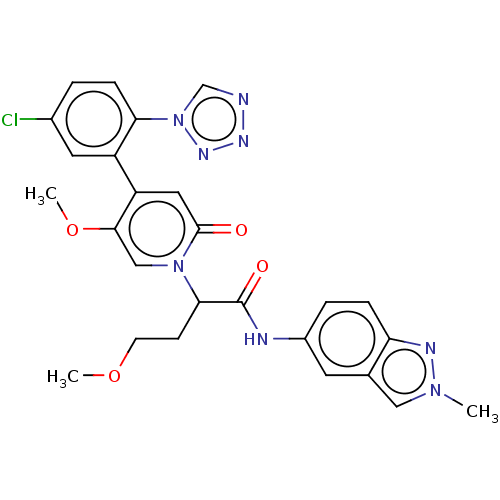
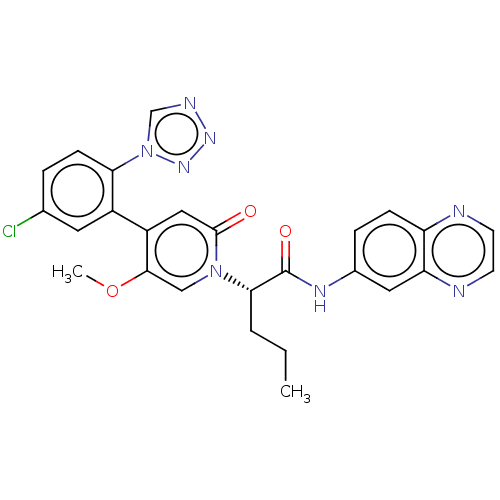
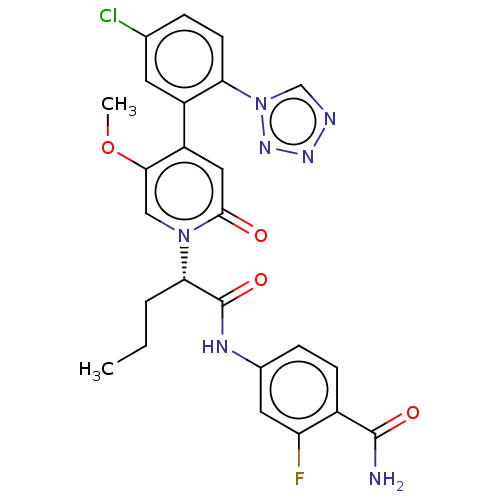
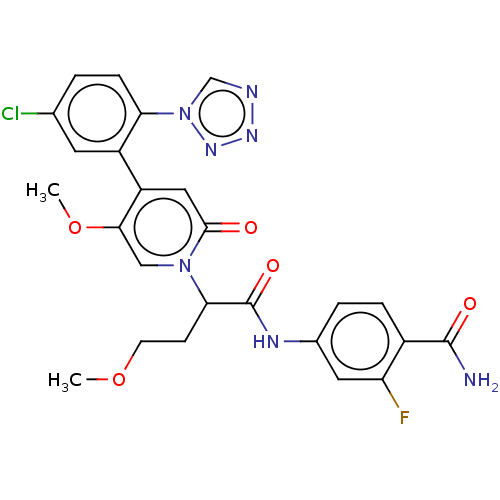
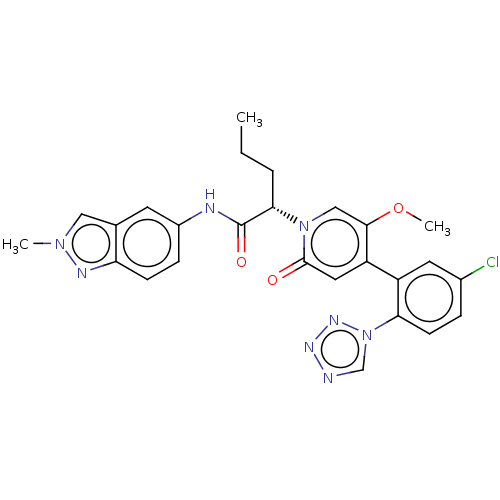
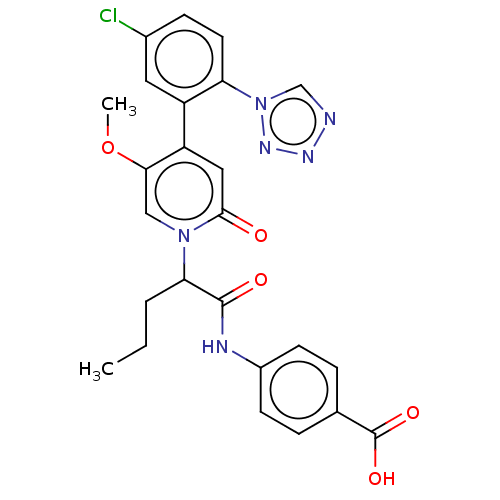
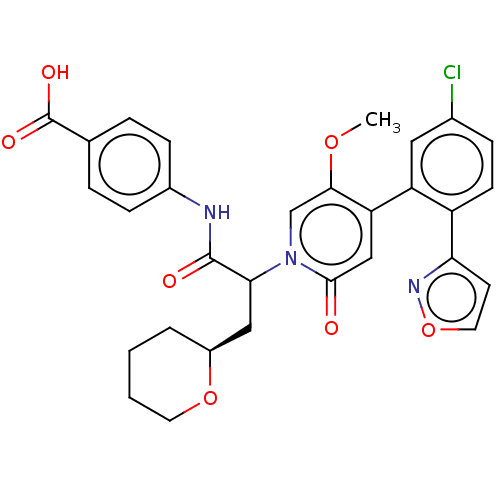
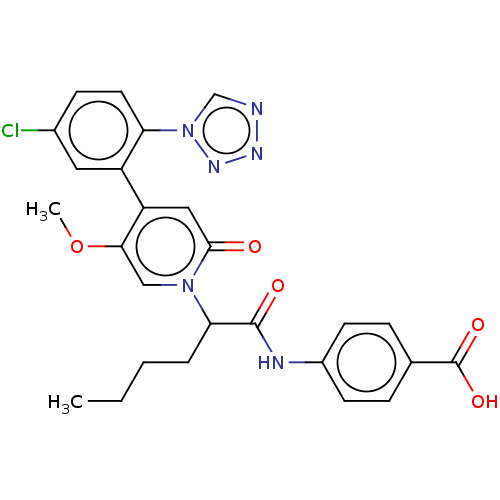
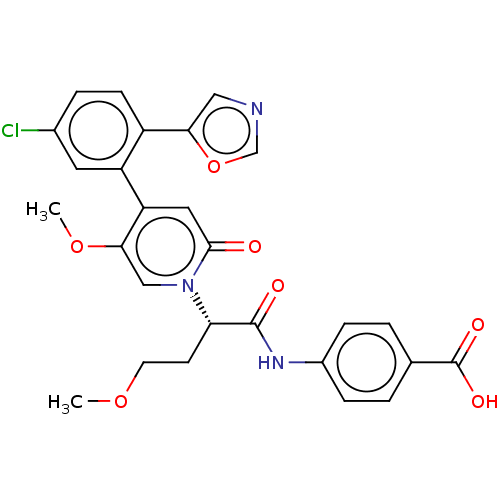
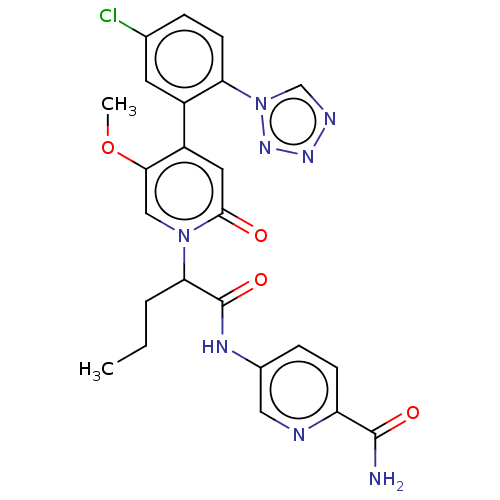
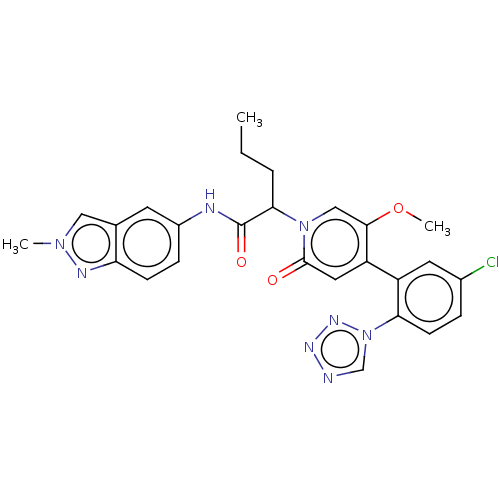
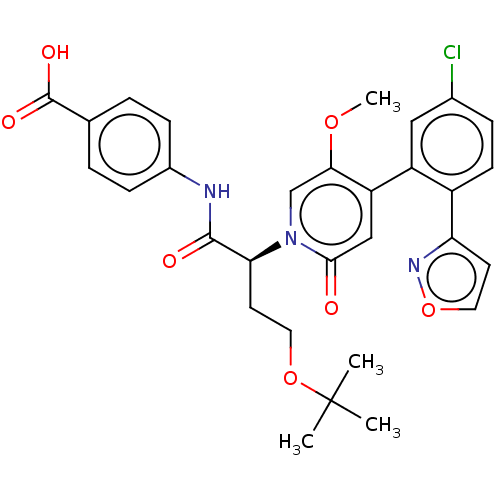
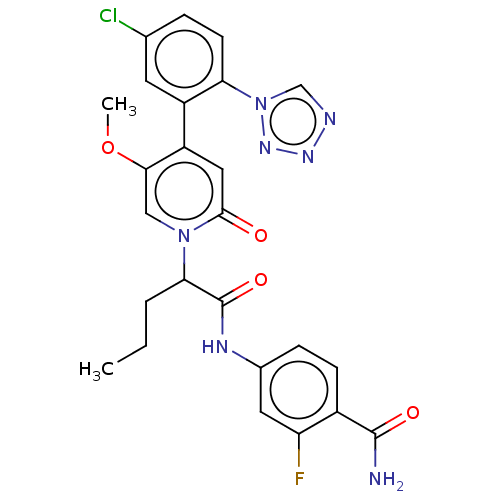
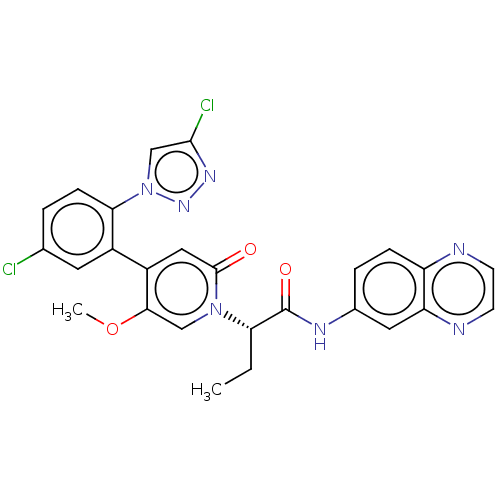
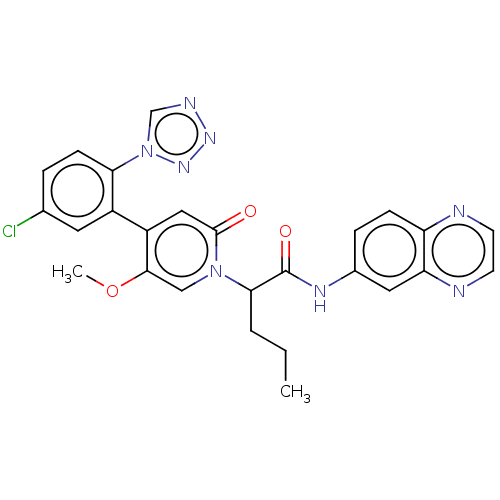
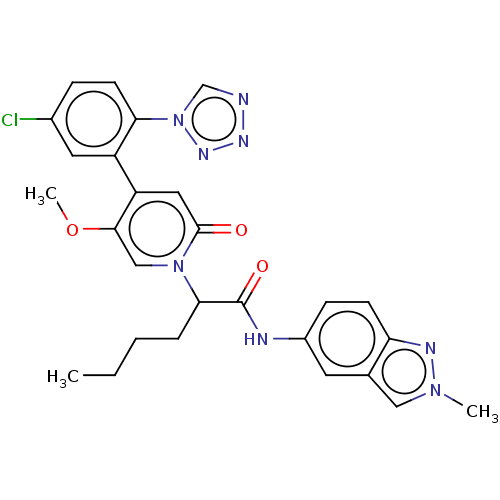
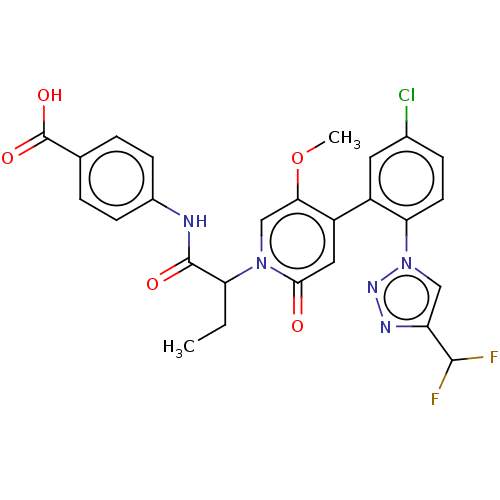
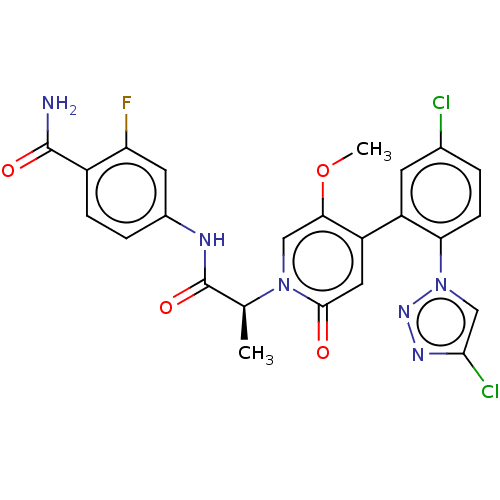
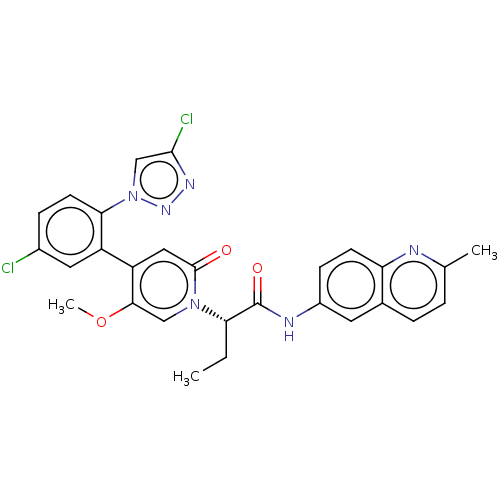
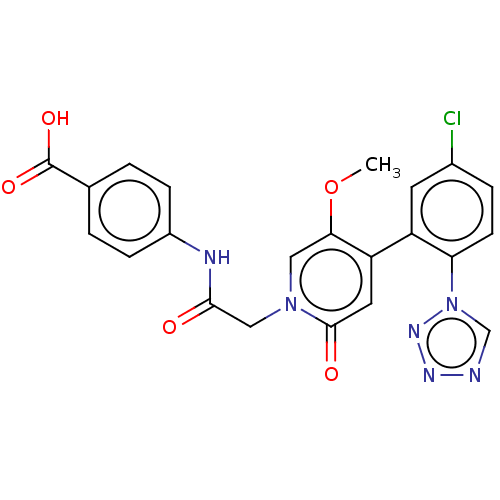
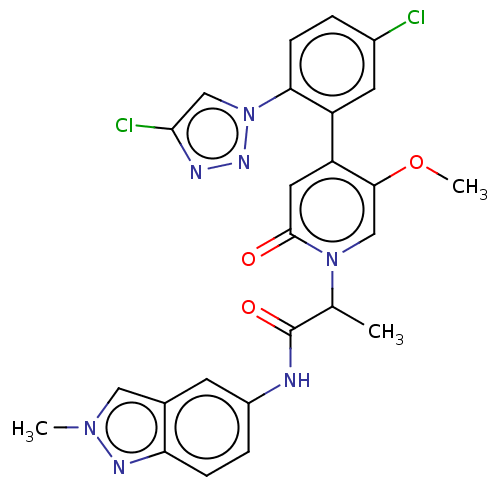
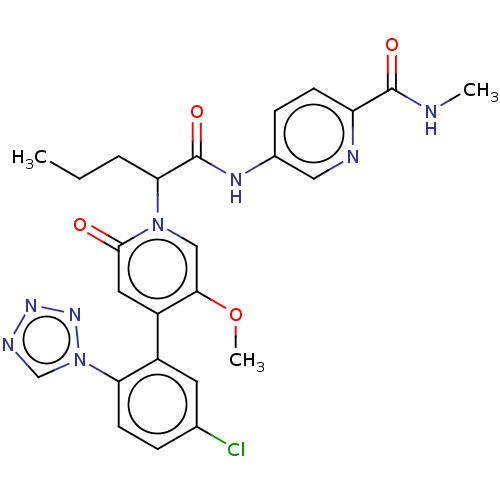
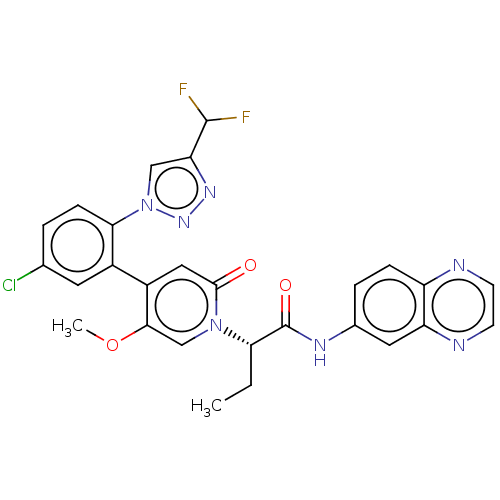
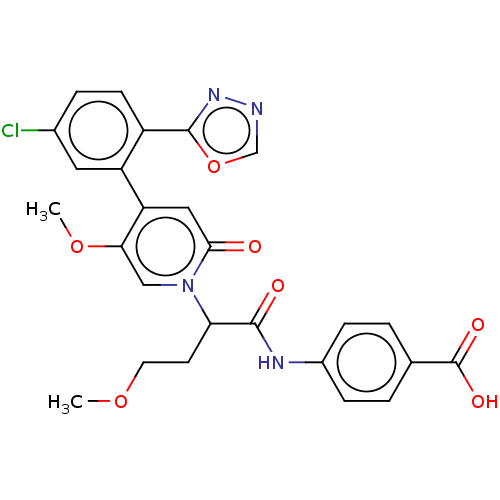
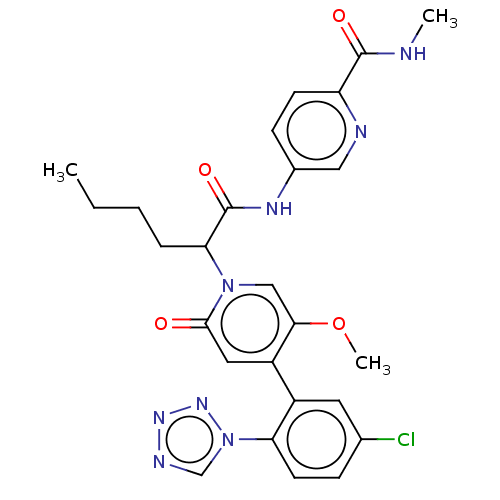
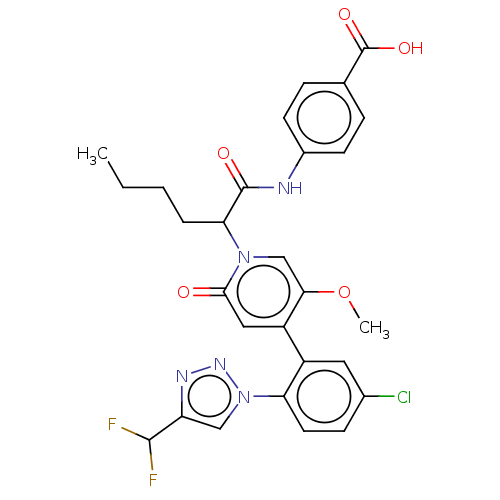
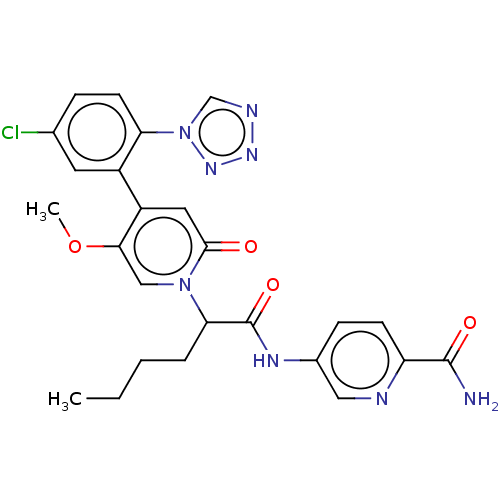
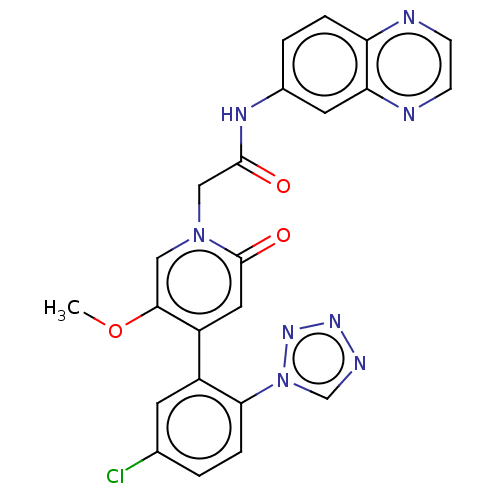
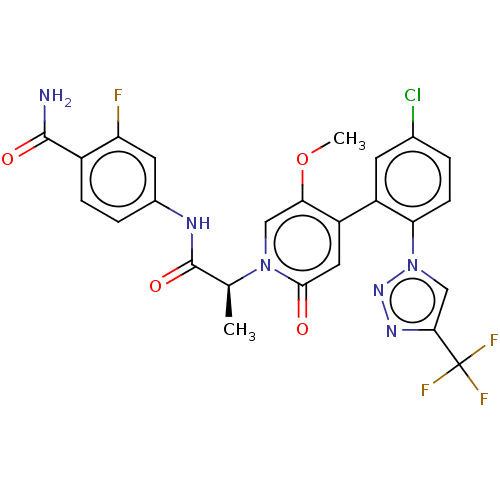
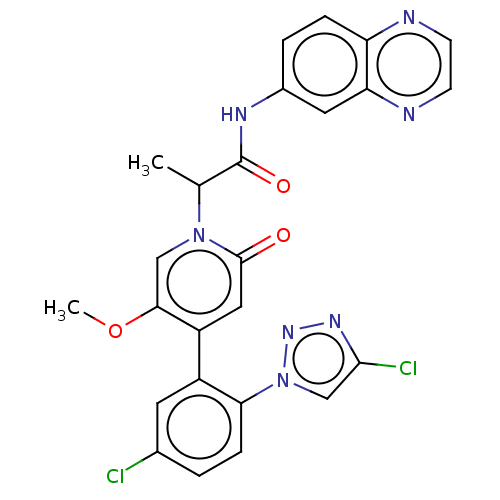
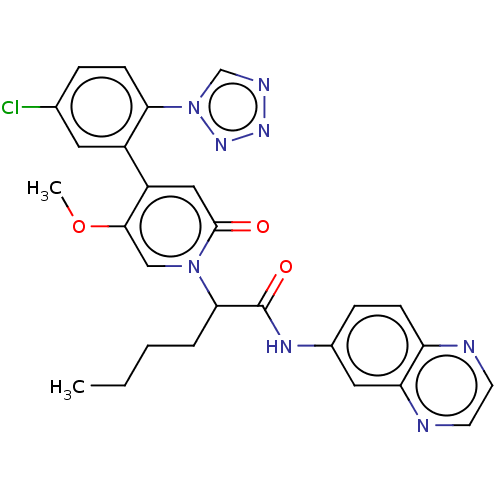
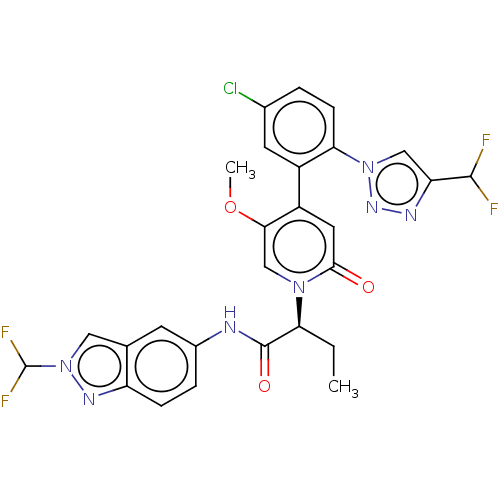
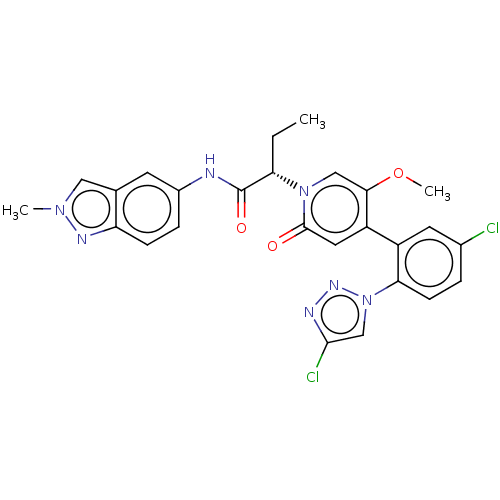
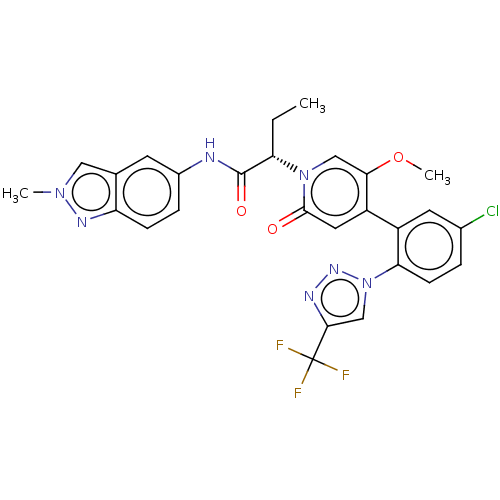
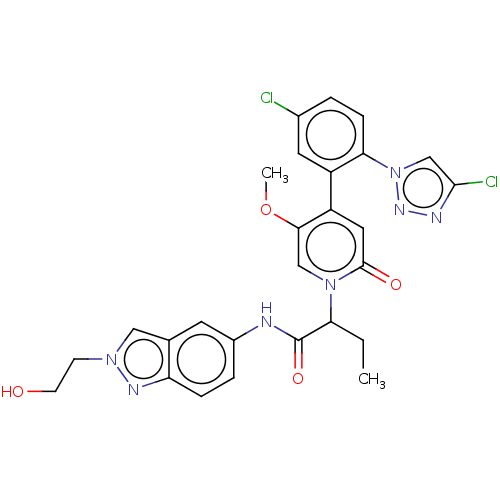
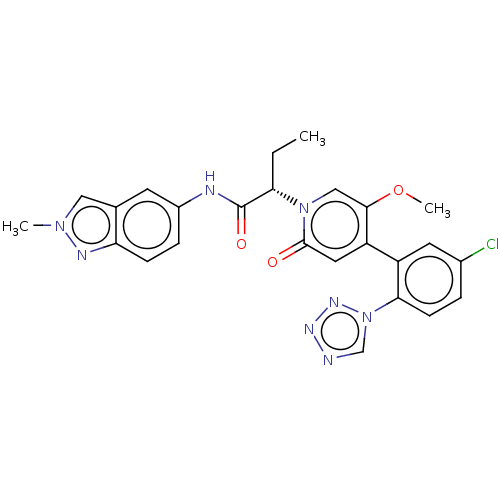
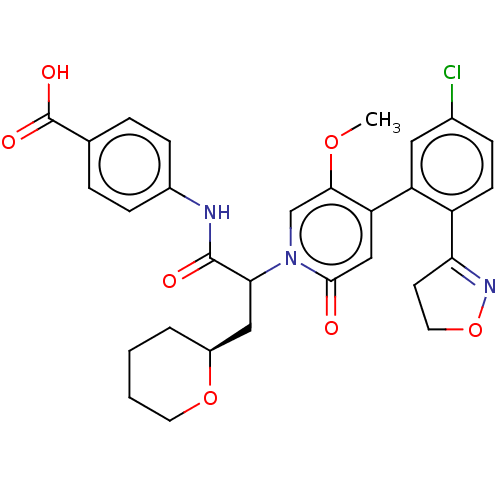
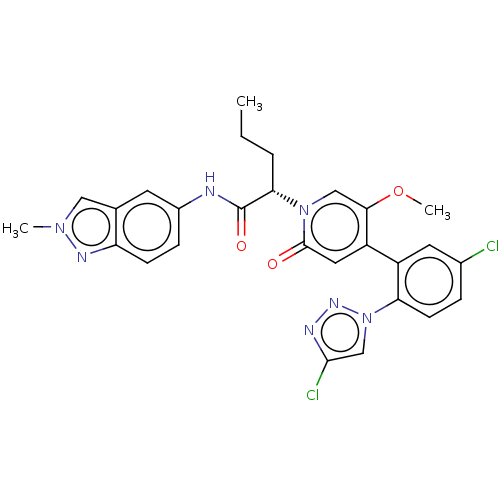
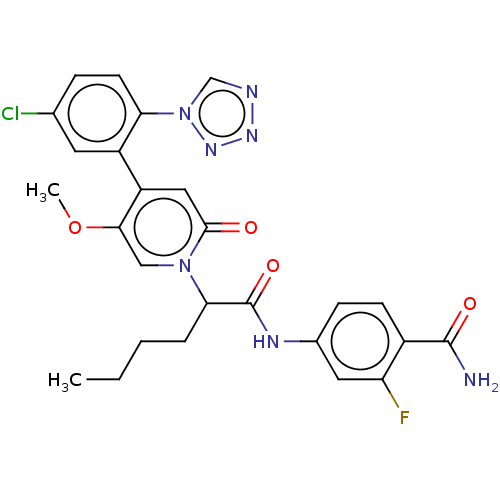
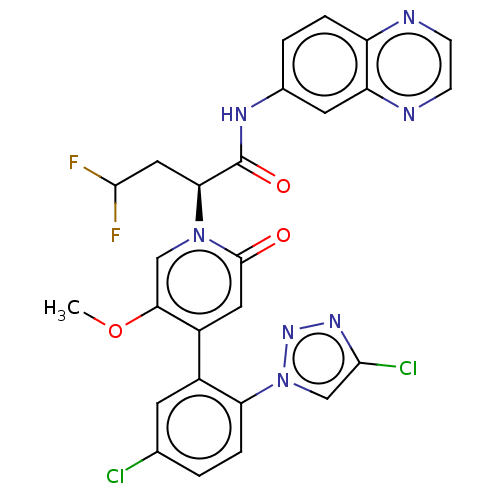
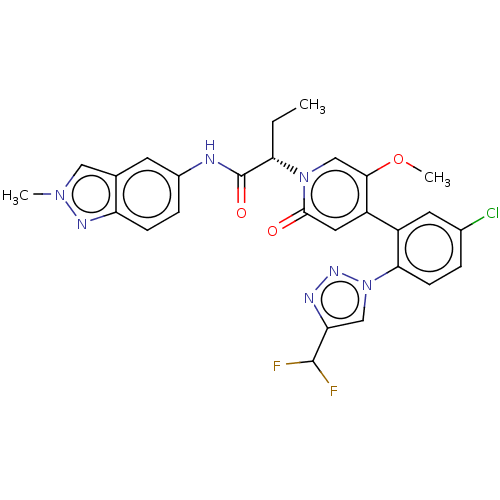
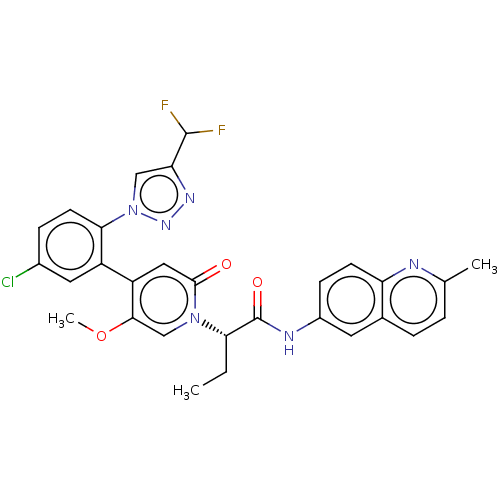
