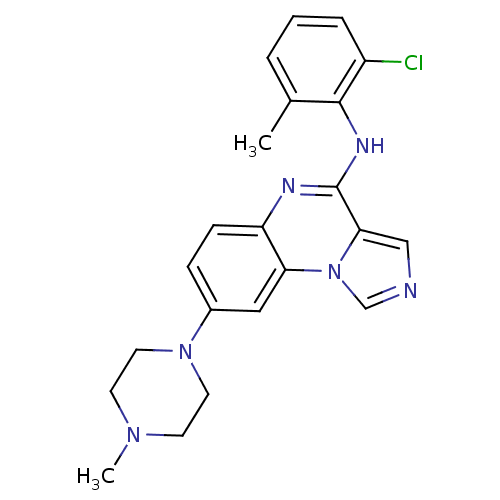
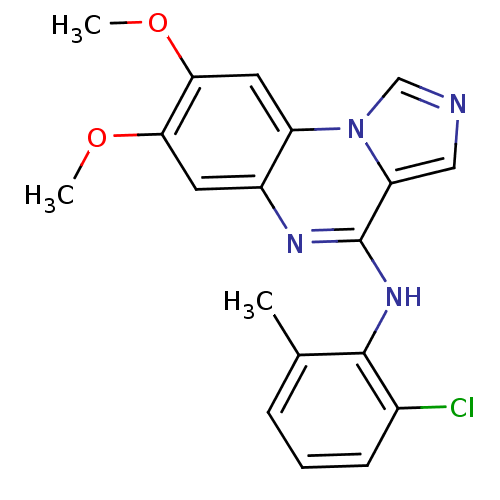
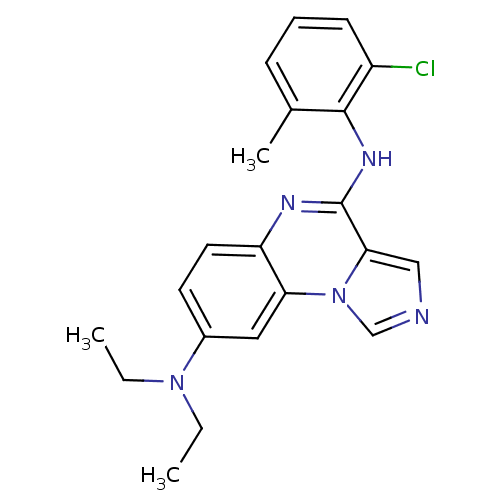
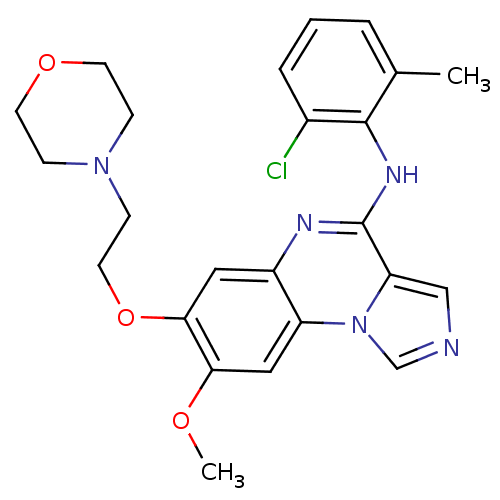
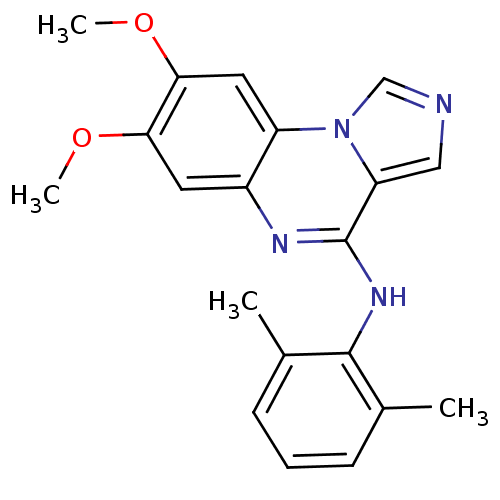
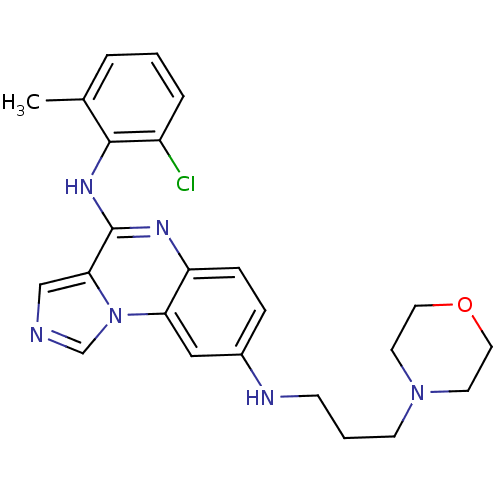
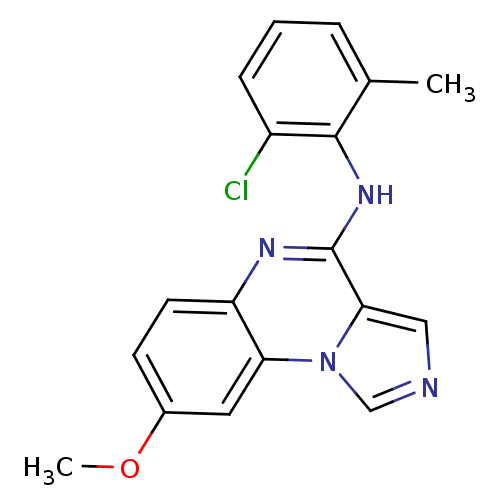
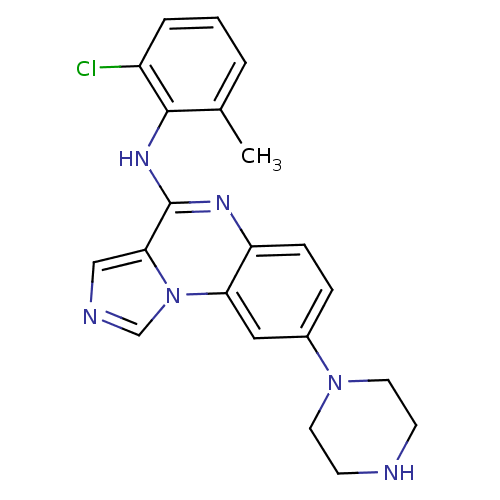
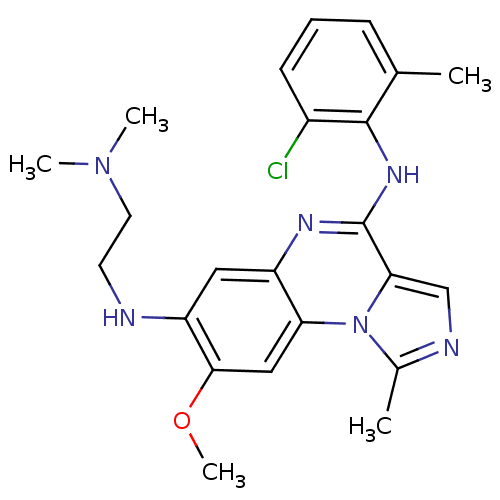
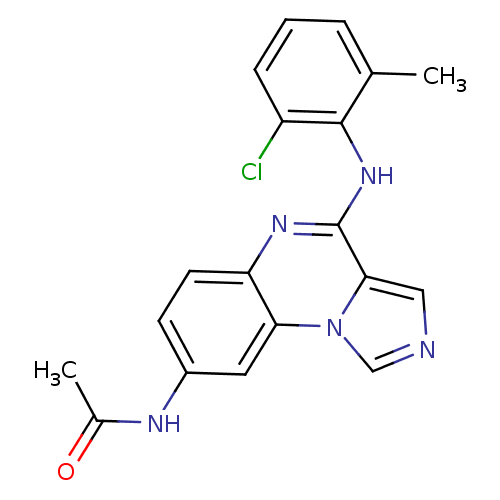
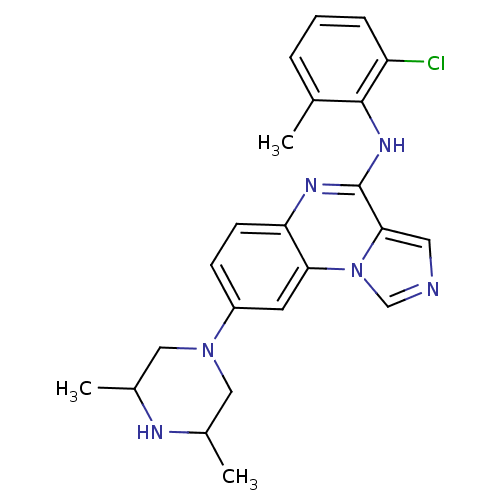
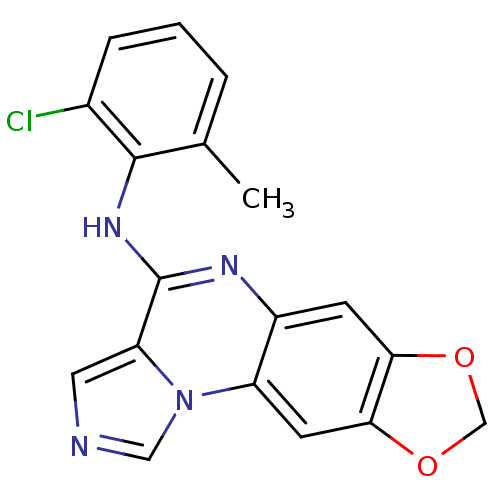
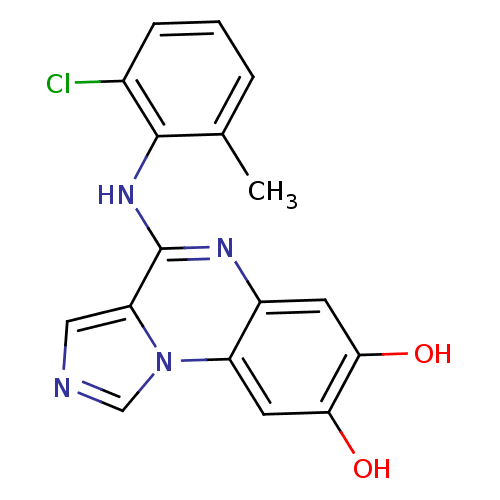
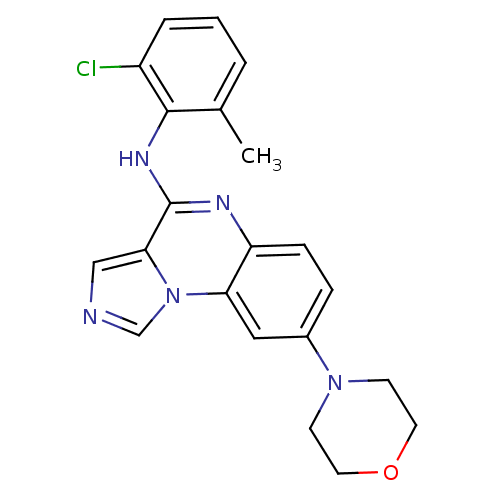
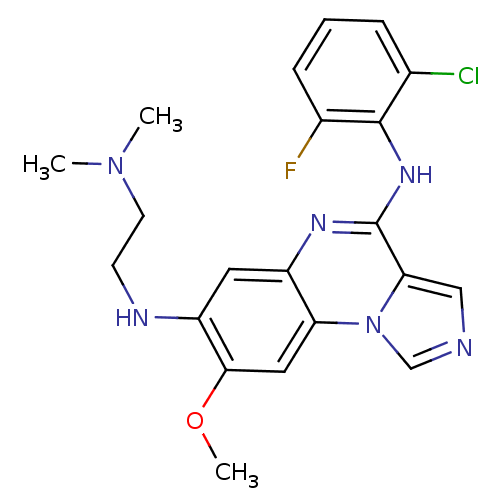
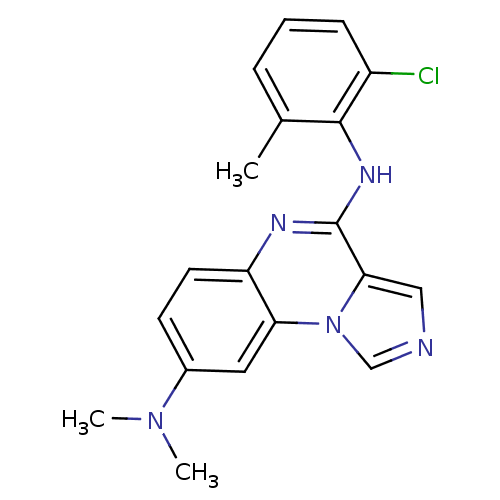
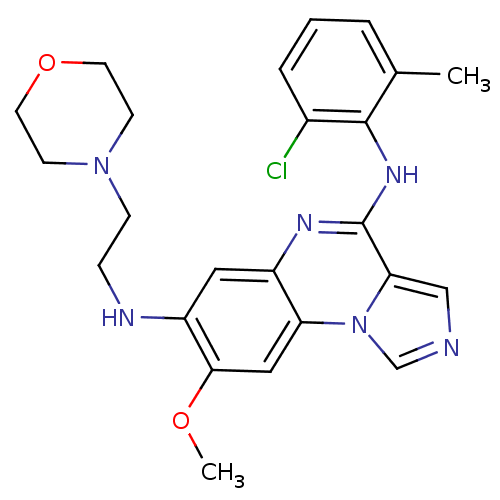
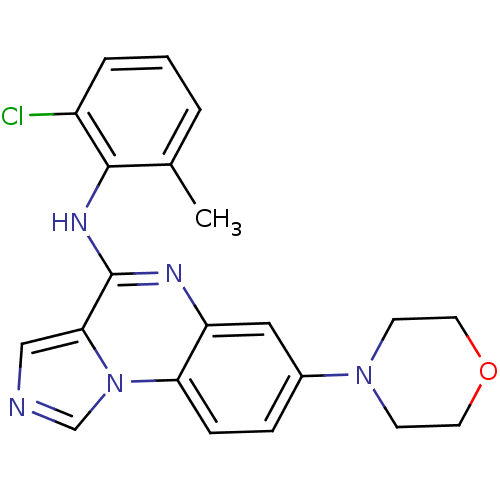
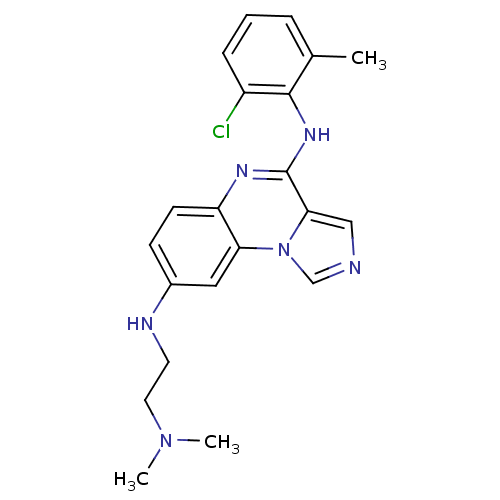
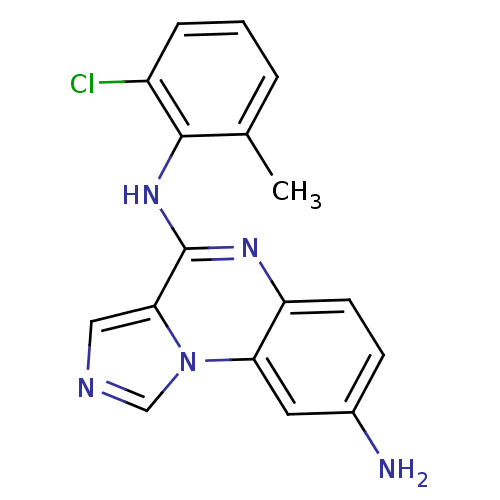
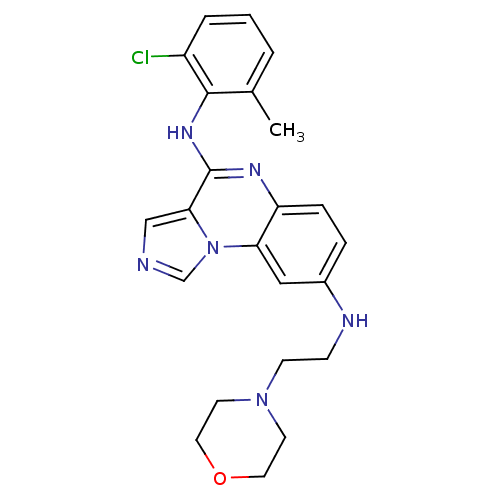
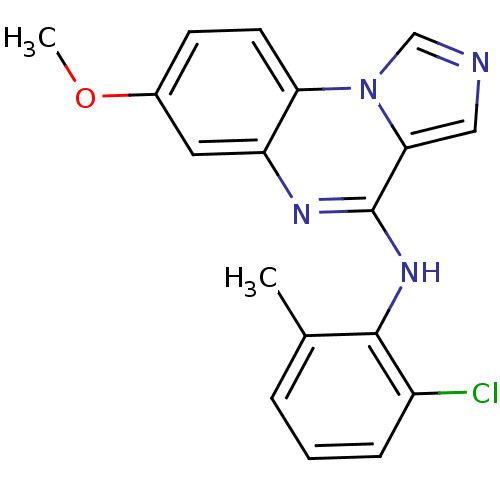
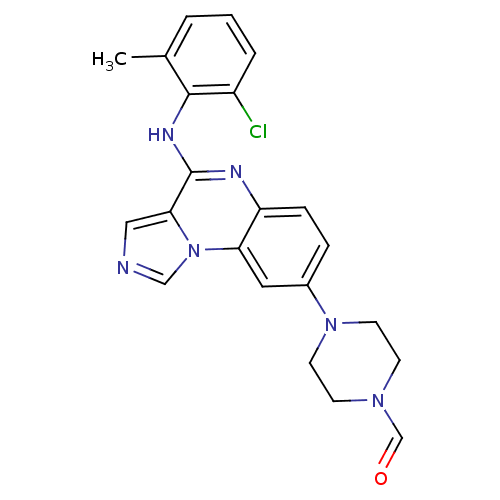
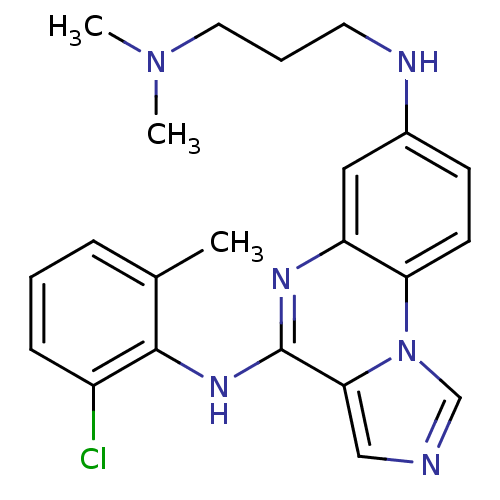
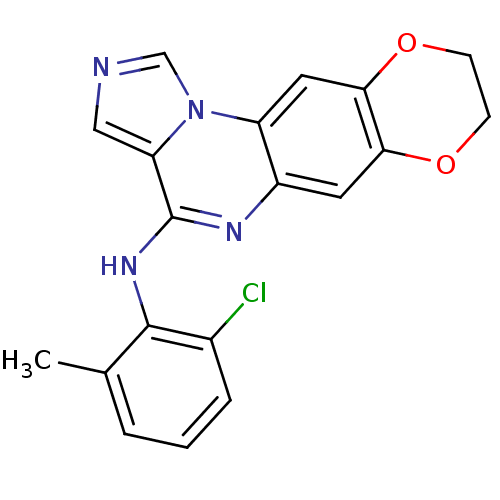
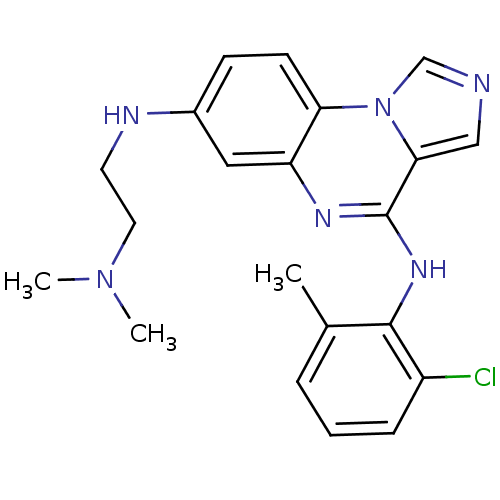
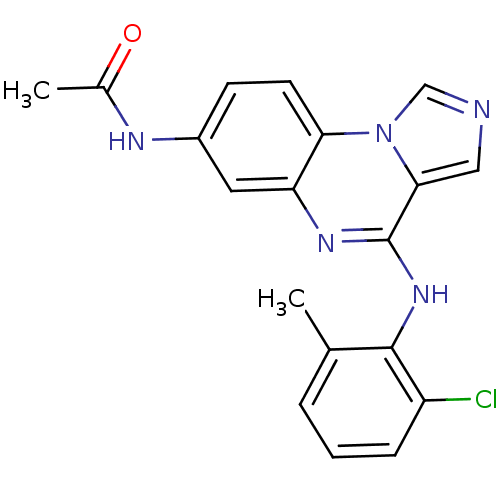
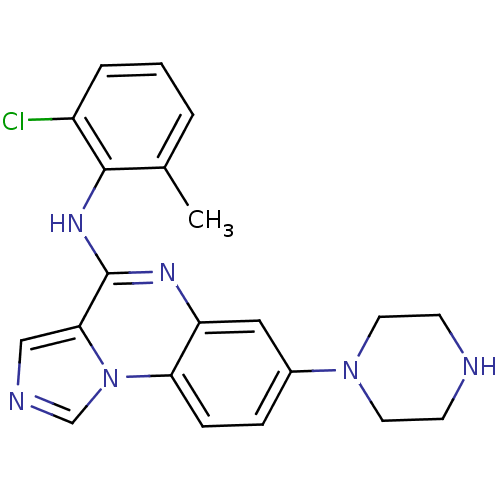
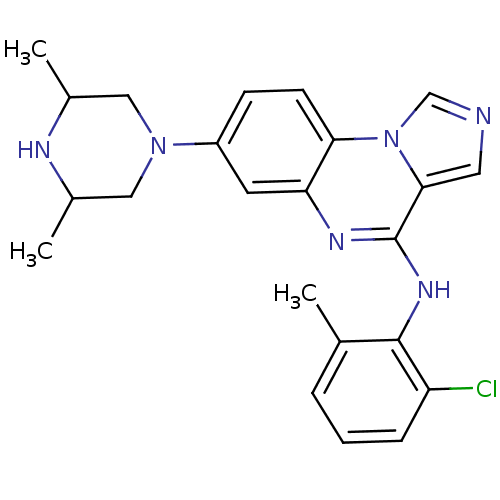
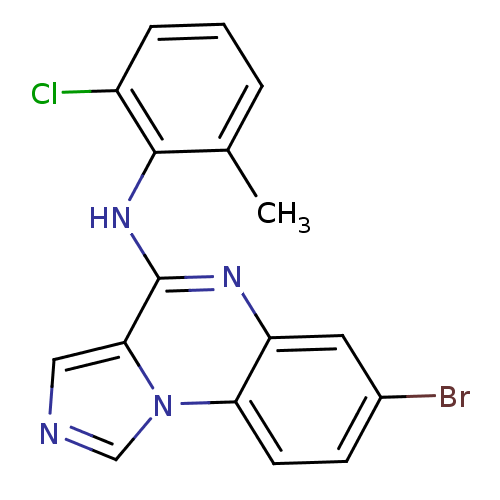
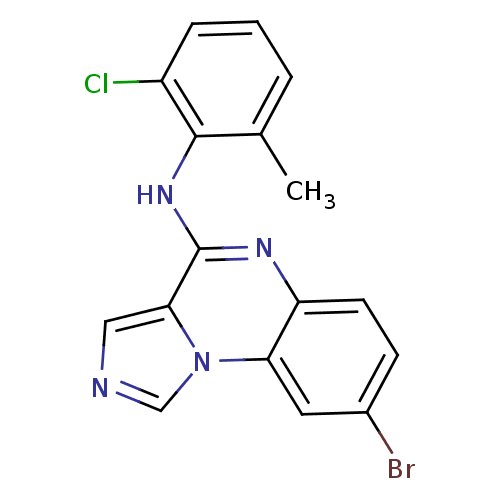
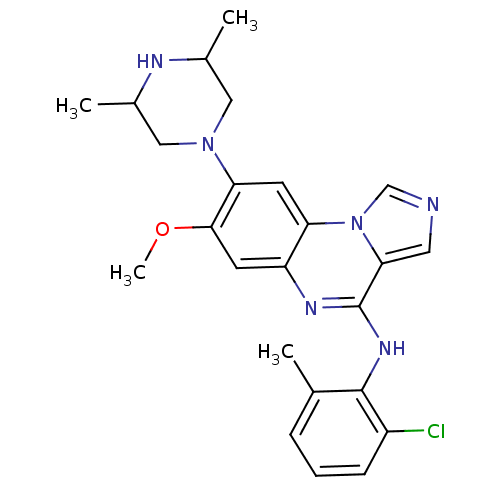
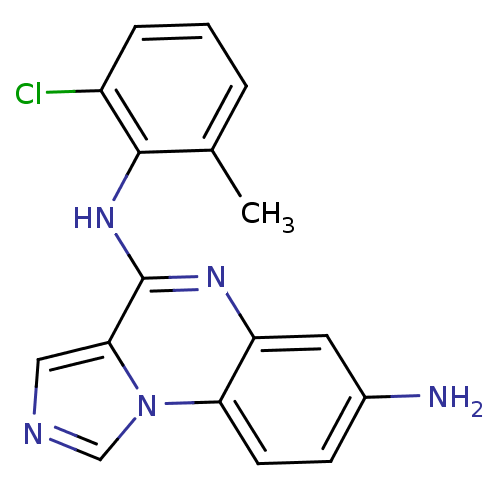
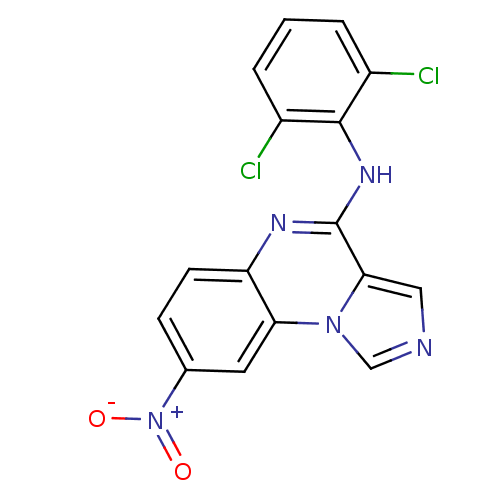
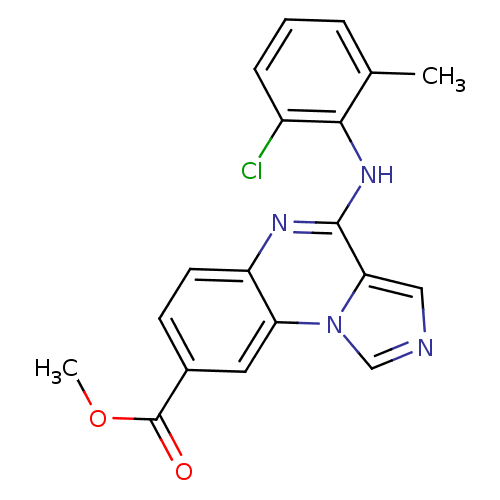
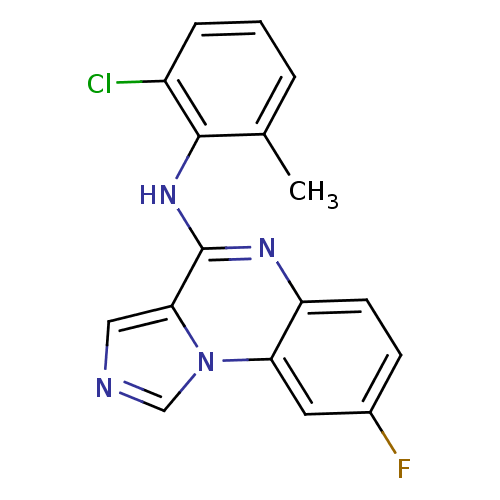
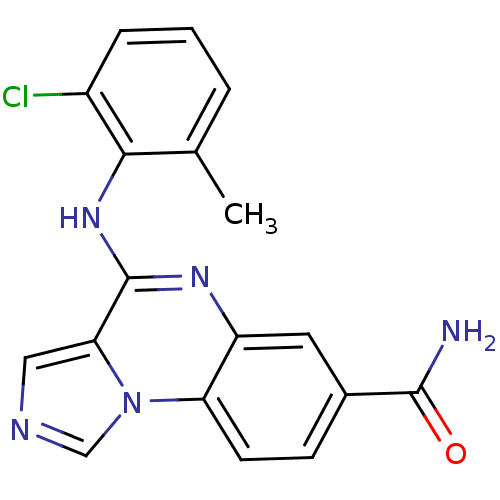
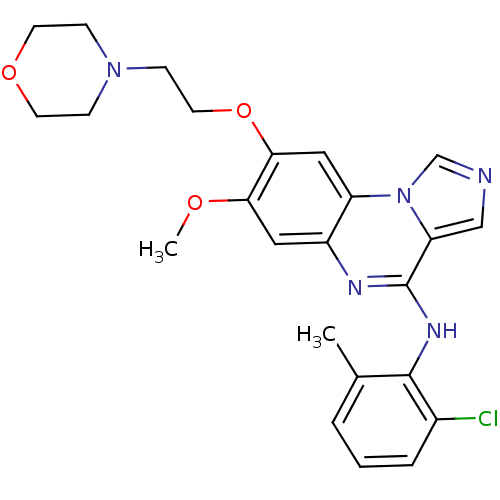
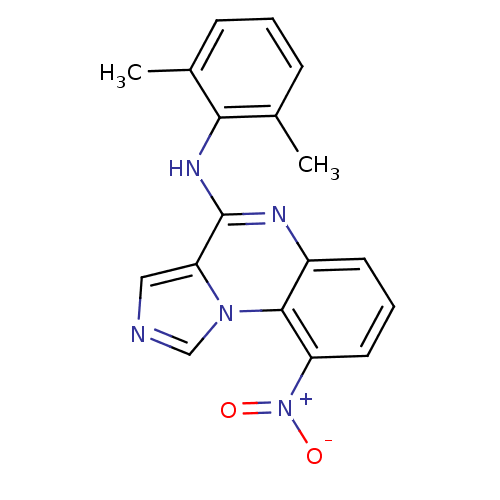
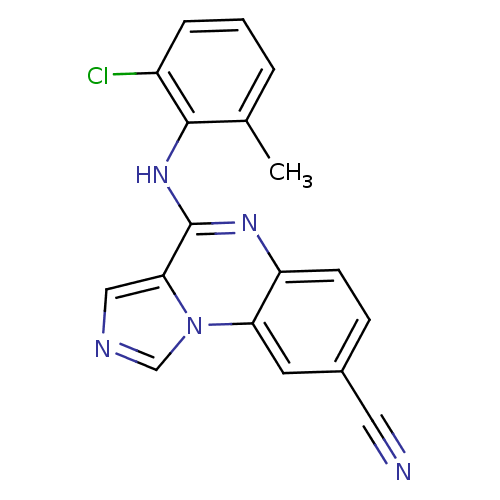
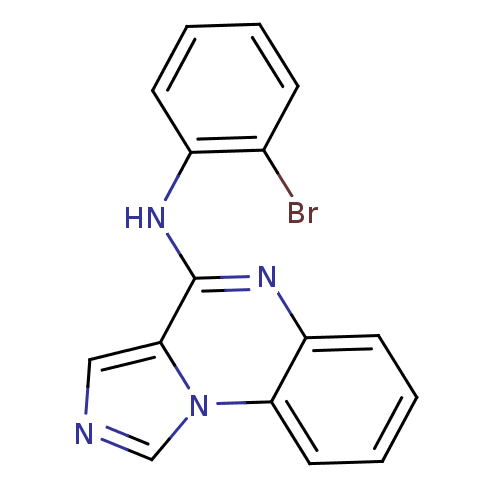
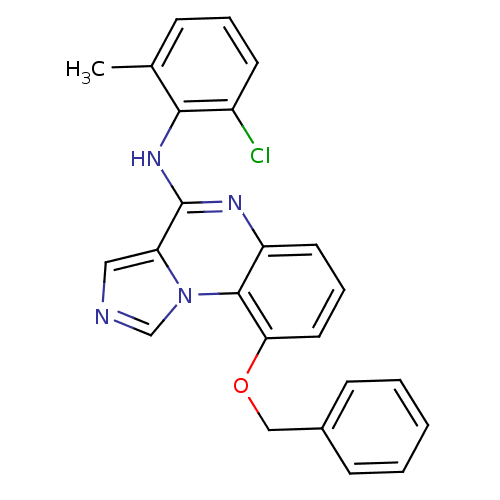
