Report error Found 149 Enz. Inhib. hit(s) with all data for entry = 9181
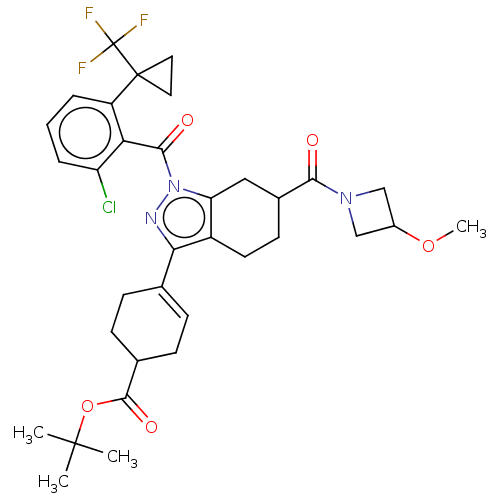
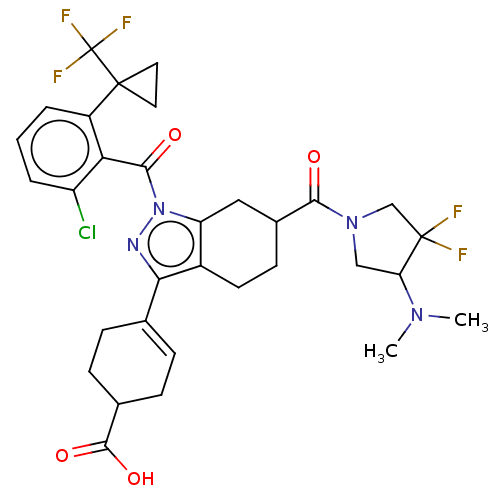
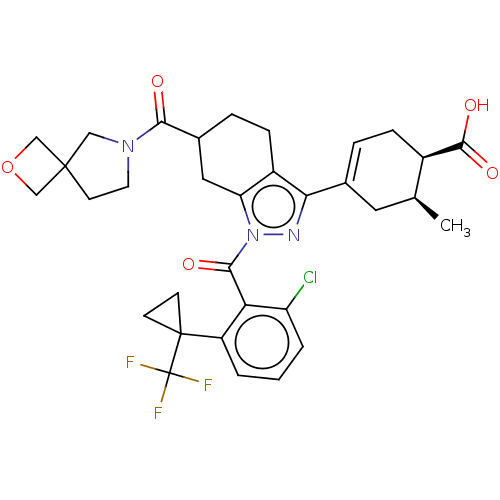
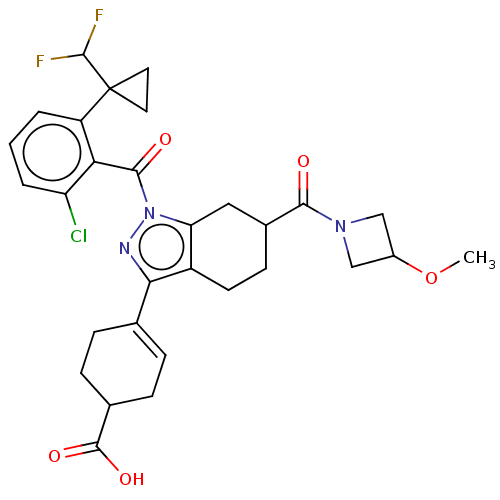
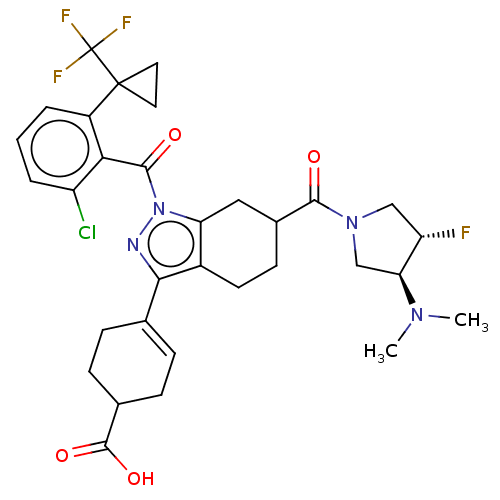
