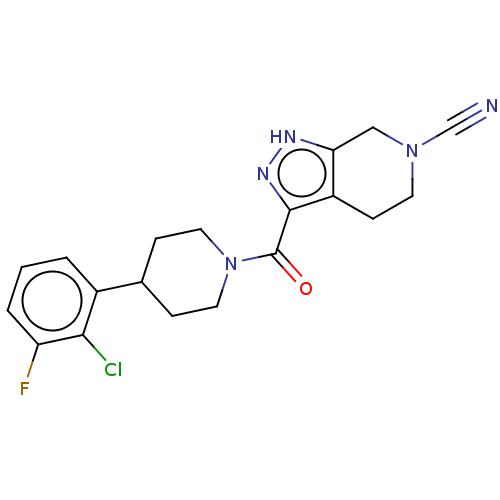
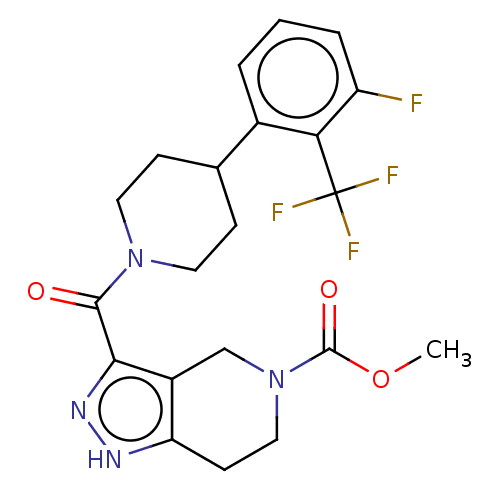
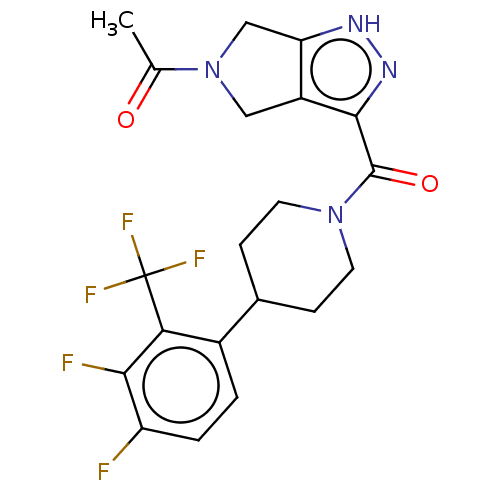
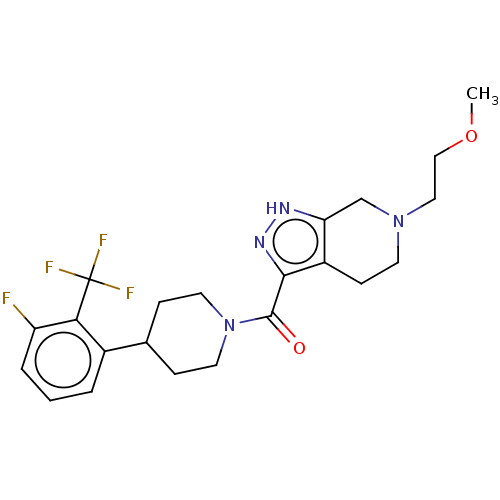
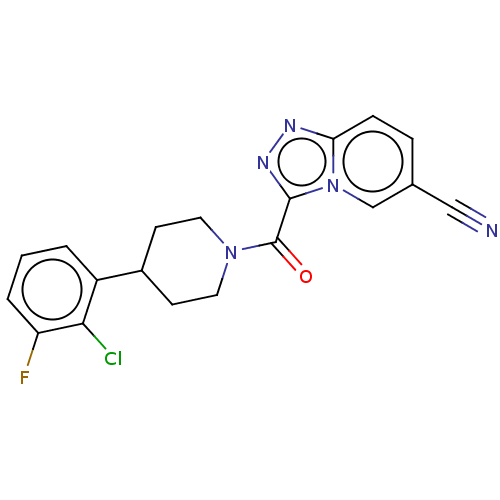
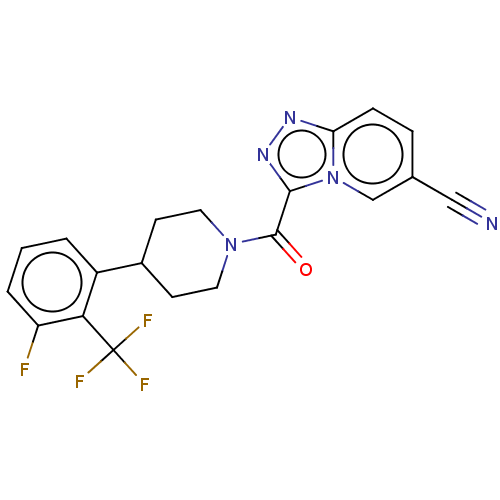
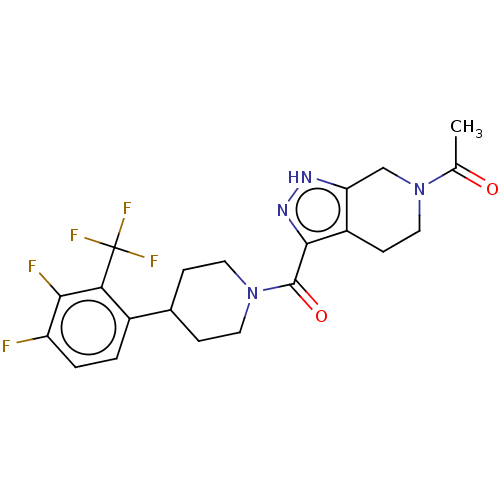
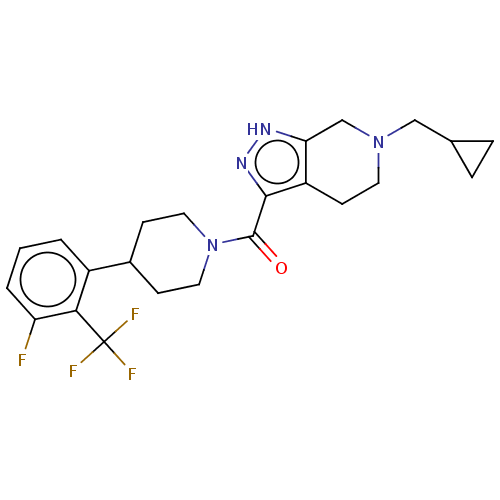
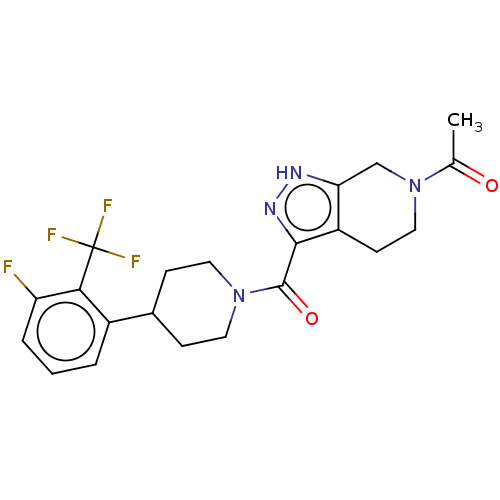
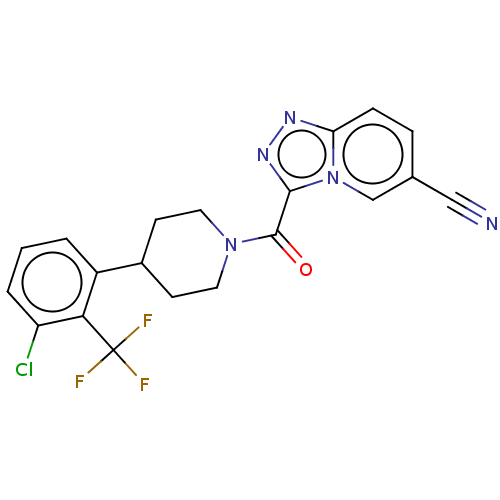
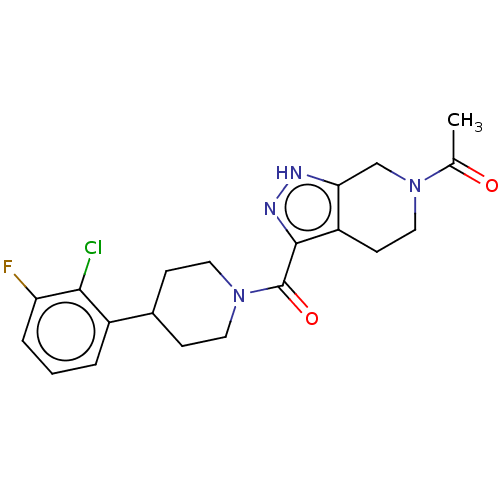
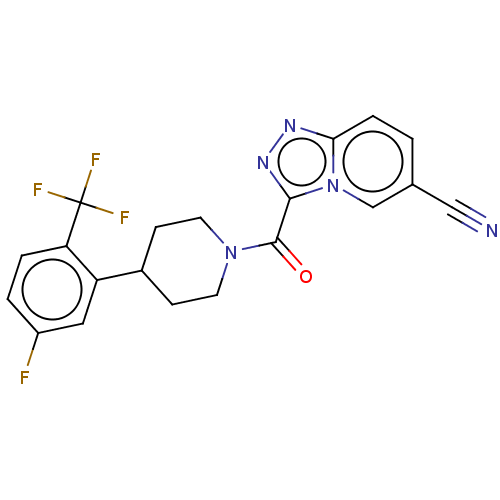
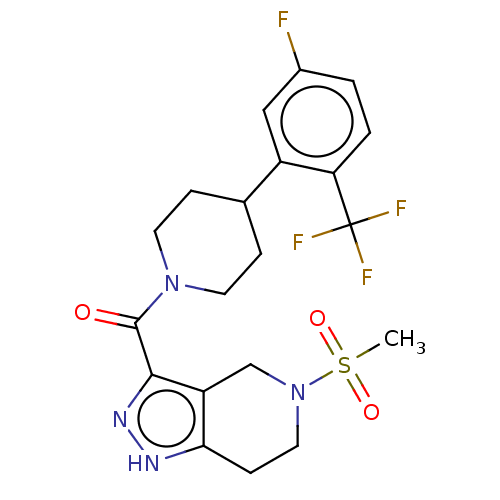
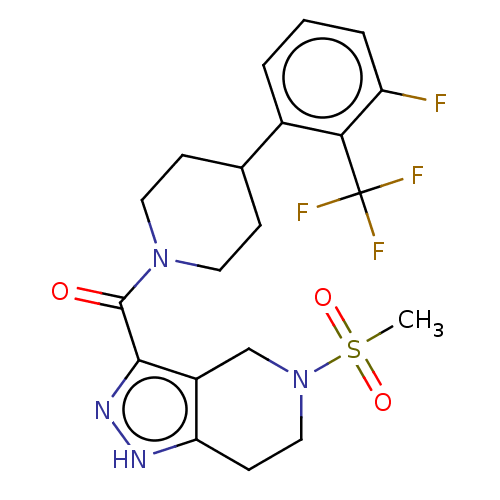
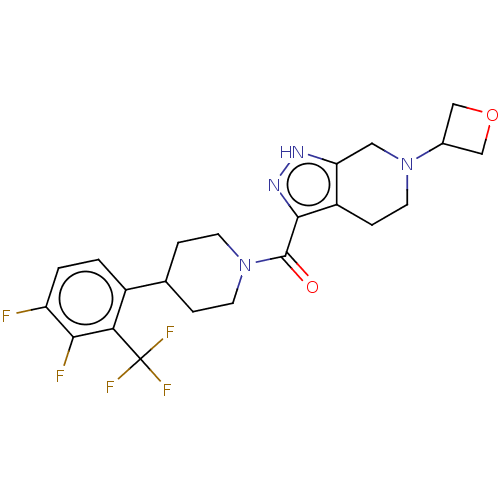
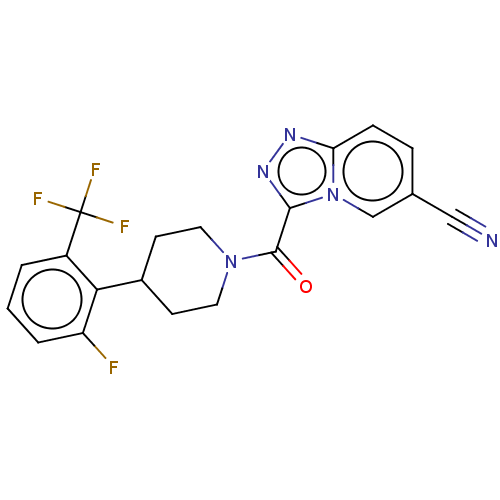
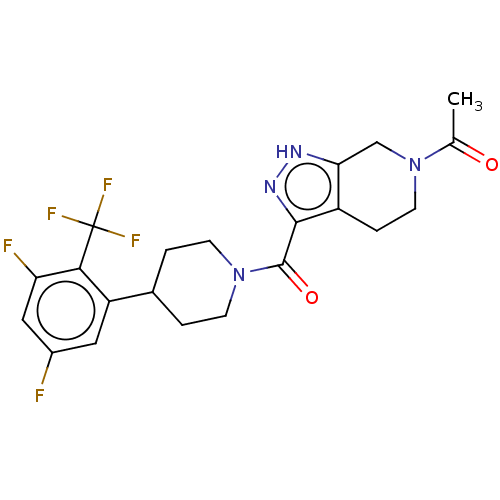
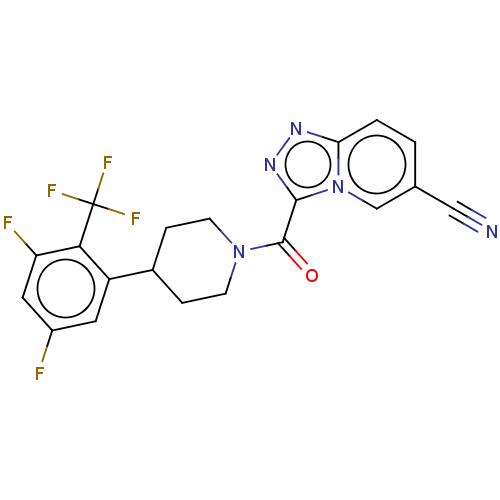
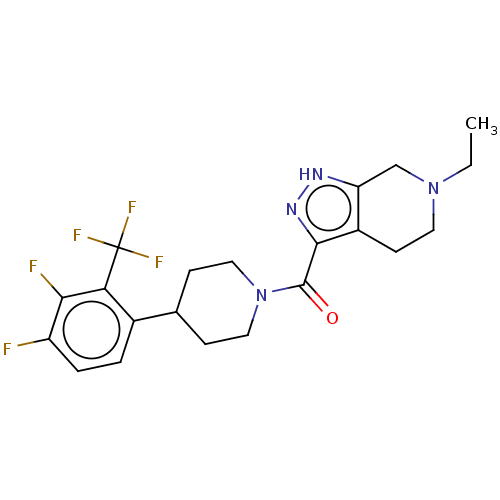
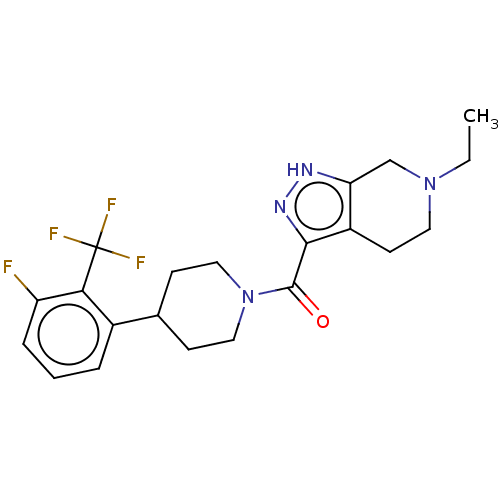
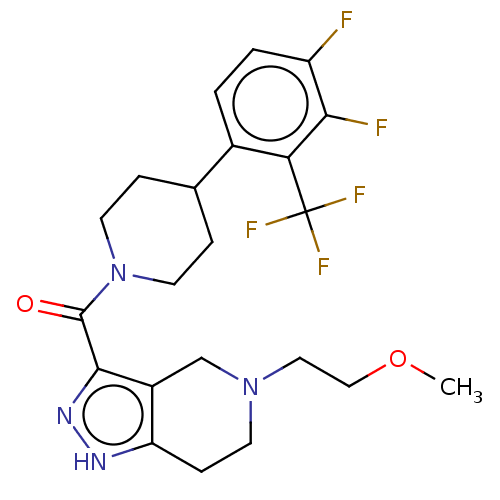
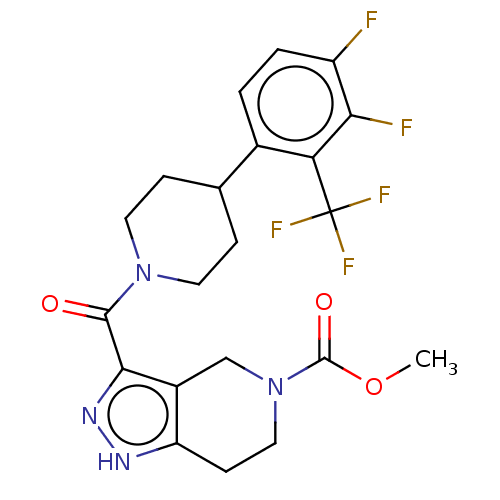
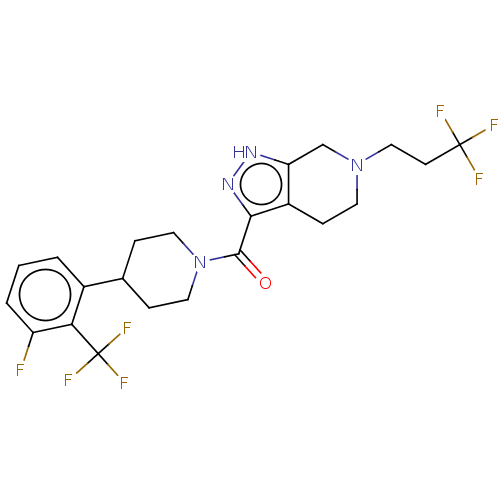
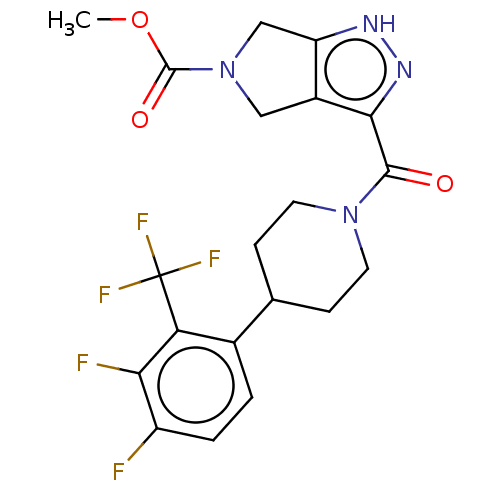
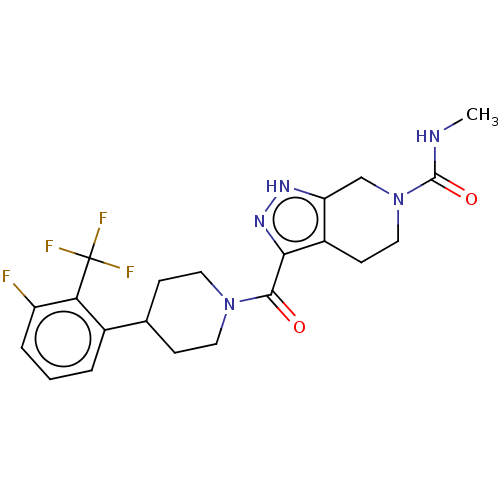
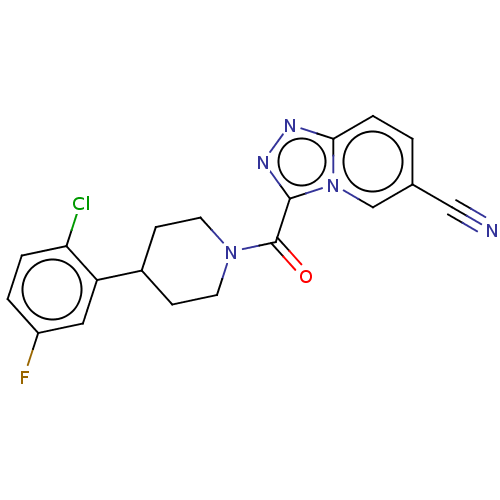
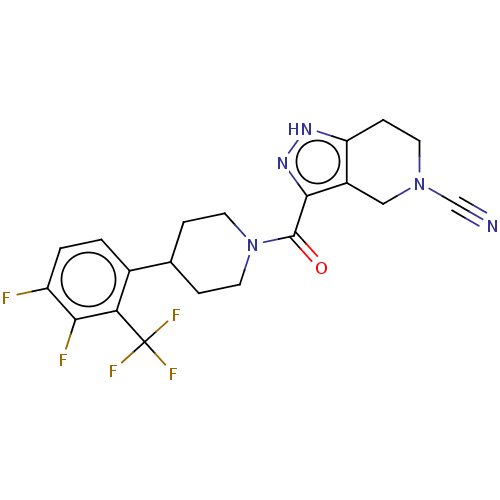
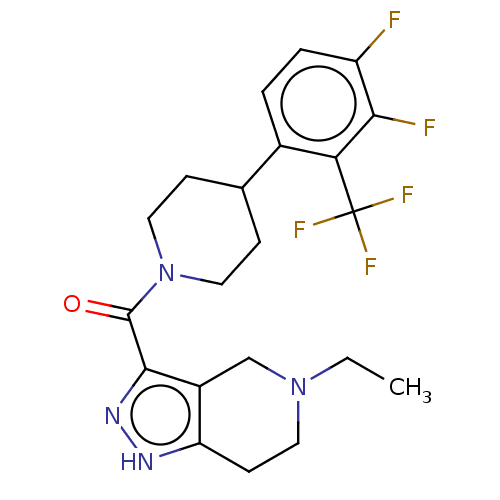
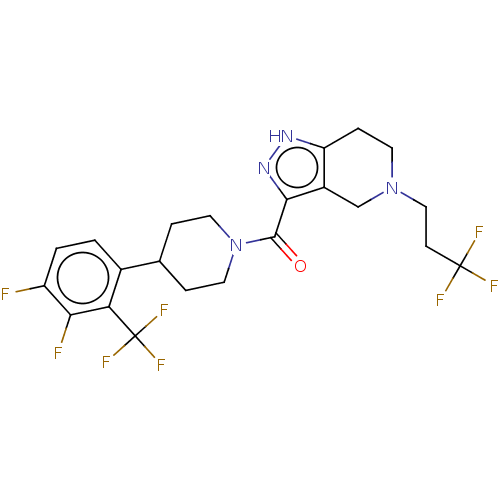
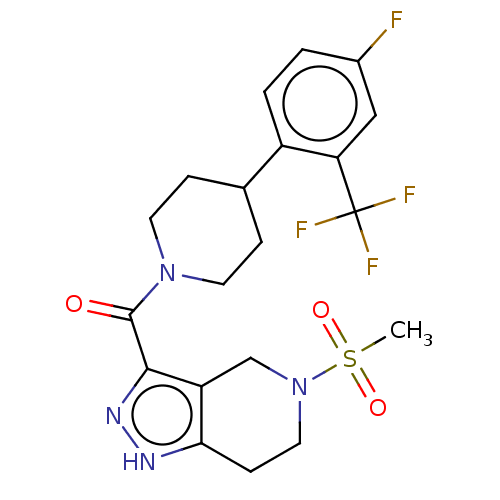
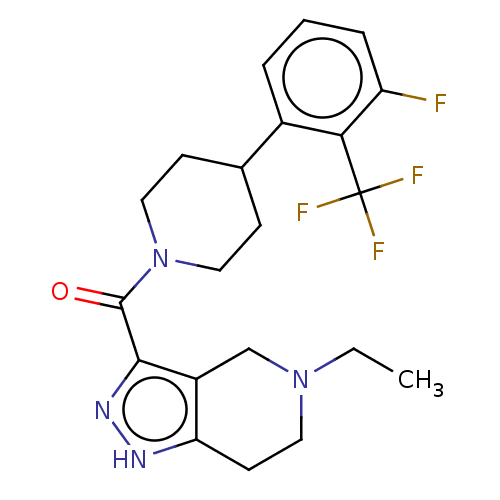
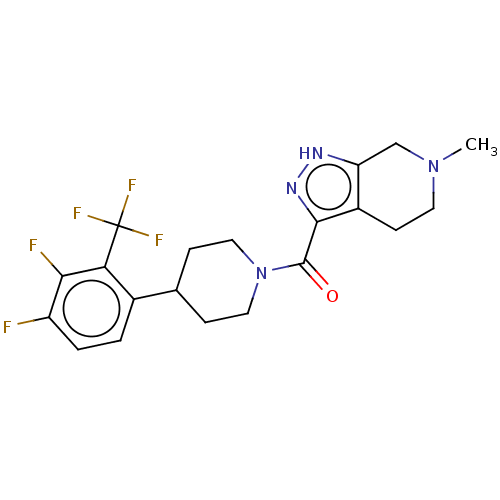
Report error Found 247 Enz. Inhib. hit(s) with all data for entry = 540
Affinity DataIC50: 0.169nMpH: 7.4 T: 2°CAssay Description:Untagged human RBP4 purified from urine of tubular proteinuria patients was purchased from Fitzgerald Industries International. It was biotinylated u...More data for this Ligand-Target Pair