Report error Found 51 Enz. Inhib. hit(s) with all data for entry = 50012528
TargetTyrosine-protein kinase Lck(Human)
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
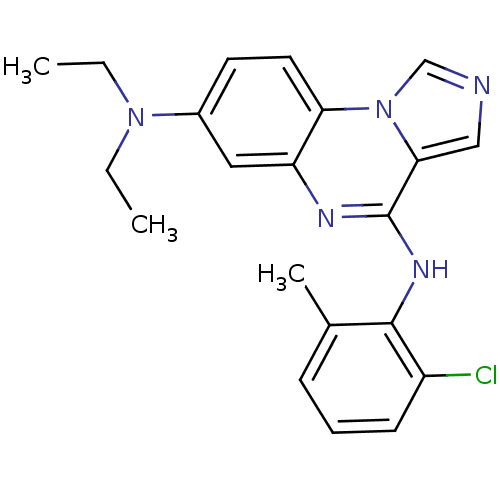
Affinity DataIC50: 1nMAssay Description:50% inhibition of the phosphorylation of an exogenous substrate by human Lck enzyme.More data for this Ligand-Target Pair
TargetTyrosine-protein kinase Lck(Human)
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
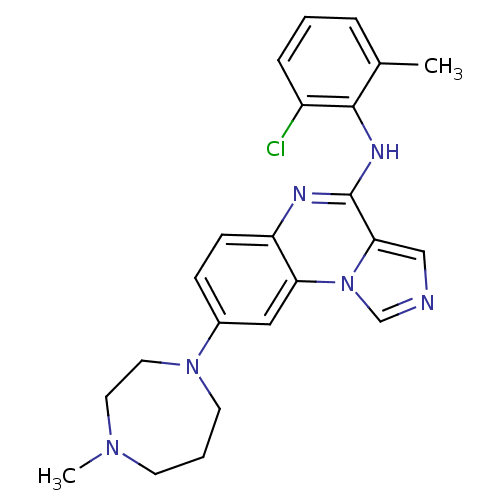
Affinity DataIC50: 1.70nMAssay Description:50% inhibition of the phosphorylation of an exogenous substrate by human Lck enzyme.More data for this Ligand-Target Pair
TargetTyrosine-protein kinase Lck(Human)
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
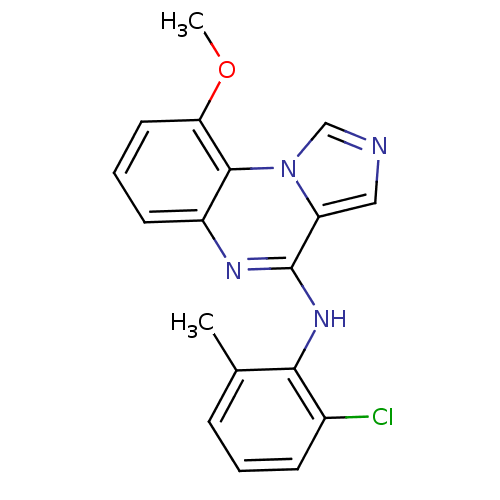
Affinity DataIC50: 2nMAssay Description:50% inhibition of the phosphorylation of an exogenous substrate by human Lck enzyme.More data for this Ligand-Target Pair
TargetTyrosine-protein kinase Lck(Human)
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
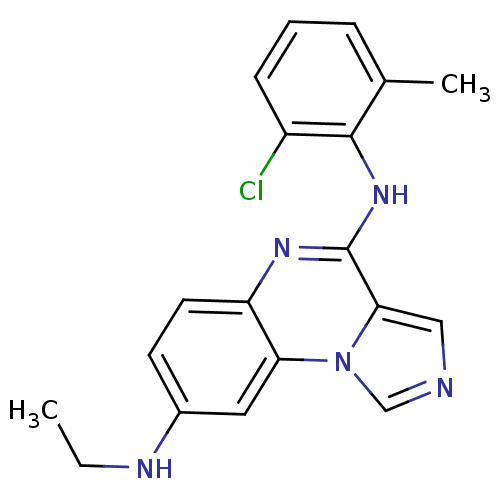
Affinity DataIC50: 2nMAssay Description:50% inhibition of the phosphorylation of an exogenous substrate by human Lck enzyme.More data for this Ligand-Target Pair
TargetTyrosine-protein kinase Lck(Human)
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 2nMAssay Description:50% inhibition of the phosphorylation of an exogenous substrate by human Lck enzyme.More data for this Ligand-Target Pair
TargetTyrosine-protein kinase Lck(Human)
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 2.40nMAssay Description:50% inhibition of the phosphorylation of an exogenous substrate by human Lck enzyme.More data for this Ligand-Target Pair
TargetTyrosine-protein kinase Lck(Human)
