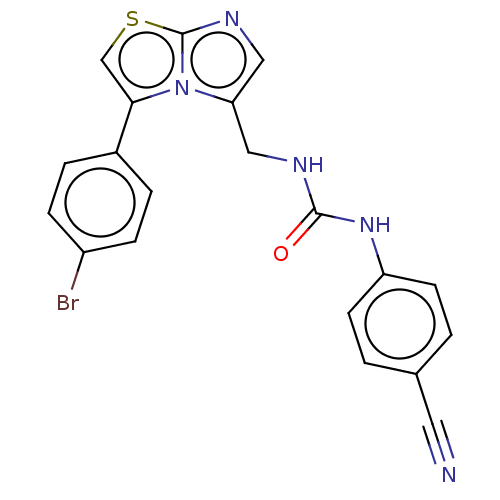
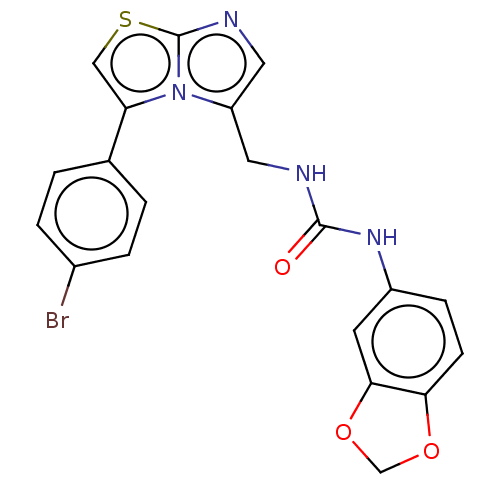
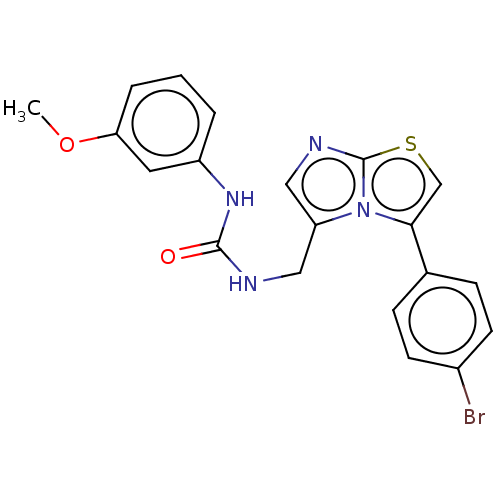
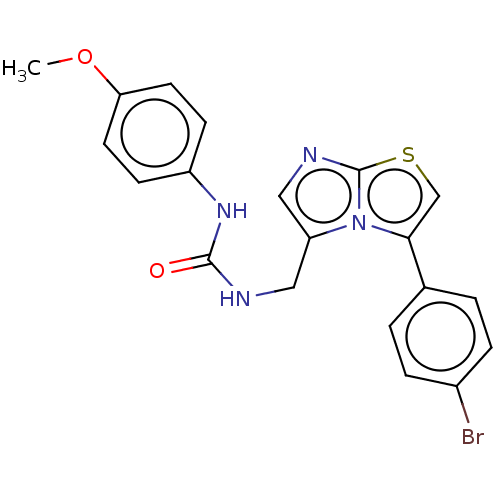
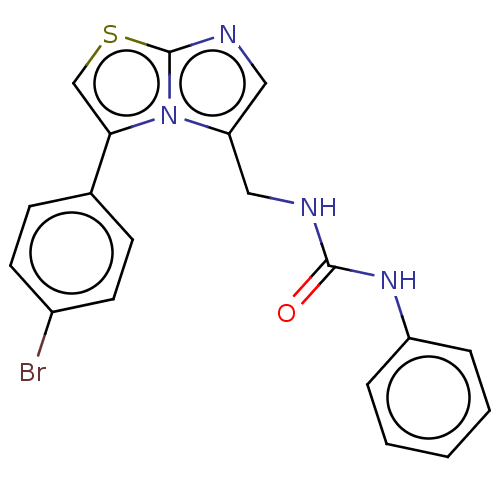
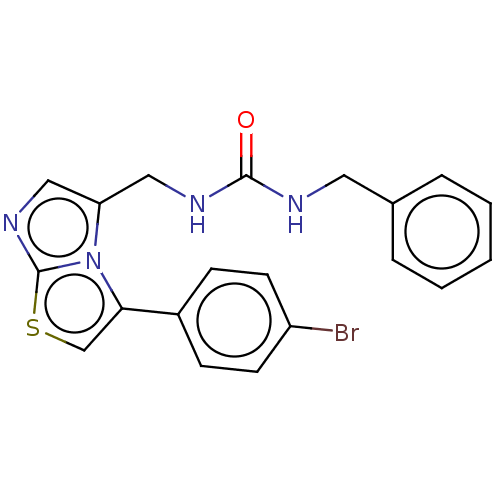
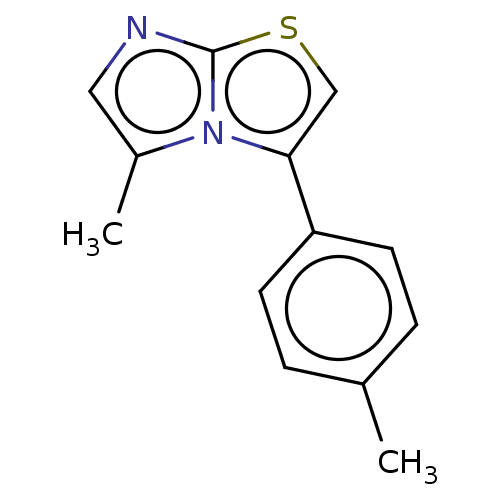
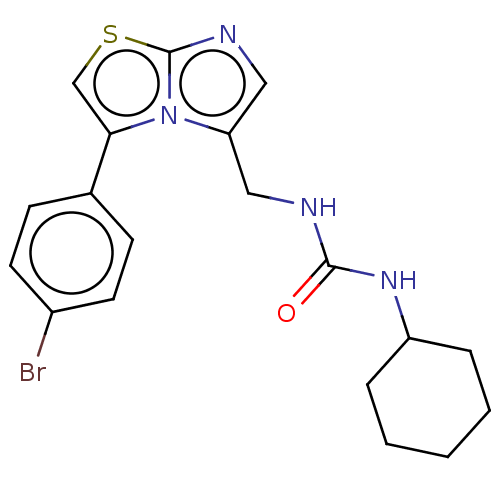
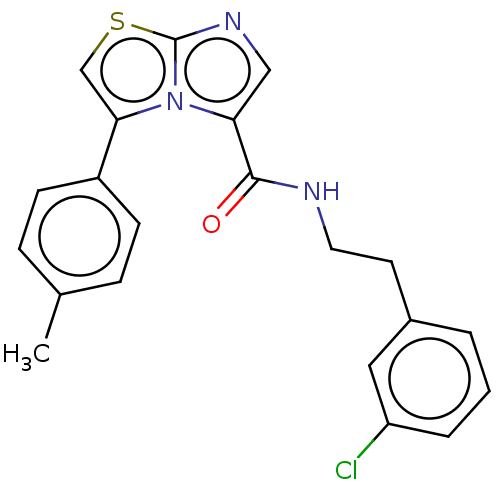
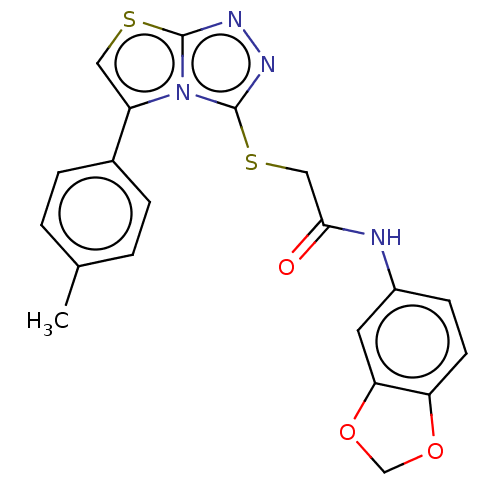
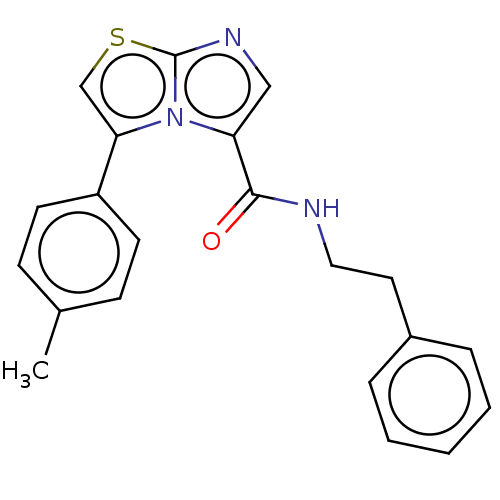
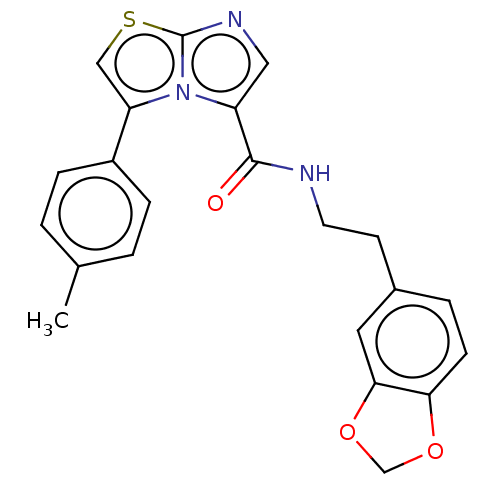
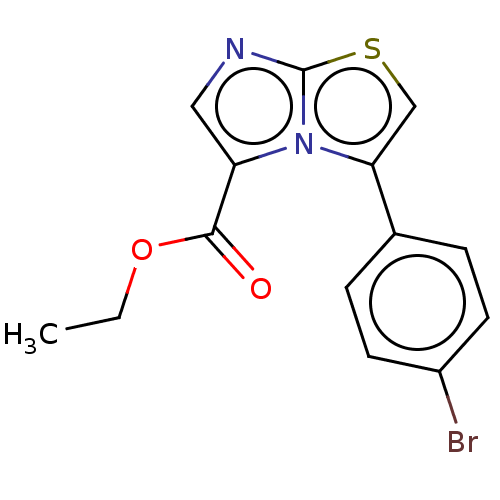
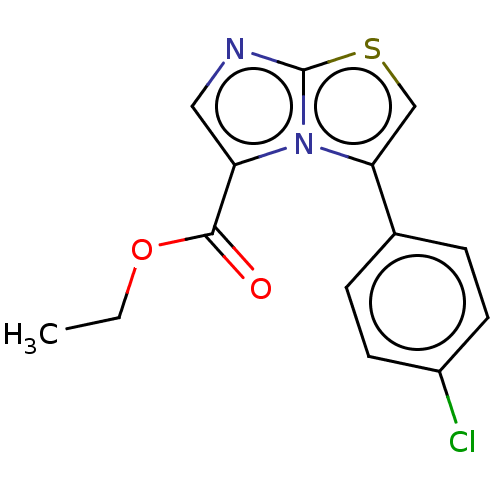
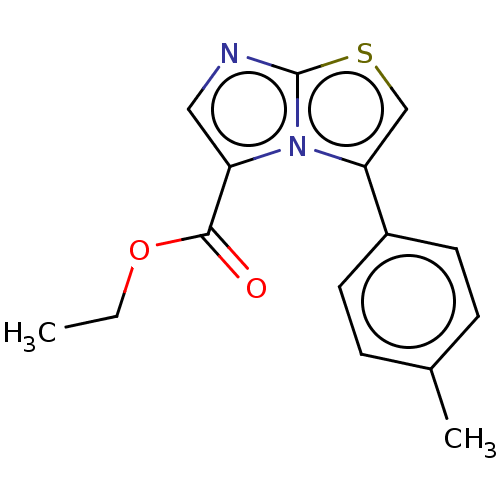
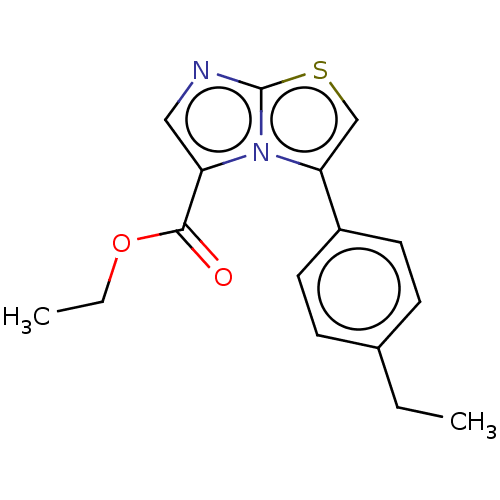
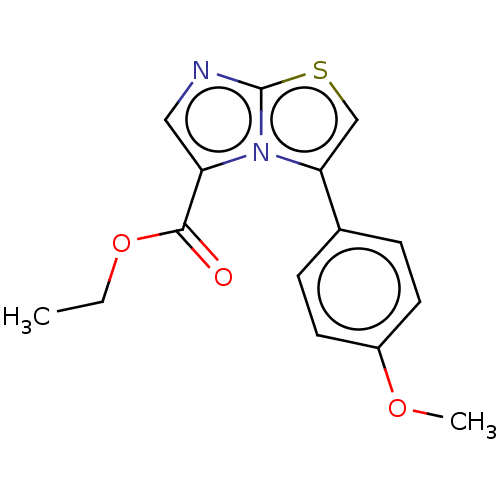
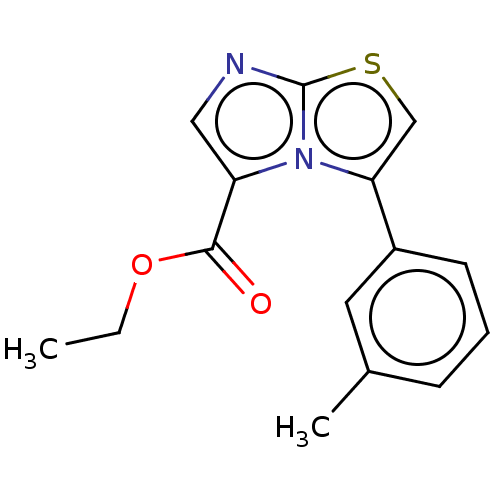
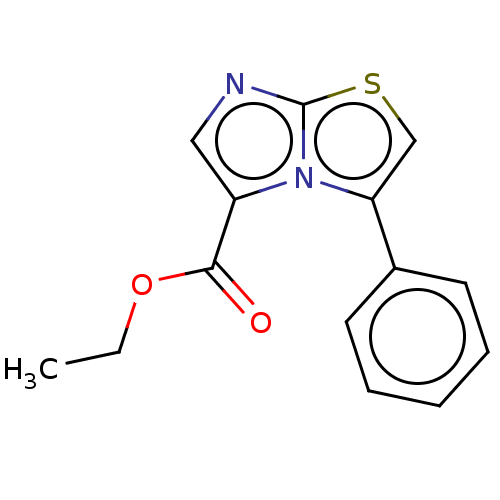
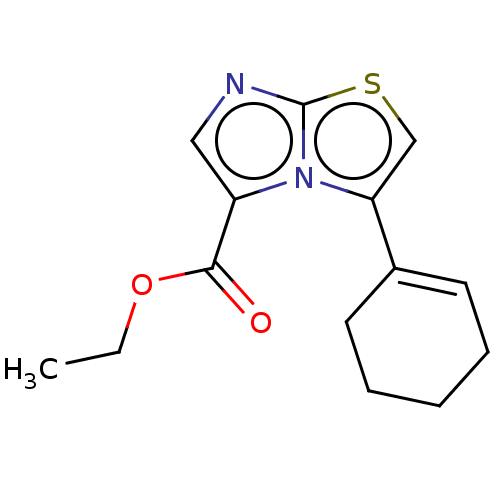
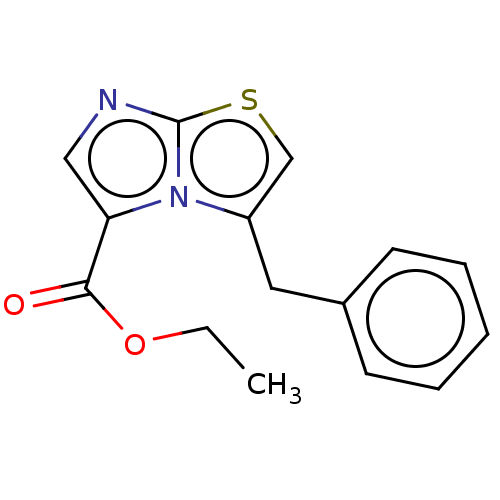
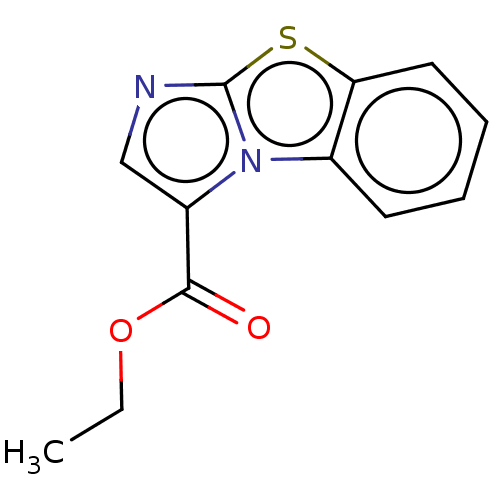
Report error Found 23 Enz. Inhib. hit(s) with all data for entry = 50044799
Affinity DataIC50: 77nMAssay Description:Inhibition of human IDO1 assessed as reduction in convertion of N-formylkynurenine to kynurenine incubated for 30 minsMore data for this Ligand-Target Pair