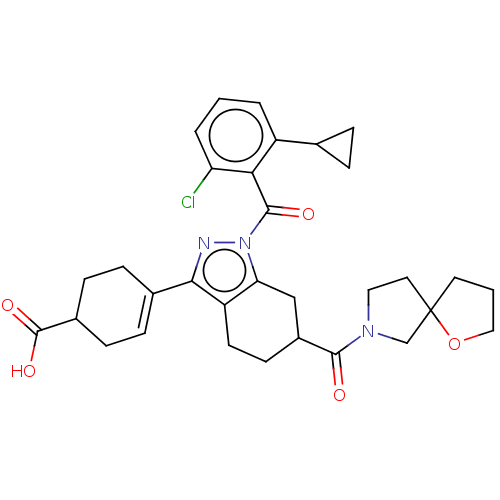
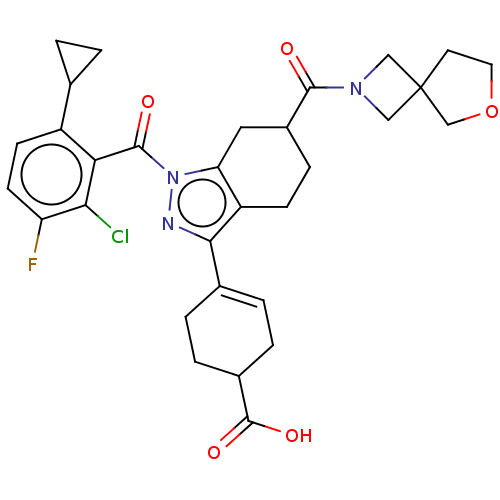
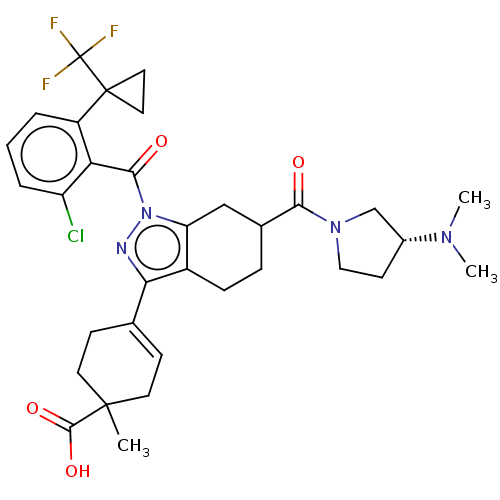
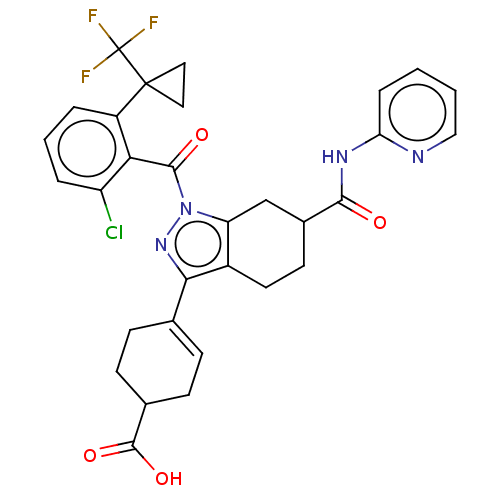
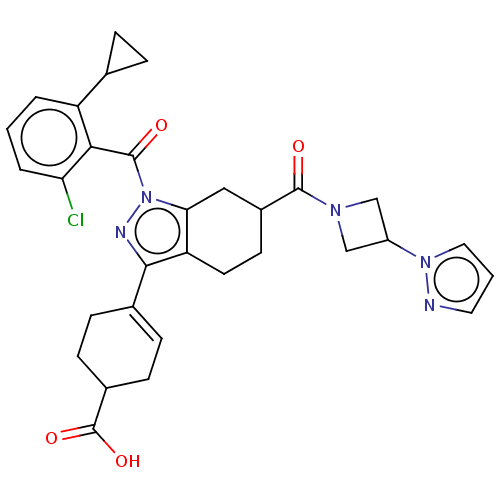
Affinity DataIC50: 1nMAssay Description:The compounds of the invention inhibit RORgammaT activity. Activation