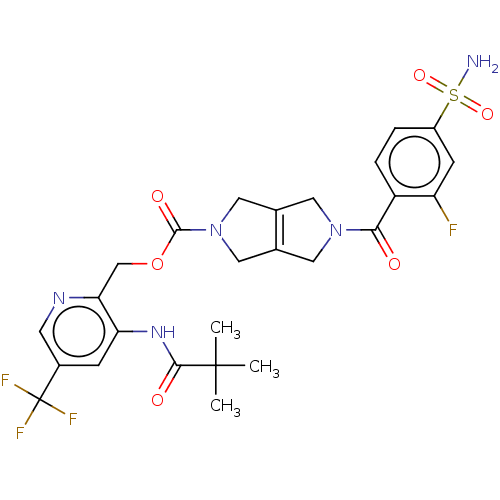
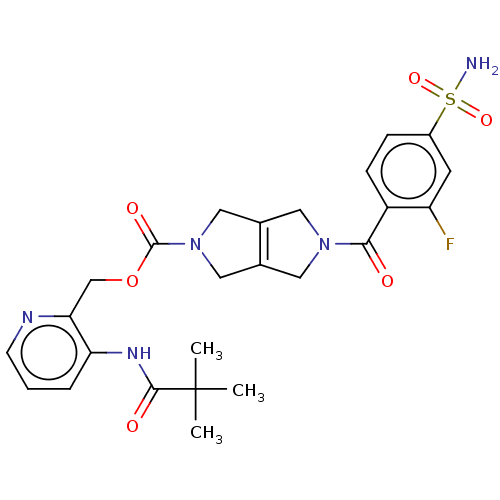
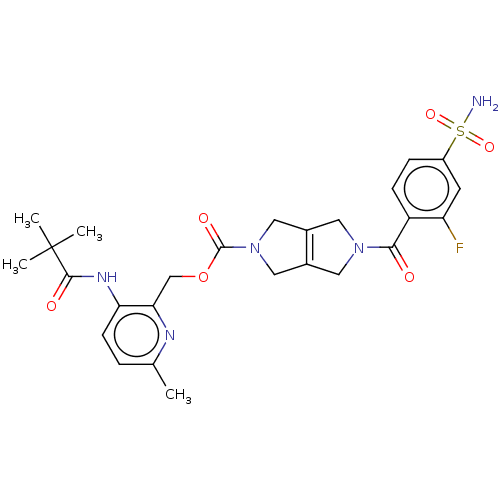
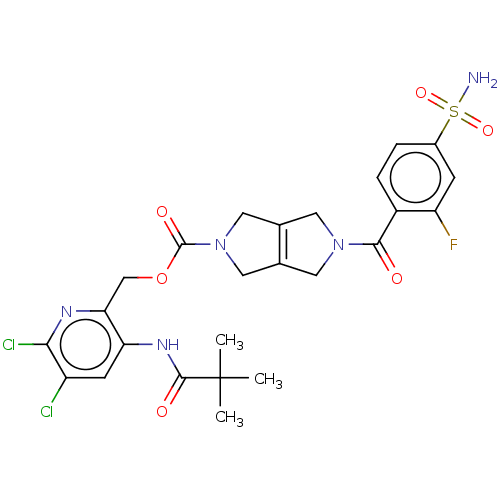
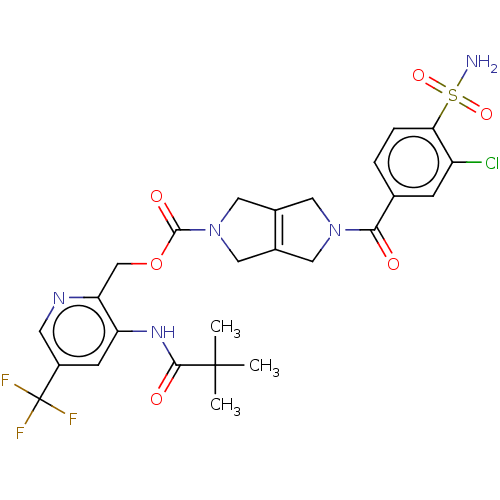
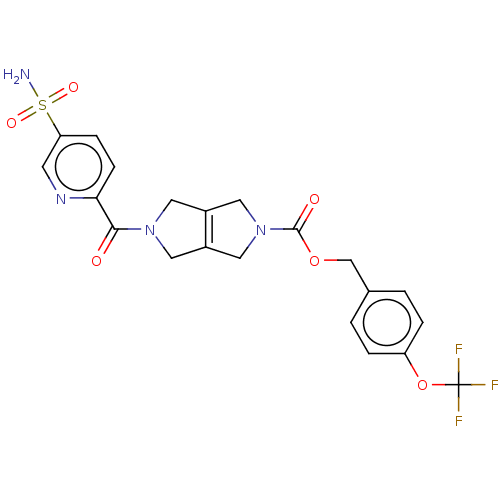
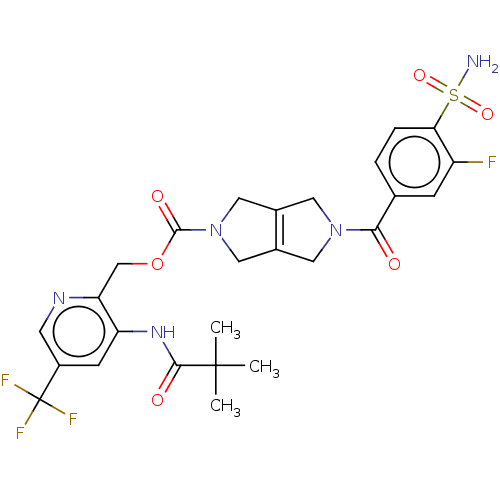
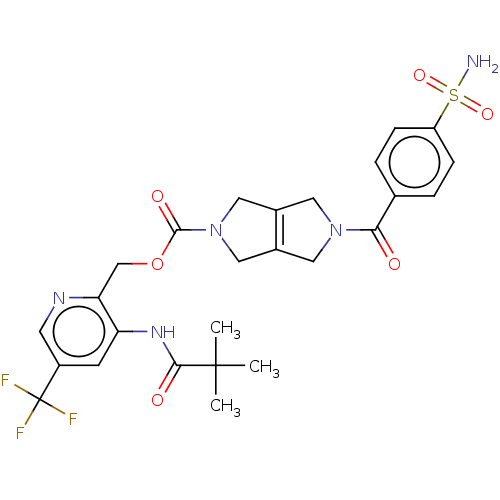
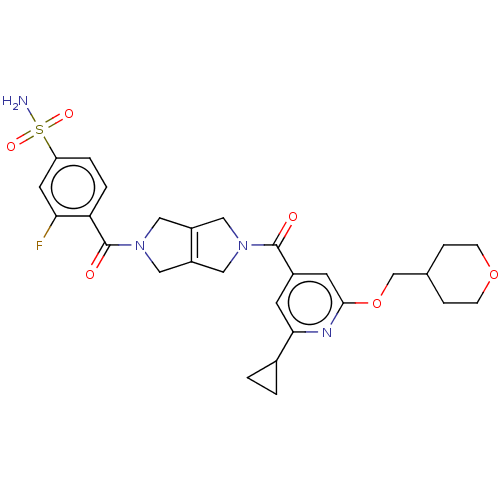
Report error Found 107 Enz. Inhib. hit(s) with all data for entry = 9109
Affinity DataIC50: 1nMAssay Description:Human carbonic anhydrase II (hCA-II) inhibition was measured by an absorbance method using 4-nitrophenyl acetate (4-NPA) as its substrate. 4-NPA can ...More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:Human carbonic anhydrase II (hCA-II) inhibition was measured by an absorbance method using 4-nitrophenyl acetate (4-NPA) as its substrate. 4-NPA can ...More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:ATX inhibition was measured by a fluorescence quenching assay using a specifically labeled substrate analogue (MR121 substrate). To obtain this MR121...More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:ATX inhibition was measured by a fluorescence quenching assay using a specifically labeled substrate analogue (MR121 substrate). To obtain this MR121...More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:ATX inhibition was measured by a fluorescence quenching assay using a specifically labeled substrate analogue (MR121 substrate). To obtain this MR121...More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:ATX inhibition was measured by a fluorescence quenching assay