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 2.40nMAssay Description:50% inhibition of the phosphorylation of an exogenous substrate by human Lck enzyme.More data for this Ligand-Target Pair
TargetTyrosine-protein kinase Lck(Human)
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 3nMAssay Description:50% inhibition of the phosphorylation of an exogenous substrate by human Lck enzyme.More data for this Ligand-Target Pair
TargetTyrosine-protein kinase Lck(Human)
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 3nMAssay Description:50% inhibition of the phosphorylation of an exogenous substrate by human Lck enzyme.More data for this Ligand-Target Pair
TargetTyrosine-protein kinase Lck(Human)
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 3nMAssay Description:50% inhibition of the phosphorylation of an exogenous substrate by human Lck enzyme.More data for this Ligand-Target Pair
TargetTyrosine-protein kinase Lck(Human)
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 3nMAssay Description:50% inhibition of the phosphorylation of an exogenous substrate by human Lck enzyme.More data for this Ligand-Target Pair
TargetTyrosine-protein kinase Lck(Human)
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 3nMAssay Description:50% inhibition of the phosphorylation of an exogenous substrate by human Lck enzyme.More data for this Ligand-Target Pair
TargetTyrosine-protein kinase Lck(Human)
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 3nMAssay Description:50% inhibition of the phosphorylation of an exogenous substrate by human Lck enzyme.More data for this Ligand-Target Pair
TargetTyrosine-protein kinase Lck(Human)