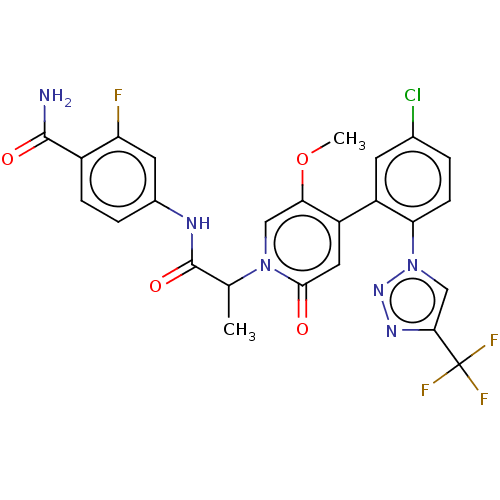
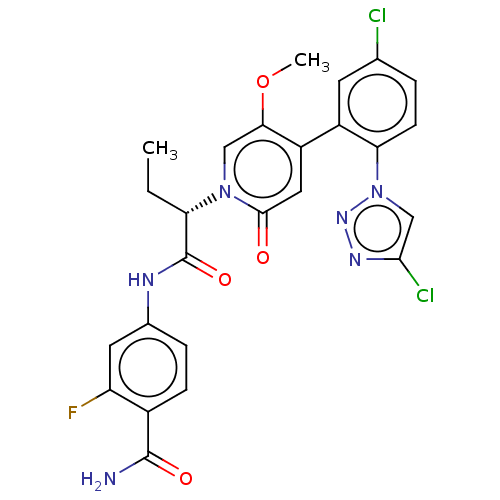
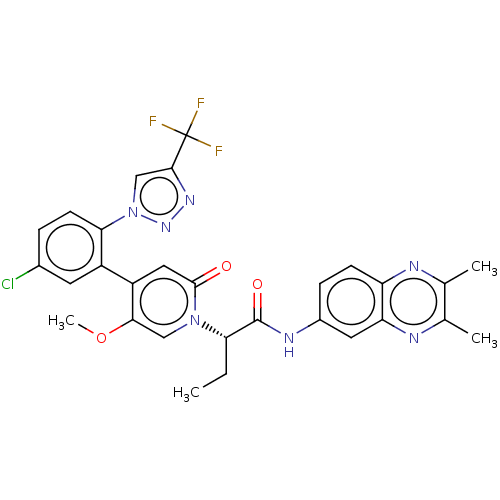
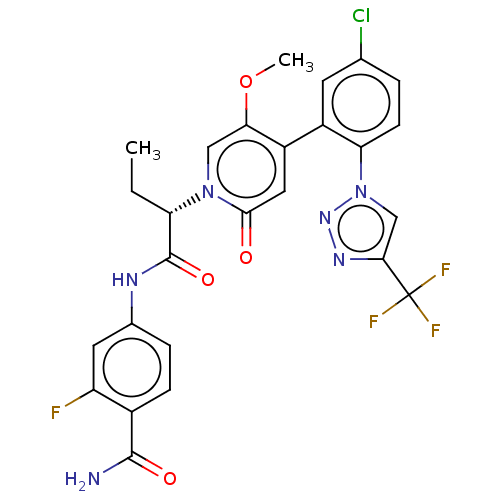
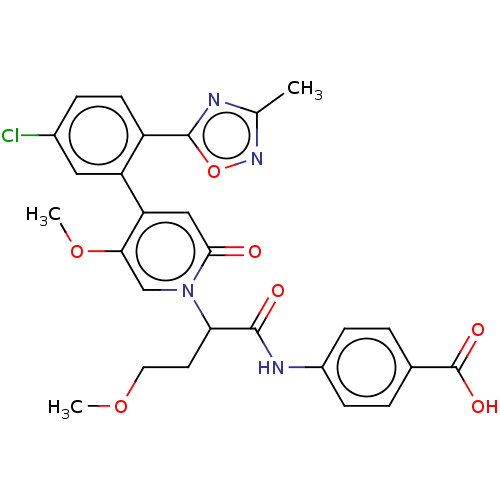
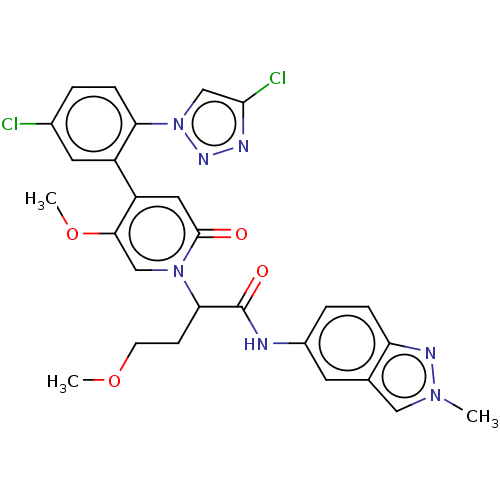
Report error Found 516 Enz. Inhib. hit(s) with all data for entry = 8704
Affinity DataIC50: 0.260nMAssay Description:The factor XIa inhibition of the substances according to the invention is determined using a biochemical test system which utilizes the reaction of a...More data for this Ligand-Target Pair
Affinity DataIC50: 0.260nMAssay Description:The factor XIa inhibition of the substances according to the invention is determined using a biochemical test system which utilizes the reaction of a...More data for this Ligand-Target Pair
Affinity DataIC50: 0.310nMAssay Description:The factor XIa inhibition of the substances according to the invention is determined using a biochemical test system which utilizes the reaction of a...More data for this Ligand-Target Pair
Affinity DataIC50: 0.340nMAssay Description:The factor XIa inhibition of the substances according to the invention is determined using a biochemical test system which utilizes the reaction of a...More data for this Ligand-Target Pair
Affinity DataIC50: 0.340nMAssay Description:The factor XIa inhibition of the substances according to the invention is determined using a biochemical test system which utilizes the reaction of a...More data for this Ligand-Target Pair
Affinity DataIC50: 0.360nMAssay Description:The factor XIa inhibition of the substances according to the invention is determined using a biochemical test system which utilizes the reaction of a...More data for this Ligand-Target Pair
