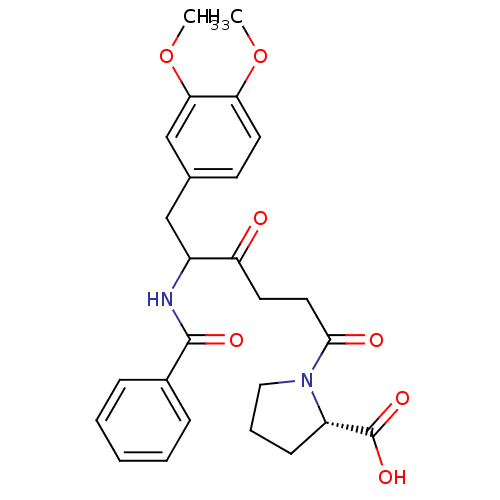
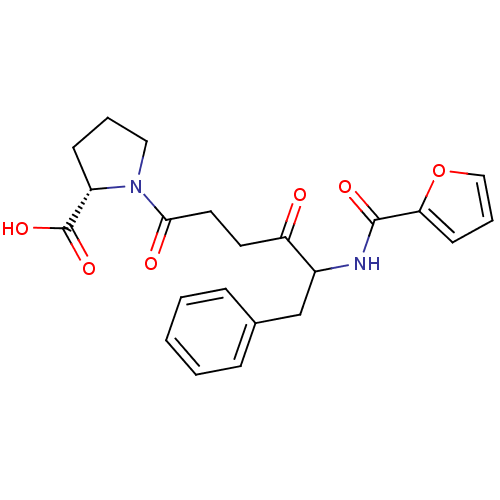
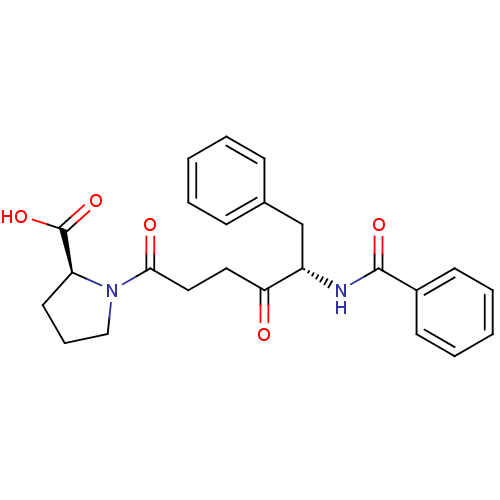
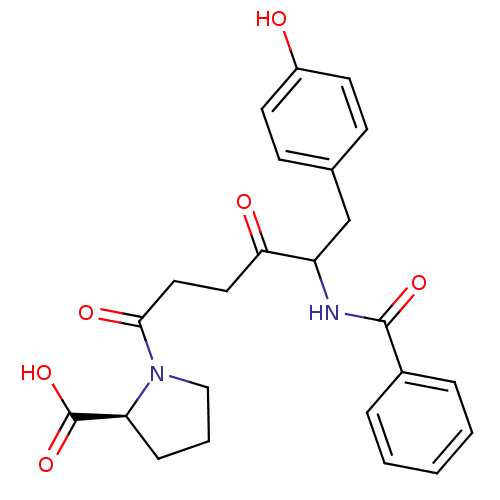
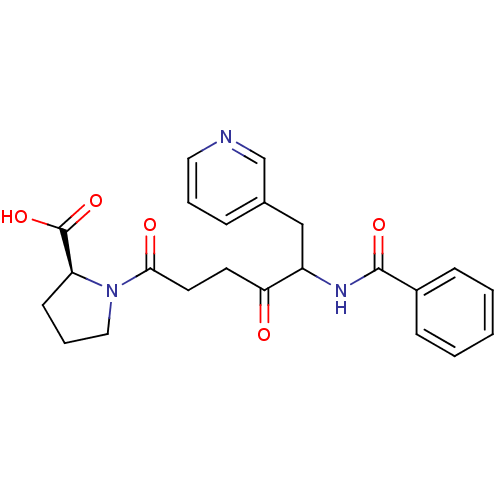
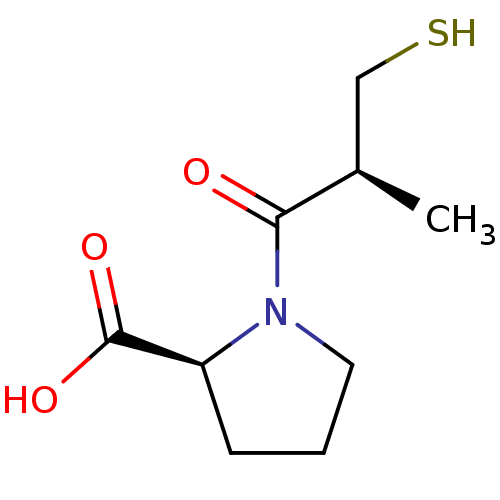
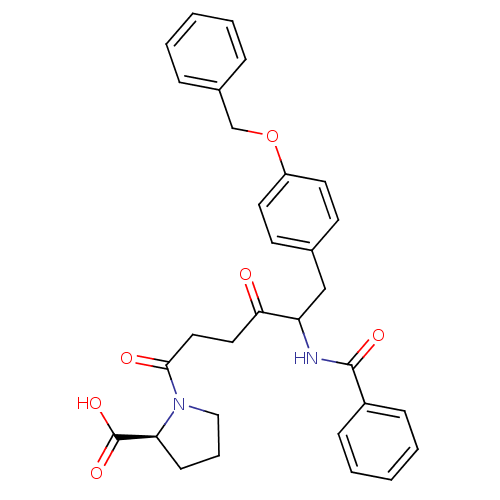
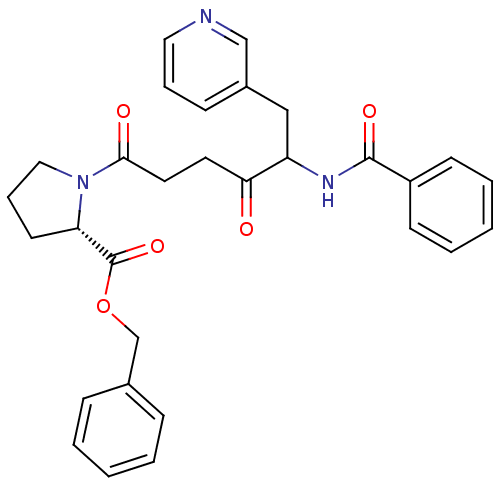
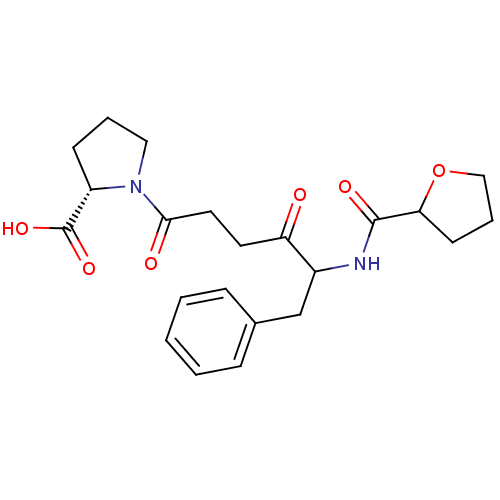
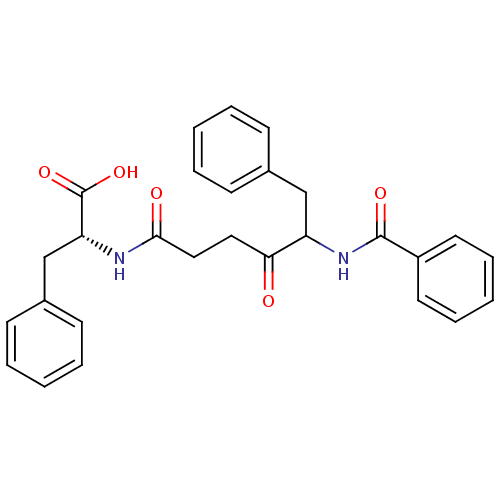
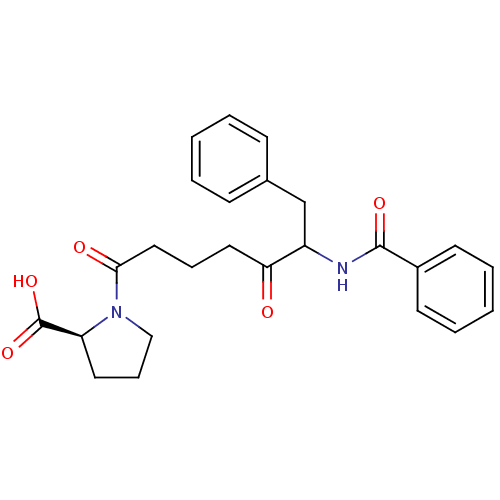
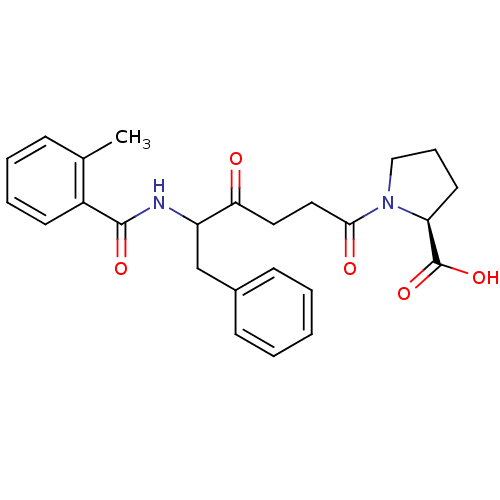
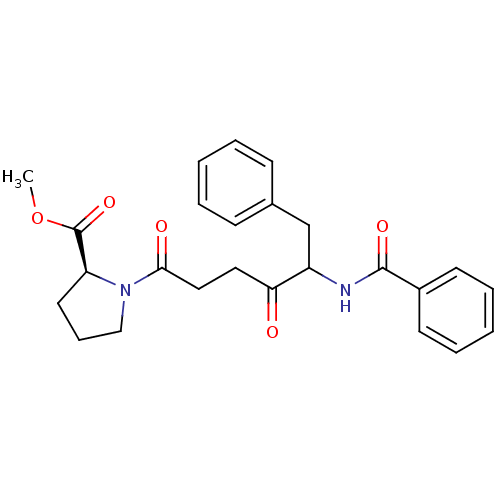
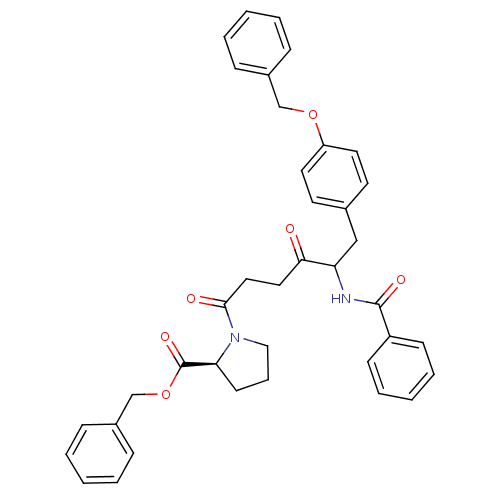
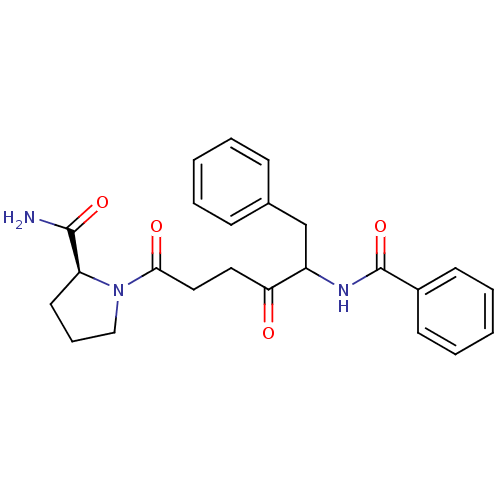
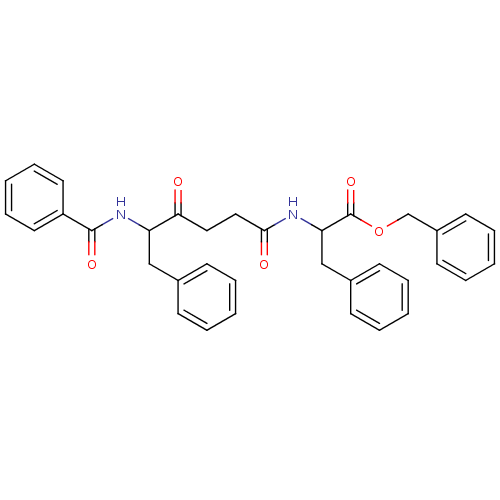
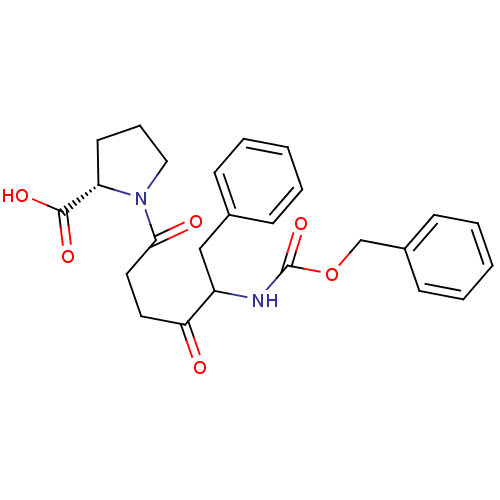
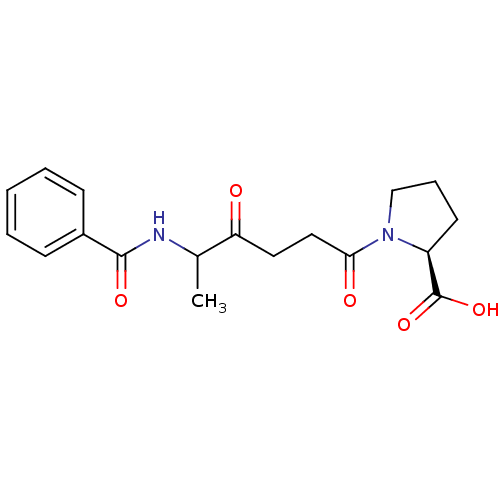
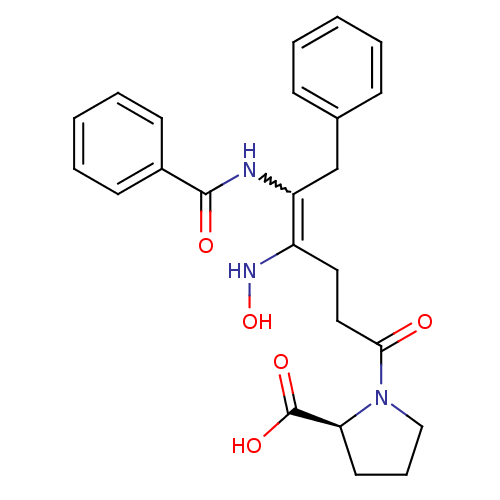
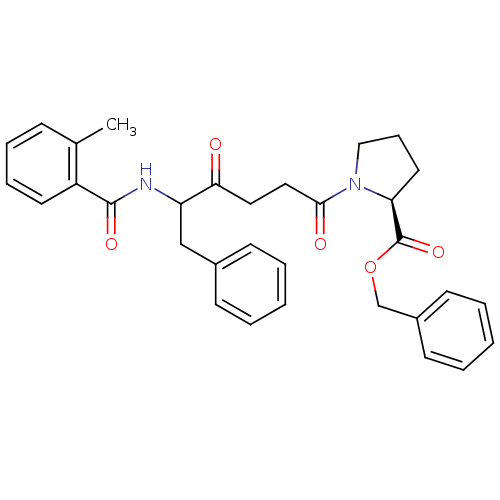
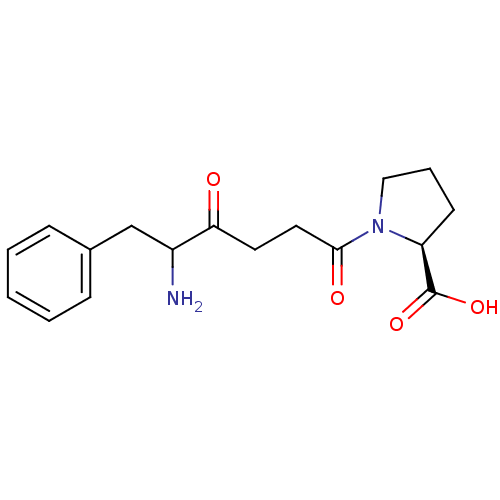
Report error Found 24 Enz. Inhib. hit(s) with all data for entry = 50035474