using a specifically labeled substrate analogue (MR121 substrate). To obtain this MR121...More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:ATX inhibition was measured by a fluorescence quenching assay using a specifically labeled substrate analogue (MR121 substrate). To obtain this MR121...More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:Human carbonic anhydrase II (hCA-II) inhibition was measured by an absorbance method using 4-nitrophenyl acetate (4-NPA) as its substrate. 4-NPA can ...More data for this Ligand-Target Pair
Affinity DataIC50: 1.20nMAssay Description:Human carbonic anhydrase II (hCA-II) inhibition was measured by an absorbance method using 4-nitrophenyl acetate (4-NPA) as its substrate. 4-NPA can ...More data for this Ligand-Target Pair
Affinity DataIC50: 1.90nMAssay Description:Human carbonic anhydrase II (hCA-II) inhibition was measured by an absorbance method using 4-nitrophenyl acetate (4-NPA) as its substrate. 4-NPA can ...More data for this Ligand-Target Pair
Affinity DataIC50: 2nMAssay Description:ATX inhibition was measured by a fluorescence quenching assay using a specifically labeled substrate analogue (MR121 substrate). To obtain this MR121...More data for this Ligand-Target Pair
Affinity DataIC50: 2nMAssay Description:Human carbonic anhydrase II (hCA-II) inhibition was measured by an absorbance method using 4-nitrophenyl acetate (4-NPA) as its substrate. 4-NPA can ...More data for this Ligand-Target Pair