Affinity DataIC50: 1.37nMpH: 7.5Assay Description:Binding of a desired RBP4 antagonist displaces retinol and induces hindrance for RBP4-TTR interaction resulting in the decreased FRET signal (FIG. 7)...More data for this Ligand-Target Pair
Affinity DataIC50: 1.37nMpH: 7.5Assay Description:Binding of a desired RBP4 antagonist displaces retinol and induces hindrance for RBP4-TTR interaction resulting in the decreased FRET signal (FIG. 7)...More data for this Ligand-Target Pair
Affinity DataIC50: 1.37nMpH: 7.5Assay Description:Binding of a desired RBP4 antagonist displaces retinol and induces hindrance for RBP4-TTR interaction resulting in the decreased FRET signal (FIG. 7)...More data for this Ligand-Target Pair
Affinity DataIC50: 1.62nMpH: 7.4 T: 2°CAssay Description:Untagged human RBP4 purified from urine of tubular proteinuria patients was purchased from Fitzgerald Industries International. It was biotinylated u...More data for this Ligand-Target Pair
Affinity DataIC50: 2.88nMpH: 7.5Assay Description:Binding of a desired RBP4 antagonist displaces retinol and induces hindrance for RBP4-TTR interaction resulting in the decreased FRET signal (FIG. 7)...More data for this Ligand-Target Pair
Affinity DataIC50: 3.10nMpH: 7.5Assay Description:Binding of a desired RBP4 antagonist displaces retinol and induces hindrance for RBP4-TTR interaction resulting in the decreased FRET signal (FIG. 7)...More data for this Ligand-Target Pair
Affinity DataIC50: 3.45nMpH: 7.5Assay Description:Binding of a desired RBP4 antagonist displaces retinol and induces hindrance for RBP4-TTR interaction resulting in the decreased FRET signal (FIG. 7)...More data for this Ligand-Target Pair