Affinity DataIC50: 0.360nMAssay Description:The factor XIa inhibition of the substances according to the invention is determined using a biochemical test system which utilizes the reaction of a...More data for this Ligand-Target Pair
Affinity DataIC50: 0.360nMAssay Description:The factor XIa inhibition of the substances according to the invention is determined using a biochemical test system which utilizes the reaction of a...More data for this Ligand-Target Pair
Affinity DataIC50: 0.400nMAssay Description:The factor XIa inhibition of the substances according to the invention is determined using a biochemical test system which utilizes the reaction of a...More data for this Ligand-Target Pair
Affinity DataIC50: 0.460nMAssay Description:The factor XIa inhibition of the substances according to the invention is determined using a biochemical test system which utilizes the reaction of a...More data for this Ligand-Target Pair
Affinity DataIC50: 0.5nMAssay Description:The factor XIa inhibition of the substances according to the invention is determined using a biochemical test system which utilizes the reaction of a...More data for this Ligand-Target Pair
Affinity DataIC50: 0.530nMAssay Description:The factor XIa inhibition of the substances according to the invention is determined using a biochemical test system which utilizes the reaction of a...More data for this Ligand-Target Pair
Affinity DataIC50: 0.550nMAssay Description:The factor XIa inhibition of the substances according to the invention is determined using a biochemical test system which utilizes the reaction of a...More data for this Ligand-Target Pair