Affinity DataIC50: 2nMAssay Description:ATX inhibition was measured by a fluorescence quenching assay using a specifically labeled substrate analogue (MR121 substrate). To obtain this MR121...More data for this Ligand-Target Pair
Affinity DataIC50: 2nMAssay Description:ATX inhibition was measured by a fluorescence quenching assay using a specifically labeled substrate analogue (MR121 substrate). To obtain this MR121...More data for this Ligand-Target Pair
Affinity DataIC50: 2nMAssay Description:Human carbonic anhydrase II (hCA-II) inhibition was measured by an absorbance method using 4-nitrophenyl acetate (4-NPA) as its substrate. 4-NPA can ...More data for this Ligand-Target Pair
Affinity DataIC50: 2nMAssay Description:ATX inhibition was measured by a fluorescence quenching assay using a specifically labeled substrate analogue (MR121 substrate). To obtain this MR121...More data for this Ligand-Target Pair
Affinity DataIC50: 2nMAssay Description:ATX inhibition was measured by a fluorescence quenching assay using a specifically labeled substrate analogue (MR121 substrate). To obtain this MR121...More data for this Ligand-Target Pair
Affinity DataIC50: 2.30nMAssay Description:Human carbonic anhydrase II (hCA-II) inhibition was measured by an absorbance method using 4-nitrophenyl acetate (4-NPA) as its substrate. 4-NPA can ...More data for this Ligand-Target Pair
Affinity DataIC50: 2.5nMAssay Description:Human carbonic anhydrase II (hCA-II) inhibition was measured by an absorbance method using 4-nitrophenyl acetate (4-NPA) as its substrate. 4-NPA can ...More data for this Ligand-Target Pair