Affinity DataIC50: 4.13nMpH: 7.5Assay Description:Binding of a desired RBP4 antagonist displaces retinol and induces hindrance for RBP4-TTR interaction resulting in the decreased FRET signal (FIG. 7)...More data for this Ligand-Target Pair
Affinity DataIC50: 4.33nMpH: 7.5Assay Description:Binding of a desired RBP4 antagonist displaces retinol and induces hindrance for RBP4-TTR interaction resulting in the decreased FRET signal (FIG. 7)...More data for this Ligand-Target Pair
Affinity DataIC50: 4.33nMpH: 7.5Assay Description:Binding of a desired RBP4 antagonist displaces retinol and induces hindrance for RBP4-TTR interaction resulting in the decreased FRET signal (FIG. 7)...More data for this Ligand-Target Pair
Affinity DataIC50: 4.33nMpH: 7.5Assay Description:Binding of a desired RBP4 antagonist displaces retinol and induces hindrance for RBP4-TTR interaction resulting in the decreased FRET signal (FIG. 7)...More data for this Ligand-Target Pair
Affinity DataIC50: 4.35nMpH: 7.5Assay Description:Binding of a desired RBP4 antagonist displaces retinol and induces hindrance for RBP4-TTR interaction resulting in the decreased FRET signal (FIG. 7)...More data for this Ligand-Target Pair
Affinity DataIC50: 4.46nMpH: 7.5Assay Description:Binding of a desired RBP4 antagonist displaces retinol and induces hindrance for RBP4-TTR interaction resulting in the decreased FRET signal (FIG. 7)...More data for this Ligand-Target Pair
Affinity DataIC50: 4.82nMpH: 7.5Assay Description:Binding of a desired RBP4 antagonist displaces retinol and induces hindrance for RBP4-TTR interaction resulting in the decreased FRET signal (FIG. 7)...More data for this Ligand-Target Pair