Affinity DataIC50: 0.550nMAssay Description:The factor XIa inhibition of the substances according to the invention is determined using a biochemical test system which utilizes the reaction of a...More data for this Ligand-Target Pair
Affinity DataIC50: 0.560nMAssay Description:The factor XIa inhibition of the substances according to the invention is determined using a biochemical test system which utilizes the reaction of a...More data for this Ligand-Target Pair
Affinity DataIC50: 0.570nMAssay Description:To determine the plasma kallikrein inhibition of the substances according to the invention, a biochemical test system is used which utilizes the reac...More data for this Ligand-Target Pair
Affinity DataIC50: 0.590nMAssay Description:The factor XIa inhibition of the substances according to the invention is determined using a biochemical test system which utilizes the reaction of a...More data for this Ligand-Target Pair
Affinity DataIC50: 0.620nMAssay Description:The factor XIa inhibition of the substances according to the invention is determined using a biochemical test system which utilizes the reaction of a...More data for this Ligand-Target Pair
Affinity DataIC50: 0.640nMAssay Description:The factor XIa inhibition of the substances according to the invention is determined using a biochemical test system which utilizes the reaction of a...More data for this Ligand-Target Pair
Affinity DataIC50: 0.650nMAssay Description:The factor XIa inhibition of the substances according to the invention is determined using a biochemical test system which utilizes the reaction of a...More data for this Ligand-Target Pair
Affinity DataIC50: 0.650nMAssay Description:The factor XIa inhibition of the substances according to the invention is determined using a biochemical test system which utilizes the reaction of a...More data for this Ligand-Target Pair