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 4nMAssay Description:50% inhibition of the phosphorylation of an exogenous substrate by human Lck enzyme.More data for this Ligand-Target Pair
TargetTyrosine-protein kinase Lck(Human)
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 4nMAssay Description:50% inhibition of the phosphorylation of an exogenous substrate by human Lck enzyme.More data for this Ligand-Target Pair
TargetTyrosine-protein kinase Lck(Human)
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 4nMAssay Description:50% inhibition of the phosphorylation of an exogenous substrate by human Lck enzyme.More data for this Ligand-Target Pair
TargetTyrosine-protein kinase Lck(Human)
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 5nMAssay Description:50% inhibition of the phosphorylation of an exogenous substrate by human Lck enzyme.More data for this Ligand-Target Pair
TargetTyrosine-protein kinase Lck(Human)
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 5nMAssay Description:50% inhibition of the phosphorylation of an exogenous substrate by human Lck enzyme.More data for this Ligand-Target Pair
TargetTyrosine-protein kinase Lck(Human)
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 5.40nMAssay Description:50% inhibition of the phosphorylation of an exogenous substrate by human Lck enzyme.More data for this Ligand-Target Pair
TargetTyrosine-protein kinase Lck(Human)
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 6nMAssay Description:50% inhibition of the phosphorylation of an exogenous substrate by human Lck enzyme.More data for this Ligand-Target Pair
TargetTyrosine-protein kinase Lck(Human)
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 6nMAssay Description:50% inhibition of the phosphorylation of an exogenous substrate by human Lck enzyme.More data for this Ligand-Target Pair