Affinity DataIC50: 4.84nMpH: 7.5Assay Description:Binding of a desired RBP4 antagonist displaces retinol and induces hindrance for RBP4-TTR interaction resulting in the decreased FRET signal (FIG. 7)...More data for this Ligand-Target Pair
Affinity DataIC50: 4.85nMpH: 7.4 T: 2°CAssay Description:Untagged human RBP4 purified from urine of tubular proteinuria patients was purchased from Fitzgerald Industries International. It was biotinylated u...More data for this Ligand-Target Pair
Affinity DataIC50: 4.88nMpH: 7.5Assay Description:Binding of a desired RBP4 antagonist displaces retinol and induces hindrance for RBP4-TTR interaction resulting in the decreased FRET signal (FIG. 7)...More data for this Ligand-Target Pair
Affinity DataIC50: 5.12nMpH: 7.5Assay Description:Binding of a desired RBP4 antagonist displaces retinol and induces hindrance for RBP4-TTR interaction resulting in the decreased FRET signal (FIG. 7)...More data for this Ligand-Target Pair
Affinity DataIC50: 5.24nMpH: 7.5Assay Description:Binding of a desired RBP4 antagonist displaces retinol and induces hindrance for RBP4-TTR interaction resulting in the decreased FRET signal (FIG. 7)...More data for this Ligand-Target Pair
Affinity DataIC50: 5.36nMpH: 7.5Assay Description:Binding of a desired RBP4 antagonist displaces retinol and induces hindrance for RBP4-TTR interaction resulting in the decreased FRET signal (FIG. 7)...More data for this Ligand-Target Pair
Affinity DataIC50: 5.36nMpH: 7.5Assay Description:Binding of a desired RBP4 antagonist displaces retinol and induces hindrance for RBP4-TTR interaction resulting in the decreased FRET signal (FIG. 7)...More data for this Ligand-Target Pair
