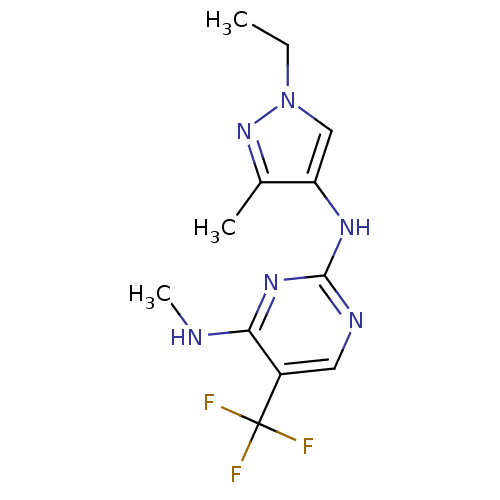
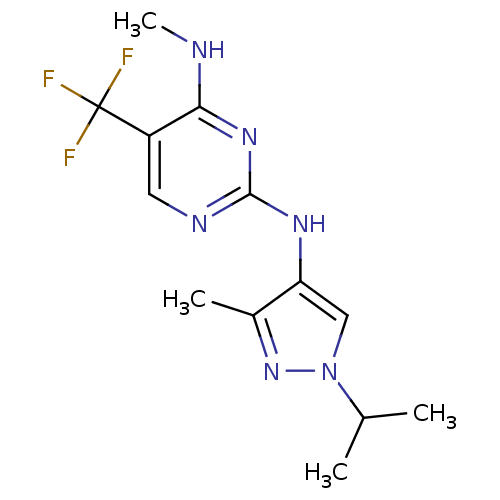
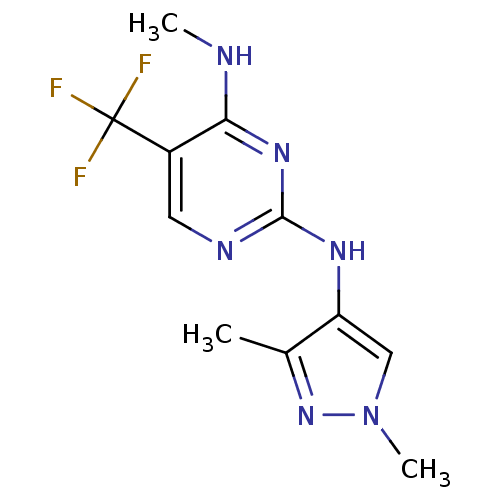
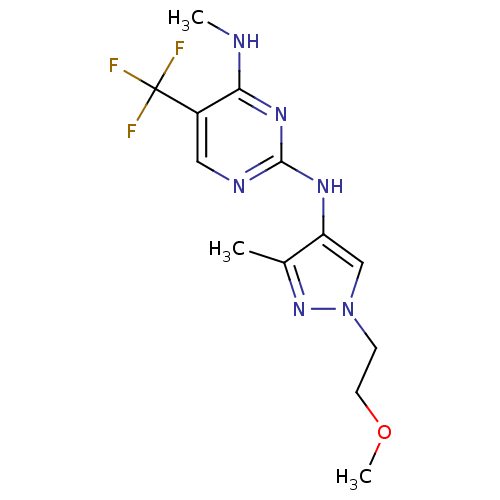
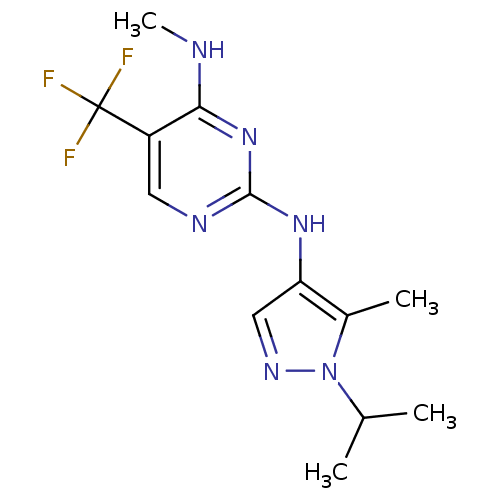
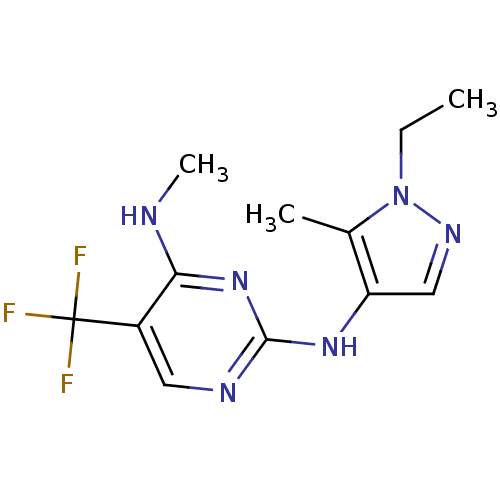
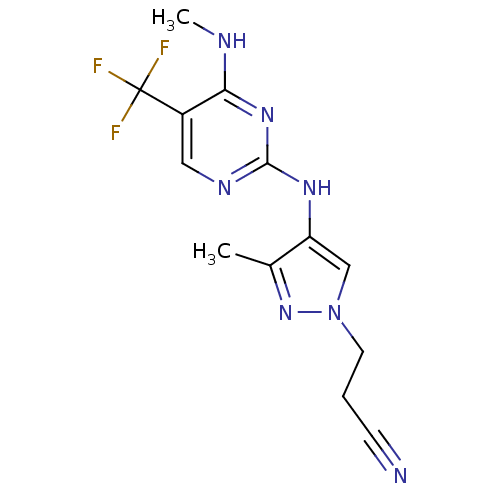
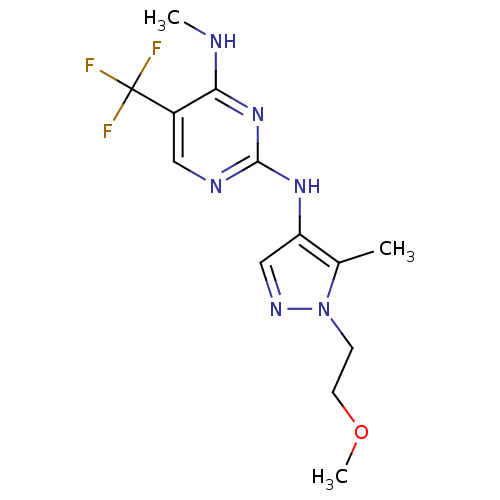
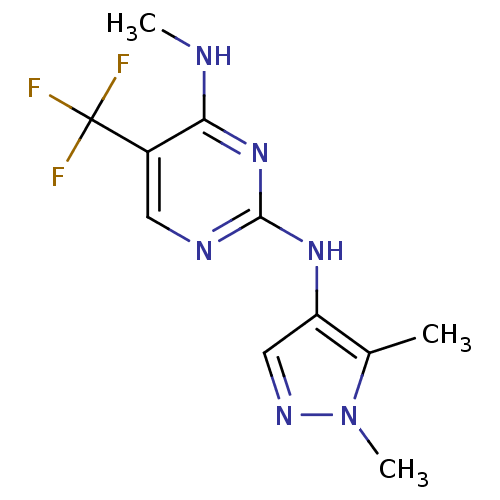
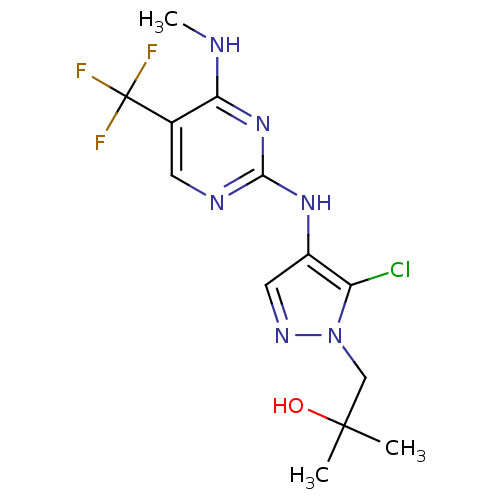
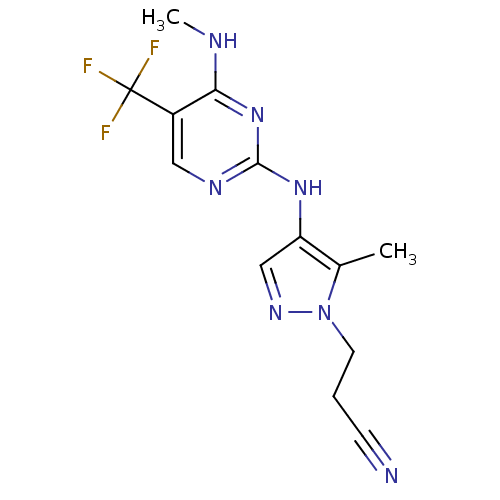
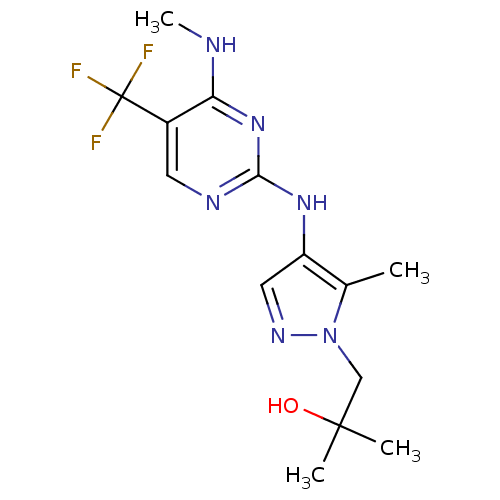
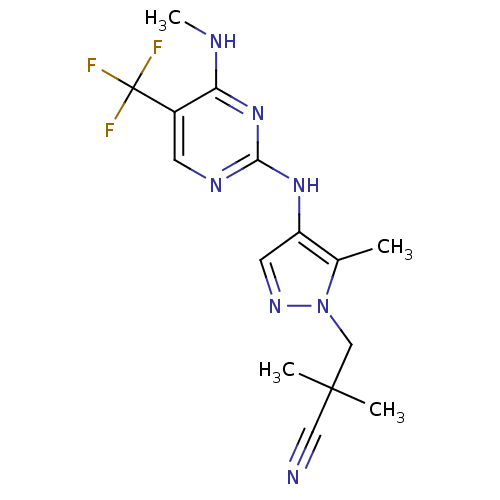
Report error Found 14 Enz. Inhib. hit(s) with all data for entry = 50042453
Affinity DataIC50: 400nMAssay Description:Reversible inhibition of CYP1A2 (unknown origin) using phenacetin as substrate by LC-MS/MS analysisMore data for this Ligand-Target Pair
Affinity DataIC50: 700nMAssay Description:Reversible inhibition of CYP1A2 (unknown origin) using phenacetin as substrate by LC-MS/MS analysisMore data for this Ligand-Target Pair