Affinity DataIC50: 0.650nMAssay Description:The factor XIa inhibition of the substances according to the invention is determined using a biochemical test system which utilizes the reaction of a...More data for this Ligand-Target Pair
Affinity DataIC50: 0.660nMAssay Description:The factor XIa inhibition of the substances according to the invention is determined using a biochemical test system which utilizes the reaction of a...More data for this Ligand-Target Pair
Affinity DataIC50: 0.700nMAssay Description:The factor XIa inhibition of the substances according to the invention is determined using a biochemical test system which utilizes the reaction of a...More data for this Ligand-Target Pair
Affinity DataIC50: 0.700nMAssay Description:The factor XIa inhibition of the substances according to the invention is determined using a biochemical test system which utilizes the reaction of a...More data for this Ligand-Target Pair
Affinity DataIC50: 0.710nMAssay Description:The factor XIa inhibition of the substances according to the invention is determined using a biochemical test system which utilizes the reaction of a...More data for this Ligand-Target Pair
Affinity DataIC50: 0.710nMAssay Description:The factor XIa inhibition of the substances according to the invention is determined using a biochemical test system which utilizes the reaction of a...More data for this Ligand-Target Pair
Affinity DataIC50: 0.720nMAssay Description:The factor XIa inhibition of the substances according to the invention is determined using a biochemical test system which utilizes the reaction of a...More data for this Ligand-Target Pair
