitroanilide as substrate after 3 hrs by spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 2.54E+4nMAssay Description:Inhibition of human acrosin using N-alpha-benzoyl-L-arginine p-nitroanilide as substrate after 3 hrs by spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 2.68E+4nMAssay Description:Inhibition of human acrosin using N-alpha-benzoyl-L-arginine p-nitroanilide as substrate after 3 hrs by spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 2.79E+4nMAssay Description:Inhibition of human acrosin using N-alpha-benzoyl-L-arginine p-nitroanilide as substrate after 3 hrs by spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 2.99E+4nMAssay Description:Inhibition of human acrosin using N-alpha-benzoyl-L-arginine p-nitroanilide as substrate after 3 hrs by spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 6.00E+4nMAssay Description:Inhibition of human acrosin using N-alpha-benzoyl-L-arginine p-nitroanilide as substrate after 3 hrs by spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+5nMAssay Description:Inhibition of human acrosin using N-alpha-benzoyl-L-arginine p-nitroanilide as substrate after 3 hrs by spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+5nMAssay Description:Inhibition of human acrosin using N-alpha-benzoyl-L-arginine p-nitroanilide as substrate after 3 hrs by spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 1.40E+5nMAssay Description:Inhibition of human acrosin using N-alpha-benzoyl-L-arginine p-nitroanilide as substrate after 3 hrs by spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 2.40E+5nMAssay Description:Inhibition of human acrosin using N-alpha-benzoyl-L-arginine p-nitroanilide as substrate after 3 hrs by spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 3.80E+5nMAssay Description:Inhibition of human acrosin using N-alpha-benzoyl-L-arginine p-nitroanilide as substrate after 3 hrs by spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 3.90E+5nMAssay Description:Inhibition of human acrosin using N-alpha-benzoyl-L-arginine p-nitroanilide as substrate after 3 hrs by spectrophotometryMore data for this Ligand-Target Pair
Affinity DataIC50: 4.60E+5nMAssay Description:Inhibition of human acrosin using N-alpha-benzoyl-L-arginine p-nitroanilide as substrate after 3 hrs by spectrophotometryMore data for this Ligand-Target Pair