Affinity DataIC50: 700nMAssay Description:Reversible inhibition of CYP1A2 (unknown origin) using phenacetin as substrate by LC-MS/MS analysisMore data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+3nMAssay Description:Reversible inhibition of CYP1A2 (unknown origin) using phenacetin as substrate by LC-MS/MS analysisMore data for this Ligand-Target Pair
Affinity DataIC50: 3.10E+3nMAssay Description:Reversible inhibition of CYP1A2 (unknown origin) using phenacetin as substrate by LC-MS/MS analysisMore data for this Ligand-Target Pair
Affinity DataIC50: 4.70E+3nMAssay Description:Reversible inhibition of CYP1A2 (unknown origin) using phenacetin as substrate by LC-MS/MS analysisMore data for this Ligand-Target Pair
Affinity DataIC50: 5.20E+3nMAssay Description:Reversible inhibition of CYP1A2 (unknown origin) using phenacetin as substrate by LC-MS/MS analysisMore data for this Ligand-Target Pair
Affinity DataIC50: 5.80E+3nMAssay Description:Reversible inhibition of CYP1A2 (unknown origin) using phenacetin as substrate by LC-MS/MS analysisMore data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMAssay Description:Reversible inhibition of CYP1A2 (unknown origin) using phenacetin as substrate by LC-MS/MS analysisMore data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMAssay Description:Reversible inhibition of CYP1A2 (unknown origin) using phenacetin as substrate by LC-MS/MS analysisMore data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMAssay Description:Reversible inhibition of CYP1A2 (unknown origin) using phenacetin as substrate by LC-MS/MS analysisMore data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMAssay Description:Reversible inhibition of CYP1A2 (unknown origin) using phenacetin as substrate by LC-MS/MS analysisMore data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMAssay Description:Reversible inhibition of CYP1A2 (unknown origin) using phenacetin as substrate by LC-MS/MS analysisMore data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMAssay Description:Reversible inhibition of CYP1A2 (unknown origin) using phenacetin as substrate by LC-MS/MS analysisMore data for this Ligand-Target Pair