TargetTyrosine-protein kinase Lck(Human)
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 7nMAssay Description:50% inhibition of the phosphorylation of an exogenous substrate by human Lck enzyme.More data for this Ligand-Target Pair
TargetTyrosine-protein kinase Lck(Human)
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 7nMAssay Description:50% inhibition of the phosphorylation of an exogenous substrate by human Lck enzyme.More data for this Ligand-Target Pair
TargetTyrosine-protein kinase Lck(Human)
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 8nMAssay Description:50% inhibition of the phosphorylation of an exogenous substrate by human Lck enzyme.More data for this Ligand-Target Pair
TargetTyrosine-protein kinase Lck(Human)
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 8.70nMAssay Description:50% inhibition of the phosphorylation of an exogenous substrate by human Lck enzyme.More data for this Ligand-Target Pair
TargetTyrosine-protein kinase Lck(Human)
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 9nMAssay Description:50% inhibition of the phosphorylation of an exogenous substrate by human Lck enzyme.More data for this Ligand-Target Pair
TargetTyrosine-protein kinase Lck(Human)
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 9nMAssay Description:50% inhibition of the phosphorylation of an exogenous substrate by human Lck enzyme.More data for this Ligand-Target Pair
TargetTyrosine-protein kinase Lck(Human)
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 9nMAssay Description:50% inhibition of the phosphorylation of an exogenous substrate by human Lck enzyme.More data for this Ligand-Target Pair
TargetTyrosine-protein kinase Lck(Human)