Affinity DataIC50: 5.36nMpH: 7.5Assay Description:Binding of a desired RBP4 antagonist displaces retinol and induces hindrance for RBP4-TTR interaction resulting in the decreased FRET signal (FIG. 7)...More data for this Ligand-Target Pair
Affinity DataIC50: 5.36nMpH: 7.5Assay Description:Binding of a desired RBP4 antagonist displaces retinol and induces hindrance for RBP4-TTR interaction resulting in the decreased FRET signal (FIG. 7)...More data for this Ligand-Target Pair
Affinity DataIC50: 5.48nMpH: 7.5Assay Description:Binding of a desired RBP4 antagonist displaces retinol and induces hindrance for RBP4-TTR interaction resulting in the decreased FRET signal (FIG. 7)...More data for this Ligand-Target Pair
Affinity DataIC50: 5.74nMpH: 7.5Assay Description:Binding of a desired RBP4 antagonist displaces retinol and induces hindrance for RBP4-TTR interaction resulting in the decreased FRET signal (FIG. 7)...More data for this Ligand-Target Pair
Affinity DataIC50: 5.84nMpH: 7.5Assay Description:Binding of a desired RBP4 antagonist displaces retinol and induces hindrance for RBP4-TTR interaction resulting in the decreased FRET signal (FIG. 7)...More data for this Ligand-Target Pair
Affinity DataIC50: 5.91nMpH: 7.5Assay Description:Binding of a desired RBP4 antagonist displaces retinol and induces hindrance for RBP4-TTR interaction resulting in the decreased FRET signal (FIG. 7)...More data for this Ligand-Target Pair
Affinity DataIC50: 6.04nMpH: 7.5Assay Description:Binding of a desired RBP4 antagonist displaces retinol and induces hindrance for RBP4-TTR interaction resulting in the decreased FRET signal (FIG. 7)...More data for this Ligand-Target Pair