Affinity DataIC50: 0.730nMAssay Description:The factor XIa inhibition of the substances according to the invention is determined using a biochemical test system which utilizes the reaction of a...More data for this Ligand-Target Pair
Affinity DataIC50: 0.740nMAssay Description:The factor XIa inhibition of the substances according to the invention is determined using a biochemical test system which utilizes the reaction of a...More data for this Ligand-Target Pair
Affinity DataIC50: 0.75nMAssay Description:The factor XIa inhibition of the substances according to the invention is determined using a biochemical test system which utilizes the reaction of a...More data for this Ligand-Target Pair
Affinity DataIC50: 0.760nMAssay Description:The factor XIa inhibition of the substances according to the invention is determined using a biochemical test system which utilizes the reaction of a...More data for this Ligand-Target Pair
Affinity DataIC50: 0.780nMAssay Description:The factor XIa inhibition of the substances according to the invention is determined using a biochemical test system which utilizes the reaction of a...More data for this Ligand-Target Pair
Affinity DataIC50: 0.780nMAssay Description:The factor XIa inhibition of the substances according to the invention is determined using a biochemical test system which utilizes the reaction of a...More data for this Ligand-Target Pair
Affinity DataIC50: 0.780nMAssay Description:To determine the plasma kallikrein inhibition of the substances according to the invention, a biochemical test system is used which utilizes the reac...More data for this Ligand-Target Pair