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 9nMAssay Description:50% inhibition of the phosphorylation of an exogenous substrate by human Lck enzyme.More data for this Ligand-Target Pair
TargetTyrosine-protein kinase Lck(Human)
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 9.40nMAssay Description:50% inhibition of the phosphorylation of an exogenous substrate by human Lck enzyme.More data for this Ligand-Target Pair
TargetTyrosine-protein kinase Lck(Human)
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 10nMAssay Description:50% inhibition of the phosphorylation of an exogenous substrate by human Lck enzyme.More data for this Ligand-Target Pair
TargetTyrosine-protein kinase Lck(Human)
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 10nMAssay Description:50% inhibition of the phosphorylation of an exogenous substrate by human Lck enzyme.More data for this Ligand-Target Pair
TargetTyrosine-protein kinase Lck(Human)
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 10nMAssay Description:50% inhibition of the phosphorylation of an exogenous substrate by human Lck enzyme.More data for this Ligand-Target Pair
TargetTyrosine-protein kinase Lck(Human)
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 11nMAssay Description:50% inhibition of the phosphorylation of an exogenous substrate by human Lck enzyme.More data for this Ligand-Target Pair
TargetTyrosine-protein kinase Lck(Human)
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 11nMAssay Description:50% inhibition of the phosphorylation of an exogenous substrate by human Lck enzyme.More data for this Ligand-Target Pair
TargetTyrosine-protein kinase Lck(Human)