of RORgammaT activity can be measured using, e.g., biochemical TR-FRET assay. I...More data for this Ligand-Target Pair
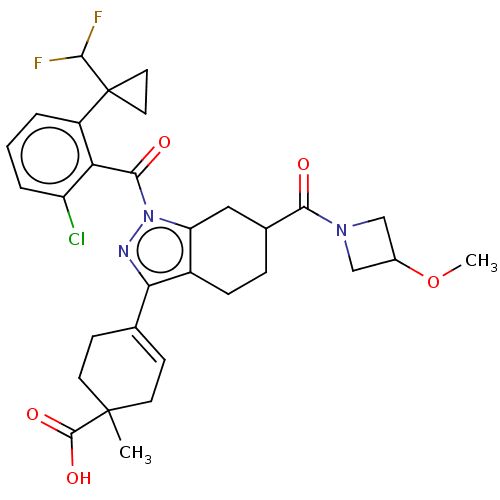
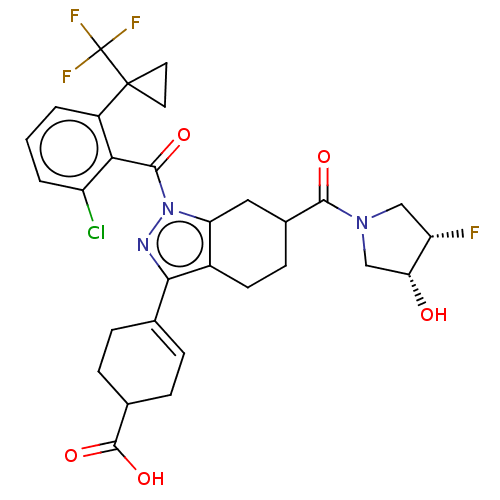
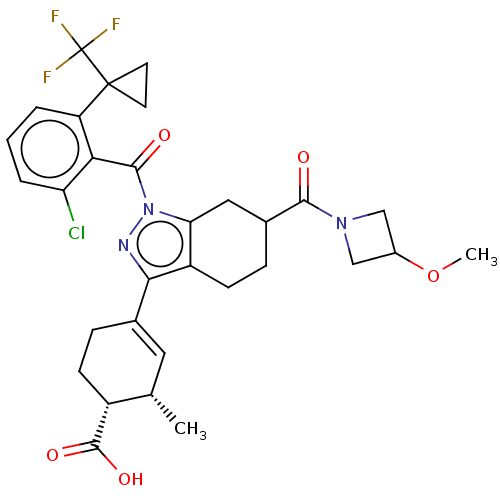
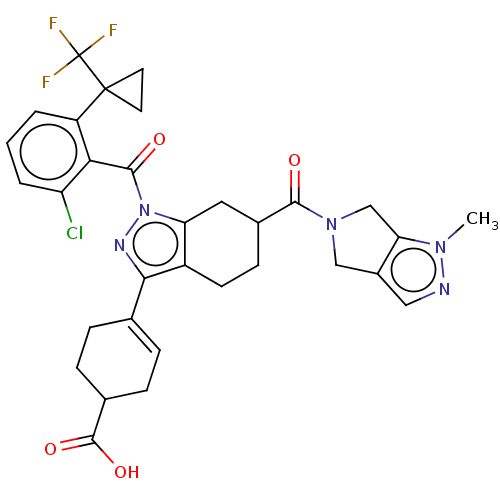