Affinity DataIC50: 0.790nMAssay Description:The factor XIa inhibition of the substances according to the invention is determined using a biochemical test system which utilizes the reaction of a...More data for this Ligand-Target Pair
Affinity DataIC50: 0.810nMAssay Description:The factor XIa inhibition of the substances according to the invention is determined using a biochemical test system which utilizes the reaction of a...More data for this Ligand-Target Pair
Affinity DataIC50: 0.820nMAssay Description:The factor XIa inhibition of the substances according to the invention is determined using a biochemical test system which utilizes the reaction of a...More data for this Ligand-Target Pair
Affinity DataIC50: 0.830nMAssay Description:The factor XIa inhibition of the substances according to the invention is determined using a biochemical test system which utilizes the reaction of a...More data for this Ligand-Target Pair
Affinity DataIC50: 0.840nMAssay Description:The factor XIa inhibition of the substances according to the invention is determined using a biochemical test system which utilizes the reaction of a...More data for this Ligand-Target Pair
Affinity DataIC50: 0.840nMAssay Description:The factor XIa inhibition of the substances according to the invention is determined using a biochemical test system which utilizes the reaction of a...More data for this Ligand-Target Pair
Affinity DataIC50: 0.850nMAssay Description:The factor XIa inhibition of the substances according to the invention is determined using a biochemical test system which utilizes the reaction of a...More data for this Ligand-Target Pair
Affinity DataIC50: 0.860nMAssay Description:The factor XIa inhibition of the substances according to the invention is determined using a biochemical test system which utilizes the reaction of a...More data for this Ligand-Target Pair