Affinity DataIC50: 1.40nMAssay Description:Concentration required to inhibit the activity of Angiotensin I converting enzyme by 50%More data for this Ligand-Target Pair
Affinity DataIC50: 1.40nMAssay Description:Concentration required to inhibit the activity of Angiotensin I converting enzyme by 50%More data for this Ligand-Target Pair
Affinity DataIC50: 3.20nMAssay Description:Concentration required to inhibit the activity of Angiotensin I converting enzyme by 50%More data for this Ligand-Target Pair
Affinity DataIC50: 4.70nMAssay Description:Concentration required to inhibit the activity of Angiotensin I converting enzyme by 50%More data for this Ligand-Target Pair
Affinity DataIC50: 5.70nMAssay Description:Concentration required to inhibit the activity of Angiotensin I converting enzyme by 50%More data for this Ligand-Target Pair
Affinity DataIC50: 9nMAssay Description:Concentration required to inhibit the activity of Angiotensin I converting enzyme by 50%More data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:Concentration required to inhibit the activity of Angiotensin I converting enzyme by 50%More data for this Ligand-Target Pair
Affinity DataIC50: 15nMAssay Description:Concentration required to inhibit the activity of Angiotensin I converting enzyme by 50%More data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:Concentration required to inhibit the activity of Angiotensin I converting enzyme by 50%More data for this Ligand-Target Pair
Affinity DataIC50: 28nMAssay Description:Concentration required to inhibit the activity of Angiotensin I converting enzyme by 50%More data for this Ligand-Target Pair
Affinity DataIC50: 42nMAssay Description:Concentration required to inhibit the activity of Angiotensin I converting enzyme by 50%More data for this Ligand-Target Pair
Affinity DataIC50: 46nMAssay Description:Concentration required to inhibit the activity of Angiotensin I converting enzyme by 50%More data for this Ligand-Target Pair