Affinity DataIC50: 3nMAssay Description:ATX inhibition was measured by a fluorescence quenching assay using a specifically labeled substrate analogue (MR121 substrate). To obtain this MR121...More data for this Ligand-Target Pair
Affinity DataIC50: 3nMAssay Description:ATX inhibition was measured by a fluorescence quenching assay using a specifically labeled substrate analogue (MR121 substrate). To obtain this MR121...More data for this Ligand-Target Pair
Affinity DataIC50: 3nMAssay Description:Human carbonic anhydrase II (hCA-II) inhibition was measured by an absorbance method using 4-nitrophenyl acetate (4-NPA) as its substrate. 4-NPA can ...More data for this Ligand-Target Pair
Affinity DataIC50: 3nMAssay Description:ATX inhibition was measured by a fluorescence quenching assay using a specifically labeled substrate analogue (MR121 substrate). To obtain this MR121...More data for this Ligand-Target Pair
Affinity DataIC50: 3nMAssay Description:ATX inhibition was measured by a fluorescence quenching assay using a specifically labeled substrate analogue (MR121 substrate). To obtain this MR121...More data for this Ligand-Target Pair
Affinity DataIC50: 3nMAssay Description:ATX inhibition was measured by a fluorescence quenching assay using a specifically labeled substrate analogue (MR121 substrate). To obtain this MR121...More data for this Ligand-Target Pair
Affinity DataIC50: 3nMAssay Description:Human carbonic anhydrase II (hCA-II) inhibition was measured by an absorbance method using 4-nitrophenyl acetate (4-NPA) as its substrate. 4-NPA can ...More data for this Ligand-Target Pair