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 13nMAssay Description:50% inhibition of the phosphorylation of an exogenous substrate by human Lck enzyme.More data for this Ligand-Target Pair
TargetTyrosine-protein kinase Lck(Human)
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 14nMAssay Description:50% inhibition of the phosphorylation of an exogenous substrate by human Lck enzyme.More data for this Ligand-Target Pair
TargetTyrosine-protein kinase Lck(Human)
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 15nMAssay Description:50% inhibition of the phosphorylation of an exogenous substrate by human Lck enzyme.More data for this Ligand-Target Pair
TargetTyrosine-protein kinase Lck(Human)
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 18nMAssay Description:50% inhibition of the phosphorylation of an exogenous substrate by human Lck enzyme.More data for this Ligand-Target Pair
TargetTyrosine-protein kinase Lck(Human)
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 21nMAssay Description:50% inhibition of the phosphorylation of an exogenous substrate by human Lck enzyme.More data for this Ligand-Target Pair
TargetTyrosine-protein kinase Lck(Human)
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 24nMAssay Description:50% inhibition of the phosphorylation of an exogenous substrate by human Lck enzyme.More data for this Ligand-Target Pair
TargetTyrosine-protein kinase Lck(Human)
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 26nMAssay Description:50% inhibition of the phosphorylation of an exogenous substrate by human Lck enzyme.More data for this Ligand-Target Pair
TargetTyrosine-protein kinase Lck(Human)
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 26nMAssay Description:50% inhibition of the phosphorylation of an exogenous substrate by human Lck enzyme.More data for this Ligand-Target Pair