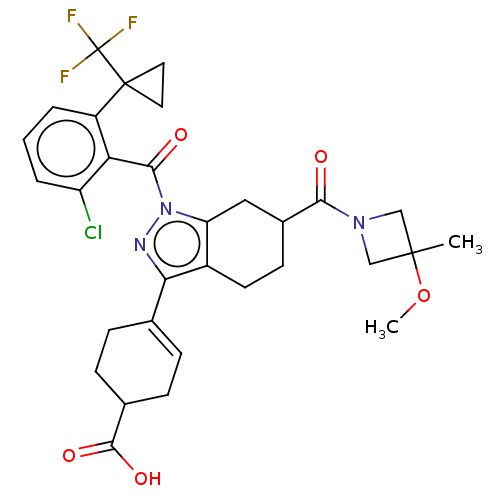
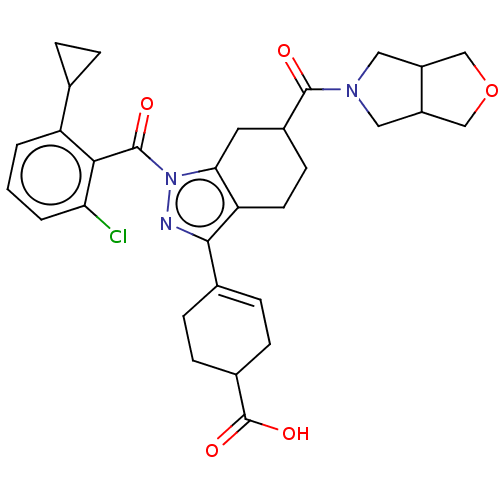
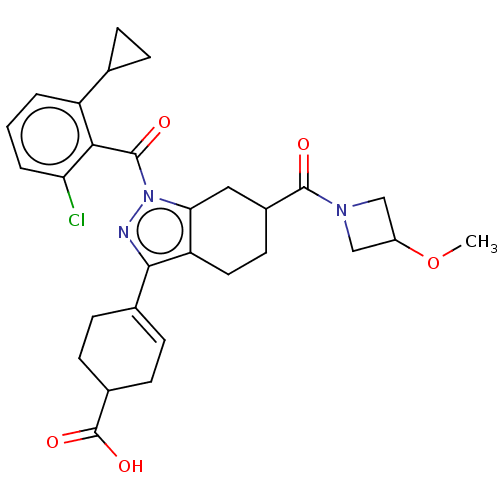
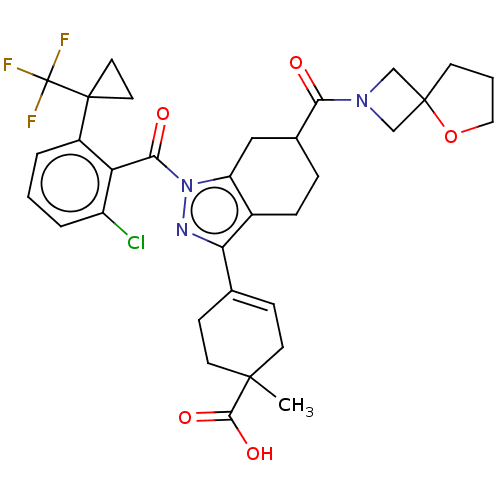
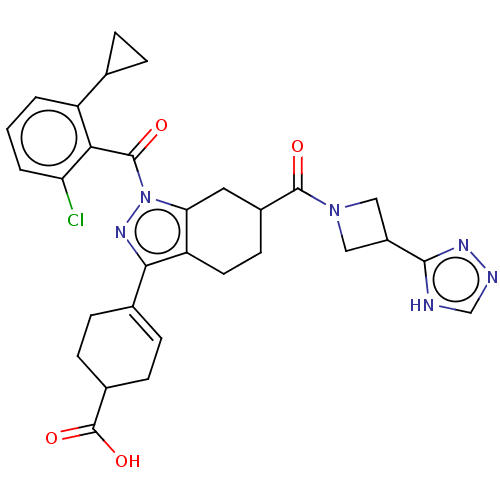
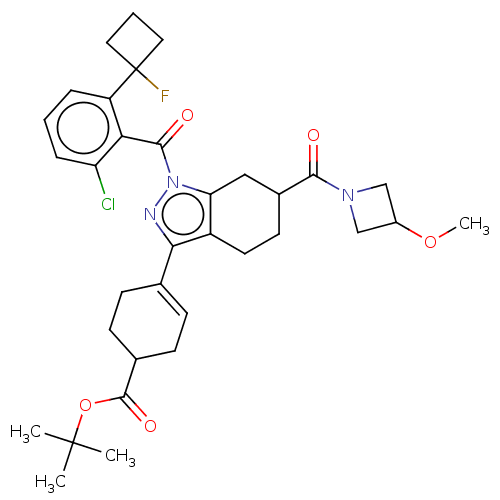
Affinity DataIC50: 1nMAssay Description:The compounds of the