Affinity DataIC50: 3.60nMAssay Description:Human carbonic anhydrase II (hCA-II) inhibition was measured by an absorbance method using 4-nitrophenyl acetate (4-NPA) as its substrate. 4-NPA can ...More data for this Ligand-Target Pair
Affinity DataIC50: 3.80nMAssay Description:Human carbonic anhydrase II (hCA-II) inhibition was measured by an absorbance method using 4-nitrophenyl acetate (4-NPA) as its substrate. 4-NPA can ...More data for this Ligand-Target Pair
Affinity DataIC50: 4nMAssay Description:ATX inhibition was measured by a fluorescence quenching assay using a specifically labeled substrate analogue (MR121 substrate). To obtain this MR121...More data for this Ligand-Target Pair
Affinity DataIC50: 4nMAssay Description:ATX inhibition was measured by a fluorescence quenching assay using a specifically labeled substrate analogue (MR121 substrate). To obtain this MR121...More data for this Ligand-Target Pair
Affinity DataIC50: 4nMAssay Description:ATX inhibition was measured by a fluorescence quenching assay using a specifically labeled substrate analogue (MR121 substrate). To obtain this MR121...More data for this Ligand-Target Pair
Affinity DataIC50: 4nMAssay Description:Human carbonic anhydrase II (hCA-II) inhibition was measured by an absorbance method using 4-nitrophenyl acetate (4-NPA) as its substrate. 4-NPA can ...More data for this Ligand-Target Pair
Affinity DataIC50: 4nMAssay Description:ATX inhibition was measured by a fluorescence quenching assay using a specifically labeled substrate analogue (MR121 substrate). To obtain this MR121...More data for this Ligand-Target Pair