Affinity DataIC50: 6.10nMpH: 7.4 T: 2°CAssay Description:Untagged human RBP4 purified from urine of tubular proteinuria patients was purchased from Fitzgerald Industries International. It was biotinylated u...More data for this Ligand-Target Pair
Affinity DataIC50: 6.17nMpH: 7.5Assay Description:Binding of a desired RBP4 antagonist displaces retinol and induces hindrance for RBP4-TTR interaction resulting in the decreased FRET signal (FIG. 7)...More data for this Ligand-Target Pair
Affinity DataIC50: 6.36nMpH: 7.5Assay Description:Binding of a desired RBP4 antagonist displaces retinol and induces hindrance for RBP4-TTR interaction resulting in the decreased FRET signal (FIG. 7)...More data for this Ligand-Target Pair
Affinity DataIC50: 6.75nMpH: 7.5Assay Description:Binding of a desired RBP4 antagonist displaces retinol and induces hindrance for RBP4-TTR interaction resulting in the decreased FRET signal (FIG. 7)...More data for this Ligand-Target Pair
Affinity DataIC50: 6.89nMpH: 7.5Assay Description:Binding of a desired RBP4 antagonist displaces retinol and induces hindrance for RBP4-TTR interaction resulting in the decreased FRET signal (FIG. 7)...More data for this Ligand-Target Pair
Affinity DataIC50: 6.95nMpH: 7.5Assay Description:Binding of a desired RBP4 antagonist displaces retinol and induces hindrance for RBP4-TTR interaction resulting in the decreased FRET signal (FIG. 7)...More data for this Ligand-Target Pair
Affinity DataIC50: 6.96nMpH: 7.5Assay Description:Binding of a desired RBP4 antagonist displaces retinol and induces hindrance for RBP4-TTR interaction resulting in the decreased FRET signal (FIG. 7)...More data for this Ligand-Target Pair