Affinity DataIC50: 120nMAssay Description:Inhibition of human IDO1 assessed as reduction in convertion of N-formylkynurenine to kynurenine incubated for 30 minsMore data for this Ligand-Target Pair
Affinity DataIC50: 160nMAssay Description:Inhibition of human IDO1 assessed as reduction in convertion of N-formylkynurenine to kynurenine incubated for 30 minsMore data for this Ligand-Target Pair
Affinity DataIC50: 190nMAssay Description:Inhibition of human IDO1 assessed as reduction in convertion of N-formylkynurenine to kynurenine incubated for 30 minsMore data for this Ligand-Target Pair
Affinity DataIC50: 220nMAssay Description:Inhibition of human IDO1 assessed as reduction in convertion of N-formylkynurenine to kynurenine incubated for 30 minsMore data for this Ligand-Target Pair
Affinity DataIC50: 750nMAssay Description:Inhibition of human IDO1 assessed as reduction in convertion of N-formylkynurenine to kynurenine incubated for 30 minsMore data for this Ligand-Target Pair
Affinity DataIC50: 1.30E+3nMAssay Description:Inhibition of human IDO1 assessed as reduction in convertion of N-formylkynurenine to kynurenine incubated for 30 minsMore data for this Ligand-Target Pair
Affinity DataIC50: 1.80E+3nMAssay Description:Inhibition of human IDO1 assessed as reduction in convertion of N-formylkynurenine to kynurenine incubated for 30 minsMore data for this Ligand-Target Pair
Affinity DataIC50: 1.90E+3nMAssay Description:Inhibition of human IDO1 assessed as reduction in convertion of N-formylkynurenine to kynurenine incubated for 30 minsMore data for this Ligand-Target Pair
Affinity DataIC50: 3.00E+3nMAssay Description:Inhibition of human IDO1 assessed as reduction in convertion of N-formylkynurenine to kynurenine incubated for 30 minsMore data for this Ligand-Target Pair
Affinity DataIC50: 3.50E+3nMAssay Description:Inhibition of human IDO1 assessed as reduction in convertion of N-formylkynurenine to kynurenine incubated for 30 minsMore data for this Ligand-Target Pair
Affinity DataIC50: 3.60E+3nMAssay Description:Inhibition of human IDO1 assessed as reduction in convertion of N-formylkynurenine to kynurenine incubated for 30 minsMore data for this Ligand-Target Pair