Affinity DataIC50: 4nMAssay Description:Human carbonic anhydrase II (hCA-II) inhibition was measured by an absorbance method using 4-nitrophenyl acetate (4-NPA) as its substrate. 4-NPA can ...More data for this Ligand-Target Pair
Affinity DataIC50: 4.20nMAssay Description:Human carbonic anhydrase II (hCA-II) inhibition was measured by an absorbance method using 4-nitrophenyl acetate (4-NPA) as its substrate. 4-NPA can ...More data for this Ligand-Target Pair
Affinity DataIC50: 5nMAssay Description:ATX inhibition was measured by a fluorescence quenching assay using a specifically labeled substrate analogue (MR121 substrate). To obtain this MR121...More data for this Ligand-Target Pair
Affinity DataIC50: 5nMAssay Description:ATX inhibition was measured by a fluorescence quenching assay using a specifically labeled substrate analogue (MR121 substrate). To obtain this MR121...More data for this Ligand-Target Pair
Affinity DataIC50: 5nMAssay Description:Human carbonic anhydrase II (hCA-II) inhibition was measured by an absorbance method using 4-nitrophenyl acetate (4-NPA) as its substrate. 4-NPA can ...More data for this Ligand-Target Pair
Affinity DataIC50: 5nMAssay Description:ATX inhibition was measured by a fluorescence quenching assay using a specifically labeled substrate analogue (MR121 substrate). To obtain this MR121...More data for this Ligand-Target Pair
Affinity DataIC50: 5nMAssay Description:Human carbonic anhydrase II (hCA-II) inhibition was measured by an absorbance method using 4-nitrophenyl acetate (4-NPA) as its substrate. 4-NPA can ...More data for this Ligand-Target Pair