invention inhibit RORgammaT activity. Activation of RORgammaT activity can be measured using, e.g., biochemical TR-FRET assay. I...More data for this Ligand-Target Pair
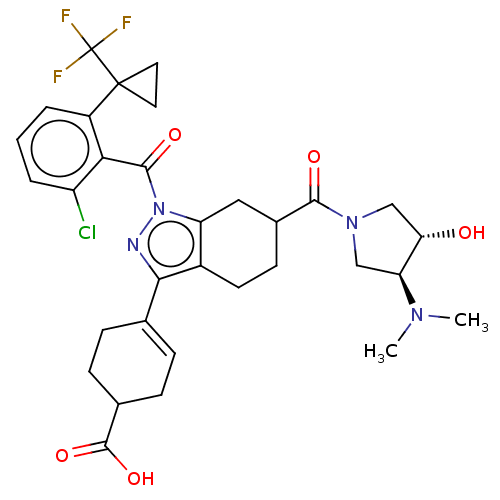
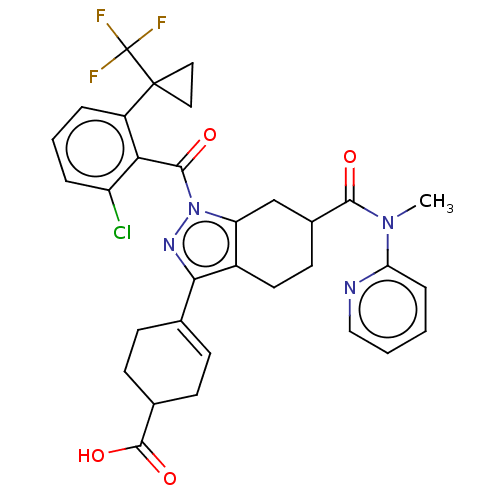
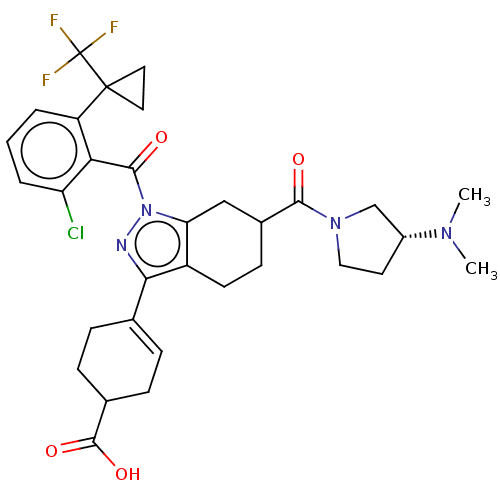