Affinity DataIC50: 82nMAssay Description:Concentration required to inhibit the activity of Angiotensin I converting enzyme by 50%More data for this Ligand-Target Pair
Affinity DataIC50: 86nMAssay Description:Concentration required to inhibit the activity of Angiotensin I converting enzyme by 50%More data for this Ligand-Target Pair
Affinity DataIC50: 150nMAssay Description:Concentration required to inhibit the activity of Angiotensin I converting enzyme by 50%More data for this Ligand-Target Pair
Affinity DataIC50: 180nMAssay Description:Concentration required to inhibit the activity of Angiotensin I converting enzyme by 50%More data for this Ligand-Target Pair
Affinity DataIC50: 240nMAssay Description:Concentration required to inhibit the activity of Angiotensin I converting enzyme by 50%More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:Concentration required to inhibit the activity of Angiotensin I converting enzyme by 50%More data for this Ligand-Target Pair
Affinity DataIC50: 330nMAssay Description:Concentration required to inhibit the activity of Angiotensin I converting enzyme by 50%More data for this Ligand-Target Pair
Affinity DataIC50: 540nMAssay Description:Concentration required to inhibit the activity of Angiotensin I converting enzyme by 50%More data for this Ligand-Target Pair
Affinity DataIC50: 1.20E+3nMAssay Description:Concentration required to inhibit the activity of Angiotensin I converting enzyme by 50%More data for this Ligand-Target Pair
Affinity DataIC50: 1.60E+3nMAssay Description:Concentration required to inhibit the activity of Angiotensin I converting enzyme by 50%More data for this Ligand-Target Pair
Affinity DataIC50: 8.60E+3nMAssay Description:Concentration required to inhibit the activity of Angiotensin I converting enzyme by 50%More data for this Ligand-Target Pair
Affinity DataIC50: 2.50E+4nMAssay Description:Concentration required to inhibit the activity of Angiotensin I converting enzyme by 50%More data for this Ligand-Target Pair