Affinity DataIC50: 5nMAssay Description:ATX inhibition was measured by a fluorescence quenching assay using a specifically labeled substrate analogue (MR121 substrate). To obtain this MR121...More data for this Ligand-Target Pair
Affinity DataIC50: 5nMAssay Description:Human carbonic anhydrase II (hCA-II) inhibition was measured by an absorbance method using 4-nitrophenyl acetate (4-NPA) as its substrate. 4-NPA can ...More data for this Ligand-Target Pair
Affinity DataIC50: 5nMAssay Description:ATX inhibition was measured by a fluorescence quenching assay using a specifically labeled substrate analogue (MR121 substrate). To obtain this MR121...More data for this Ligand-Target Pair
Affinity DataIC50: 5nMAssay Description:ATX inhibition was measured by a fluorescence quenching assay using a specifically labeled substrate analogue (MR121 substrate). To obtain this MR121...More data for this Ligand-Target Pair
Affinity DataIC50: 5nMAssay Description:Human carbonic anhydrase II (hCA-II) inhibition was measured by an absorbance method using 4-nitrophenyl acetate (4-NPA) as its substrate. 4-NPA can ...More data for this Ligand-Target Pair
Affinity DataIC50: 6nMAssay Description:ATX inhibition was measured by a fluorescence quenching assay using a specifically labeled substrate analogue (MR121 substrate). To obtain this MR121...More data for this Ligand-Target Pair
Affinity DataIC50: 6nMAssay Description:Human carbonic anhydrase II (hCA-II) inhibition was measured by an absorbance method using 4-nitrophenyl acetate (4-NPA) as its substrate. 4-NPA can ...More data for this Ligand-Target Pair