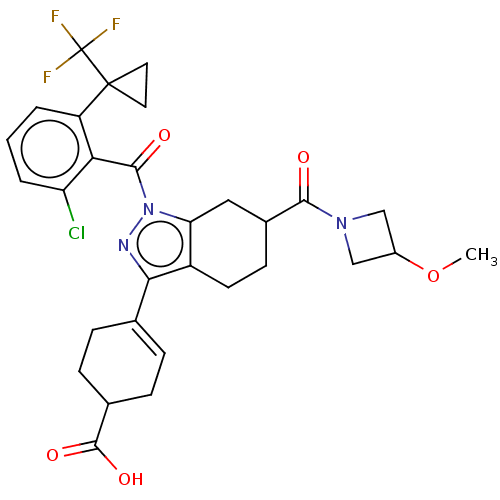
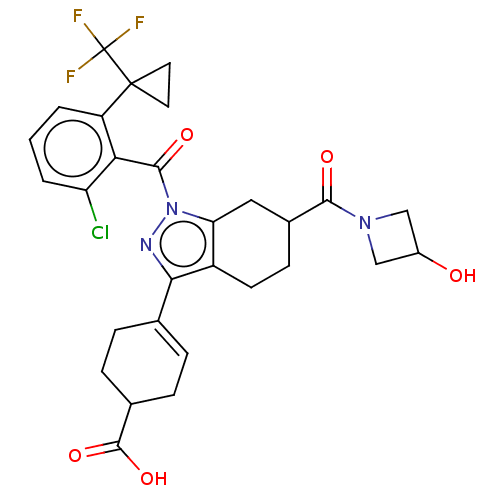
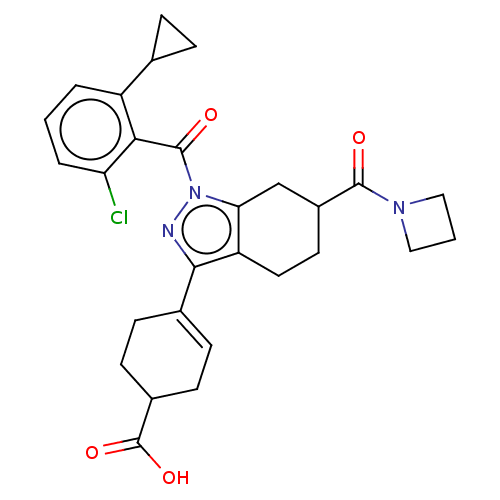
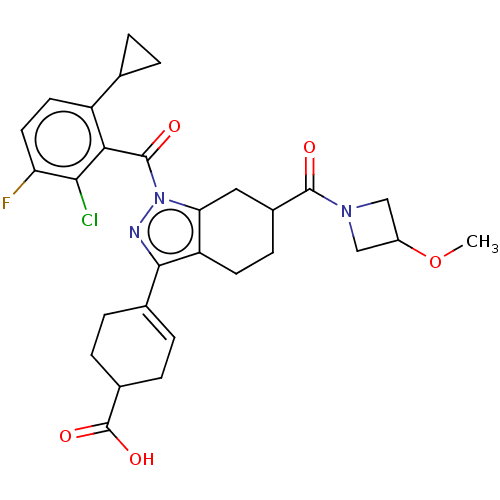
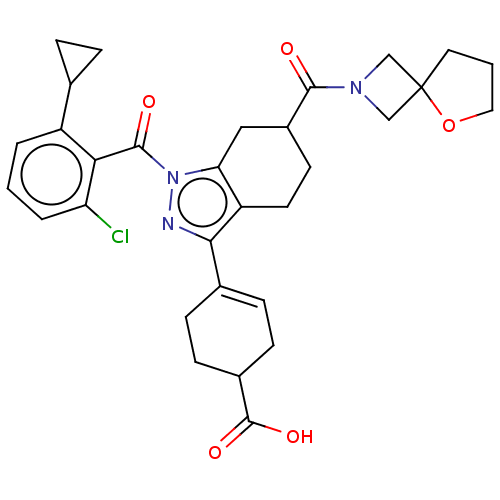
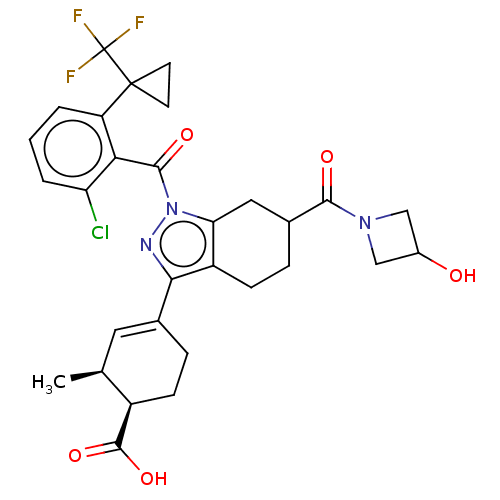
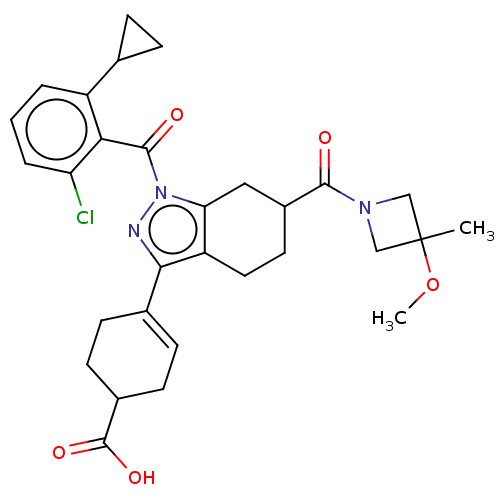