Affinity DataIC50: 7.06nMpH: 7.5Assay Description:Binding of a desired RBP4 antagonist displaces retinol and induces hindrance for RBP4-TTR interaction resulting in the decreased FRET signal (FIG. 7)...More data for this Ligand-Target Pair
Affinity DataIC50: 7.40nMpH: 7.5Assay Description:Binding of a desired RBP4 antagonist displaces retinol and induces hindrance for RBP4-TTR interaction resulting in the decreased FRET signal (FIG. 7)...More data for this Ligand-Target Pair
Affinity DataIC50: 7.41nMpH: 7.4 T: 2°CAssay Description:Untagged human RBP4 purified from urine of tubular proteinuria patients was purchased from Fitzgerald Industries International. It was biotinylated u...More data for this Ligand-Target Pair
Affinity DataIC50: 7.44nMpH: 7.5Assay Description:Binding of a desired RBP4 antagonist displaces retinol and induces hindrance for RBP4-TTR interaction resulting in the decreased FRET signal (FIG. 7)...More data for this Ligand-Target Pair
Affinity DataIC50: 7.5nMpH: 7.5Assay Description:Binding of a desired RBP4 antagonist displaces retinol and induces hindrance for RBP4-TTR interaction resulting in the decreased FRET signal (FIG. 7)...More data for this Ligand-Target Pair
Affinity DataIC50: 7.57nMpH: 7.5Assay Description:Binding of a desired RBP4 antagonist displaces retinol and induces hindrance for RBP4-TTR interaction resulting in the decreased FRET signal (FIG. 7)...More data for this Ligand-Target Pair
Affinity DataIC50: 7.78nMpH: 7.5Assay Description:Binding of a desired RBP4 antagonist displaces retinol and induces hindrance for RBP4-TTR interaction resulting in the decreased FRET signal (FIG. 7)...More data for this Ligand-Target Pair