Affinity DataIC50: 6nMAssay Description:Human carbonic anhydrase II (hCA-II) inhibition was measured by an absorbance method using 4-nitrophenyl acetate (4-NPA) as its substrate. 4-NPA can ...More data for this Ligand-Target Pair
Affinity DataIC50: 6nMAssay Description:Human carbonic anhydrase II (hCA-II) inhibition was measured by an absorbance method using 4-nitrophenyl acetate (4-NPA) as its substrate. 4-NPA can ...More data for this Ligand-Target Pair
Affinity DataIC50: 6nMAssay Description:ATX inhibition was measured by a fluorescence quenching assay using a specifically labeled substrate analogue (MR121 substrate). To obtain this MR121...More data for this Ligand-Target Pair
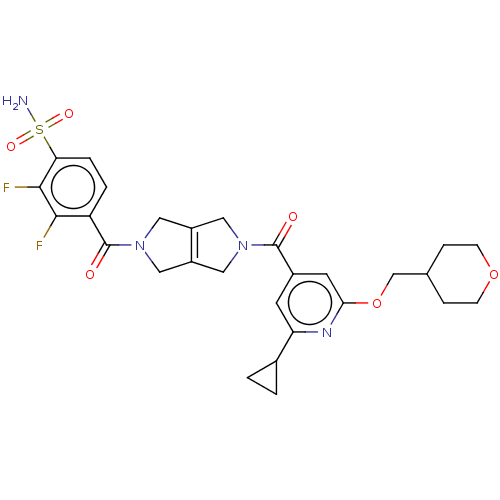
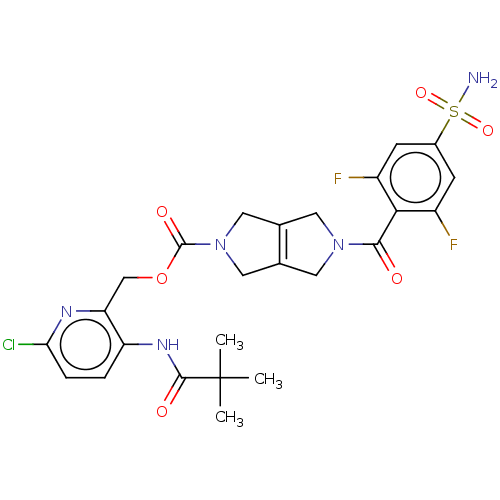
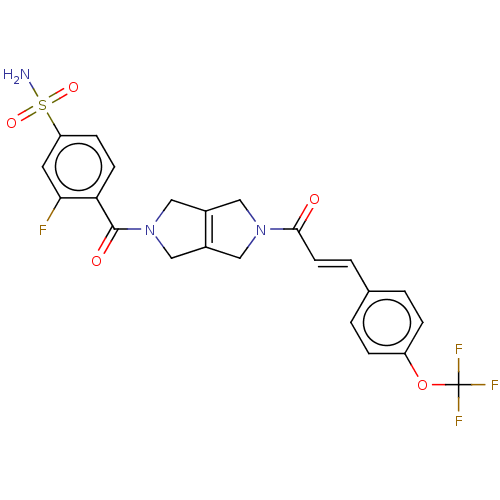
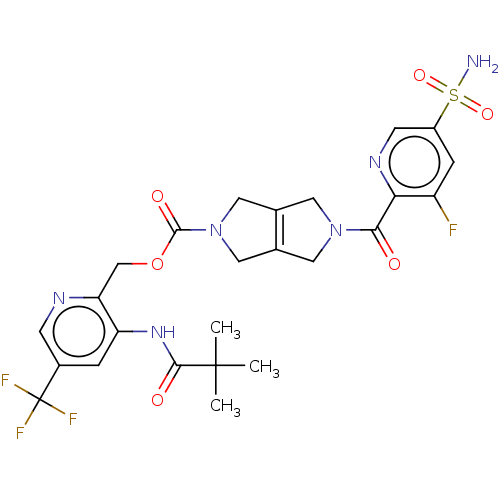
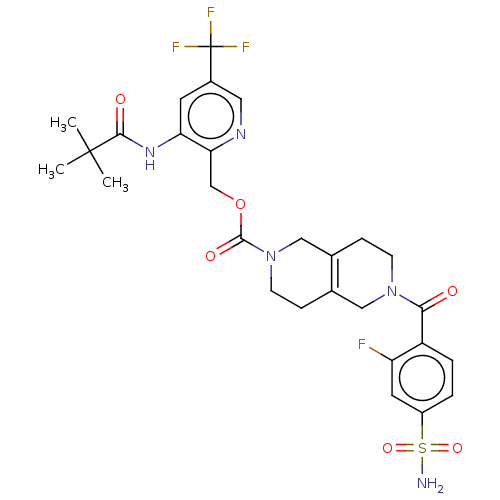
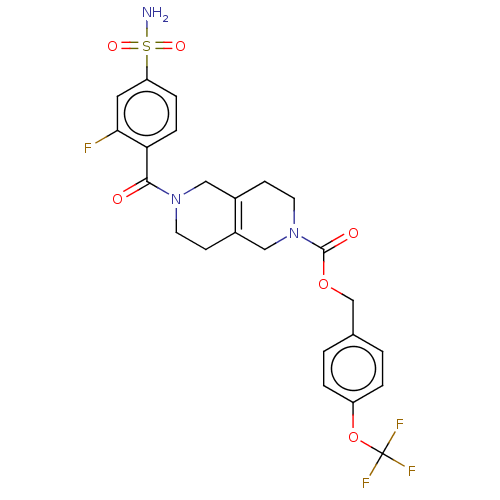
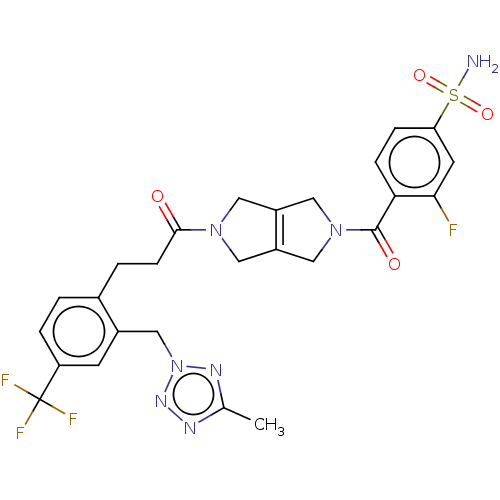
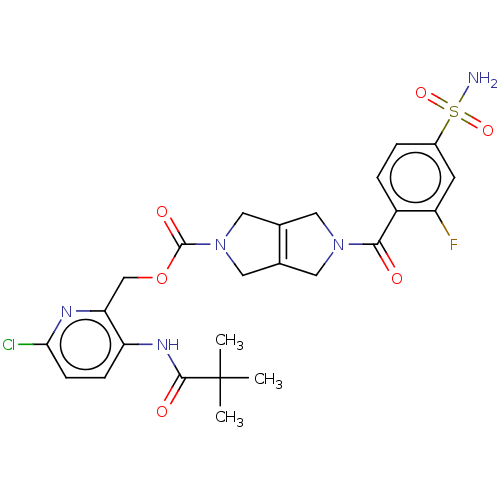
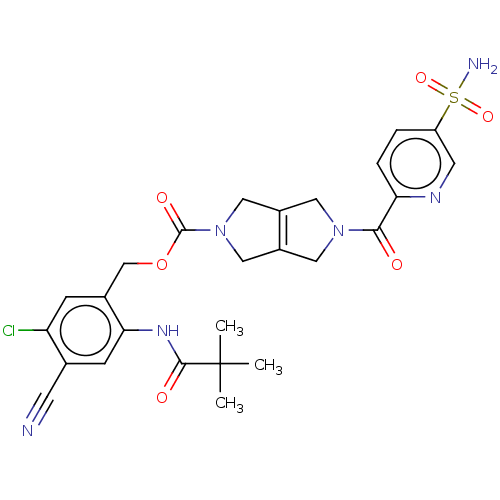
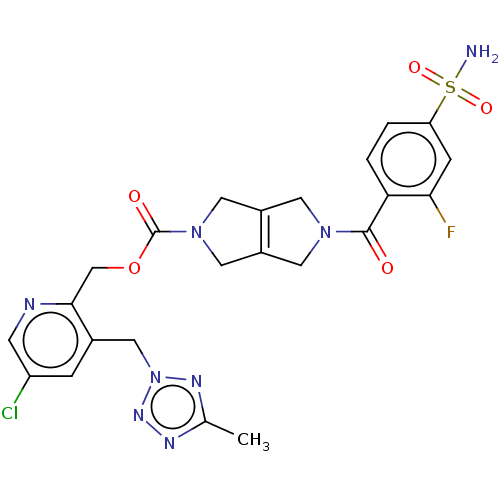
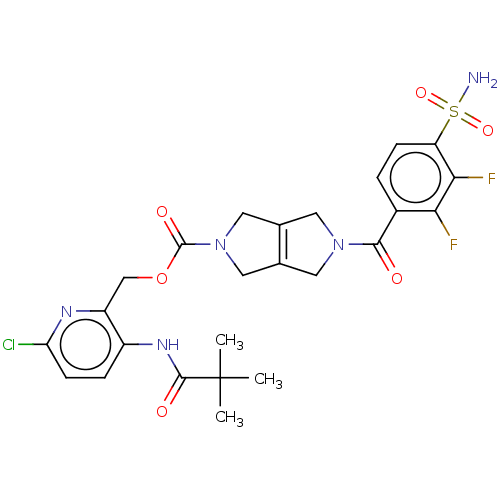
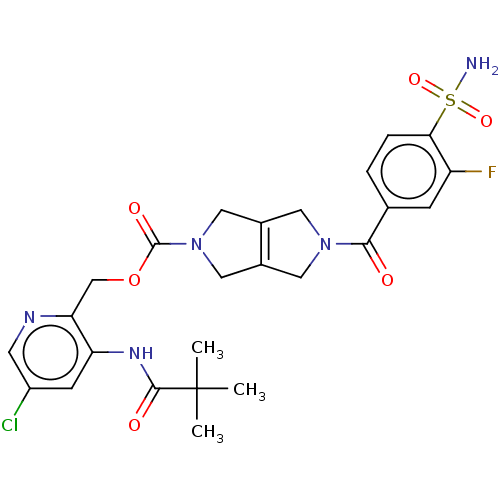
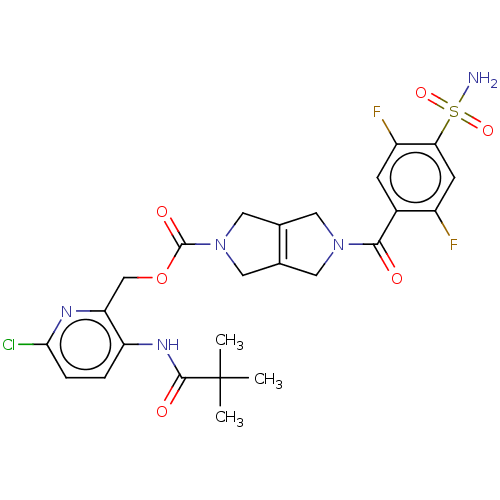
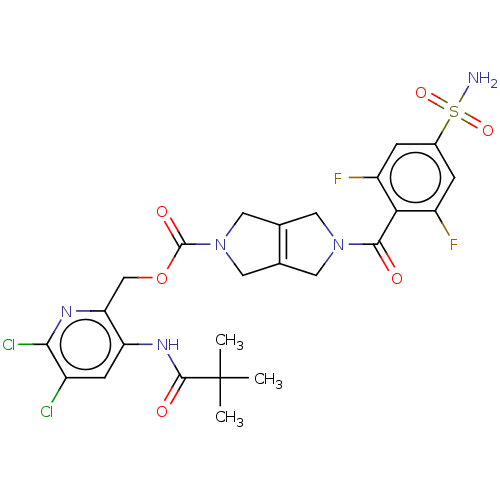
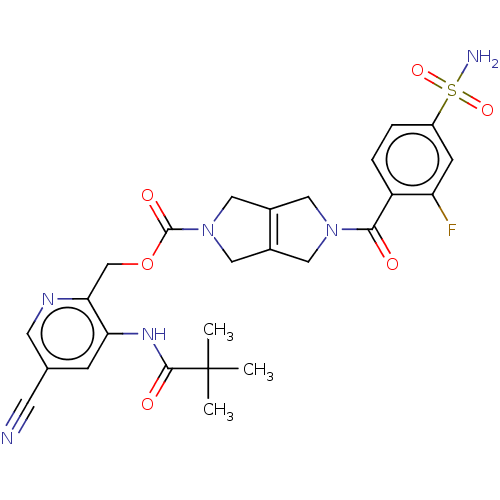
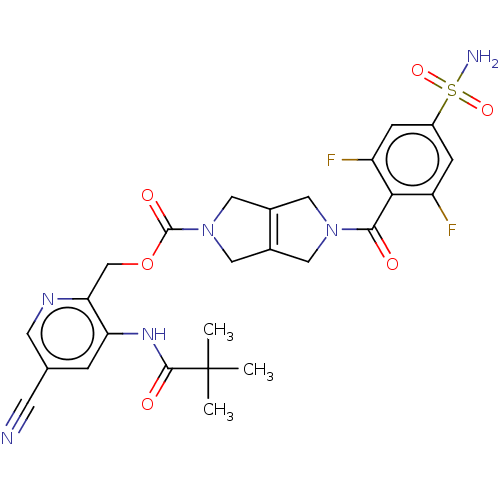
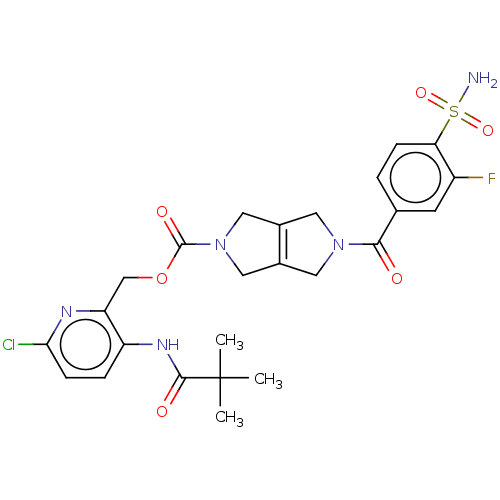
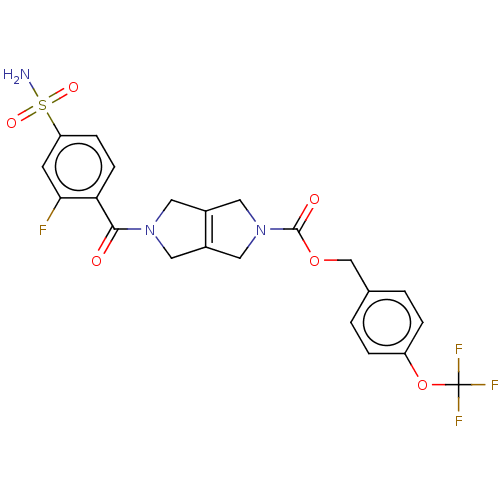
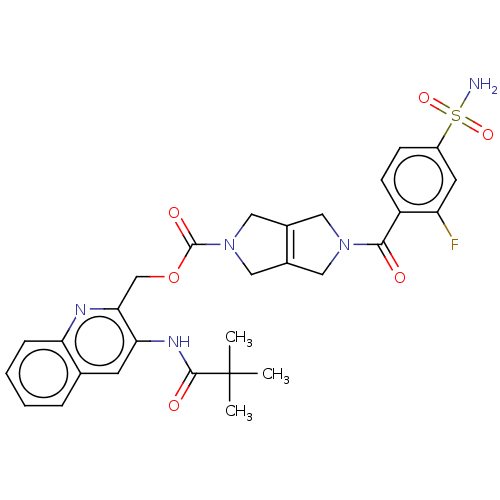
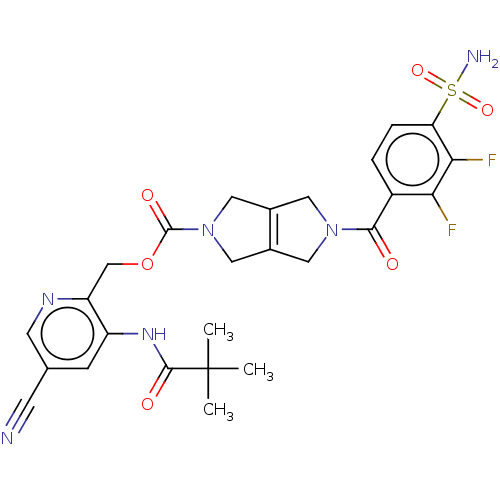
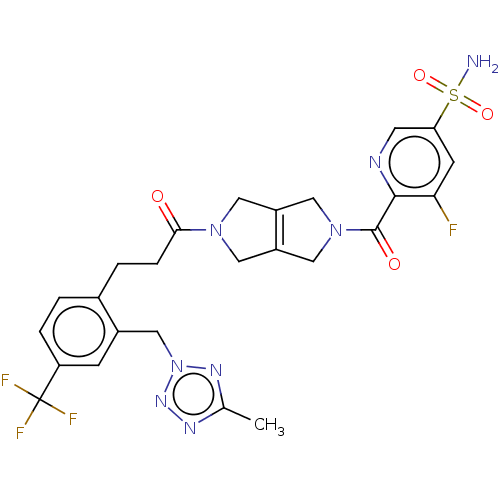
 3D Structure (crystal)
3D Structure (crystal)