Affinity DataIC50: 0.870nMAssay Description:The factor XIa inhibition of the substances according to the invention is determined using a biochemical test system which utilizes the reaction of a...More data for this Ligand-Target Pair
Affinity DataIC50: 0.890nMAssay Description:The factor XIa inhibition of the substances according to the invention is determined using a biochemical test system which utilizes the reaction of a...More data for this Ligand-Target Pair
Affinity DataIC50: 0.890nMAssay Description:The factor XIa inhibition of the substances according to the invention is determined using a biochemical test system which utilizes the reaction of a...More data for this Ligand-Target Pair
Affinity DataIC50: 0.890nMAssay Description:The factor XIa inhibition of the substances according to the invention is determined using a biochemical test system which utilizes the reaction of a...More data for this Ligand-Target Pair
Affinity DataIC50: 0.920nMAssay Description:The factor XIa inhibition of the substances according to the invention is determined using a biochemical test system which utilizes the reaction of a...More data for this Ligand-Target Pair
Affinity DataIC50: 0.920nMAssay Description:The factor XIa inhibition of the substances according to the invention is determined using a biochemical test system which utilizes the reaction of a...More data for this Ligand-Target Pair
Affinity DataIC50: 0.960nMAssay Description:The factor XIa inhibition of the substances according to the invention is determined using a biochemical test system which utilizes the reaction of a...More data for this Ligand-Target Pair