Affinity DataIC50: 7.80nMpH: 7.5Assay Description:Binding of a desired RBP4 antagonist displaces retinol and induces hindrance for RBP4-TTR interaction resulting in the decreased FRET signal (FIG. 7)...More data for this Ligand-Target Pair
Affinity DataIC50: 7.96nMpH: 7.5Assay Description:Binding of a desired RBP4 antagonist displaces retinol and induces hindrance for RBP4-TTR interaction resulting in the decreased FRET signal (FIG. 7)...More data for this Ligand-Target Pair
Affinity DataIC50: 8nMpH: 7.5Assay Description:Binding of a desired RBP4 antagonist displaces retinol and induces hindrance for RBP4-TTR interaction resulting in the decreased FRET signal (FIG. 7)...More data for this Ligand-Target Pair
Affinity DataIC50: 8.06nMpH: 7.5Assay Description:Binding of a desired RBP4 antagonist displaces retinol and induces hindrance for RBP4-TTR interaction resulting in the decreased FRET signal (FIG. 7)...More data for this Ligand-Target Pair
Affinity DataIC50: 8.31nMpH: 7.5Assay Description:Binding of a desired RBP4 antagonist displaces retinol and induces hindrance for RBP4-TTR interaction resulting in the decreased FRET signal (FIG. 7)...More data for this Ligand-Target Pair
Affinity DataIC50: 8.5nMpH: 7.5Assay Description:Binding of a desired RBP4 antagonist displaces retinol and induces hindrance for RBP4-TTR interaction resulting in the decreased FRET signal (FIG. 7)...More data for this Ligand-Target Pair
Affinity DataIC50: 8.57nMpH: 7.5Assay Description:Binding of a desired RBP4 antagonist displaces retinol and induces hindrance for RBP4-TTR interaction resulting in the decreased FRET signal (FIG. 7)...More data for this Ligand-Target Pair