Affinity DataIC50: 5.50E+3nMAssay Description:Inhibition of human IDO1 assessed as reduction in convertion of N-formylkynurenine to kynurenine incubated for 30 minsMore data for this Ligand-Target Pair
Affinity DataIC50: 8.10E+3nMAssay Description:Inhibition of human IDO1 assessed as reduction in convertion of N-formylkynurenine to kynurenine incubated for 30 minsMore data for this Ligand-Target Pair
Affinity DataIC50: 1.80E+4nMAssay Description:Inhibition of human IDO1 assessed as reduction in convertion of N-formylkynurenine to kynurenine incubated for 30 minsMore data for this Ligand-Target Pair
Affinity DataIC50: 2.40E+4nMAssay Description:Inhibition of human IDO1 assessed as reduction in convertion of N-formylkynurenine to kynurenine incubated for 30 minsMore data for this Ligand-Target Pair
Affinity DataIC50: 5.60E+4nMAssay Description:Inhibition of human IDO1 assessed as reduction in convertion of N-formylkynurenine to kynurenine incubated for 30 minsMore data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+5nMAssay Description:Inhibition of human IDO1 assessed as reduction in convertion of N-formylkynurenine to kynurenine incubated for 30 minsMore data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+5nMAssay Description:Inhibition of human IDO1 assessed as reduction in convertion of N-formylkynurenine to kynurenine incubated for 30 minsMore data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+5nMAssay Description:Inhibition of human IDO1 assessed as reduction in convertion of N-formylkynurenine to kynurenine incubated for 30 minsMore data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+5nMAssay Description:Inhibition of human IDO1 assessed as reduction in convertion of N-formylkynurenine to kynurenine incubated for 30 minsMore data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+5nMAssay Description:Inhibition of human IDO1 assessed as reduction in convertion of N-formylkynurenine to kynurenine incubated for 30 minsMore data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+5nMAssay Description:Inhibition of human IDO1 assessed as reduction in convertion of N-formylkynurenine to kynurenine incubated for 30 minsMore data for this Ligand-Target Pair