TargetTyrosine-protein kinase Lck(Human)
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 30nMAssay Description:50% inhibition of the phosphorylation of an exogenous substrate by human Lck enzyme.More data for this Ligand-Target Pair
TargetTyrosine-protein kinase Lck(Human)
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 36nMAssay Description:50% inhibition of the phosphorylation of an exogenous substrate by human Lck enzyme.More data for this Ligand-Target Pair
TargetTyrosine-protein kinase Lck(Human)
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 70nMAssay Description:50% inhibition of the phosphorylation of an exogenous substrate by human Lck enzyme.More data for this Ligand-Target Pair
TargetTyrosine-protein kinase Lck(Human)
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 100nMAssay Description:50% inhibition of the phosphorylation of an exogenous substrate by human Lck enzyme.More data for this Ligand-Target Pair
TargetTyrosine-protein kinase Lck(Human)
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 100nMAssay Description:50% inhibition of the phosphorylation of an exogenous substrate by human Lck enzyme.More data for this Ligand-Target Pair
TargetTyrosine-protein kinase Lck(Human)
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 170nMAssay Description:50% inhibition of the phosphorylation of an exogenous substrate by human Lck enzyme.More data for this Ligand-Target Pair
TargetTyrosine-protein kinase Lck(Human)
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
Affinity DataIC50: 240nMAssay Description:50% inhibition of the phosphorylation of an exogenous substrate by human Lck enzyme.More data for this Ligand-Target Pair