Affinity DataIC50: 1nMAssay Description:The compounds of the invention inhibit RORgammaT activity. Activation of RORgammaT activity can be measured using, e.g., biochemical TR-FRET assay. I...More data for this Ligand-Target Pair
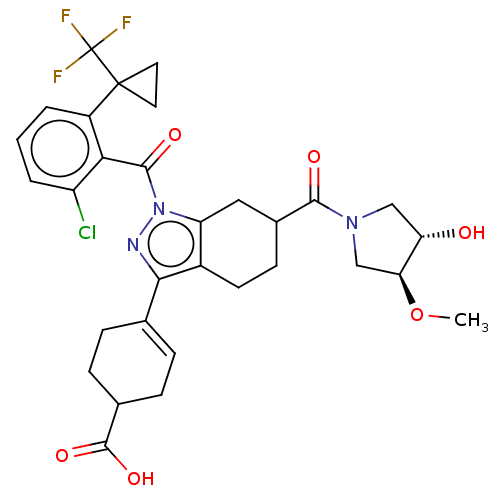
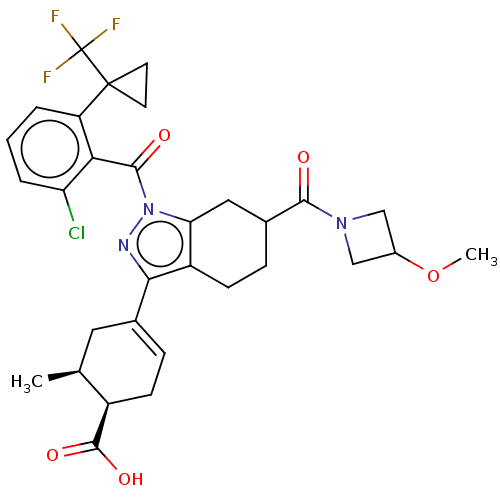
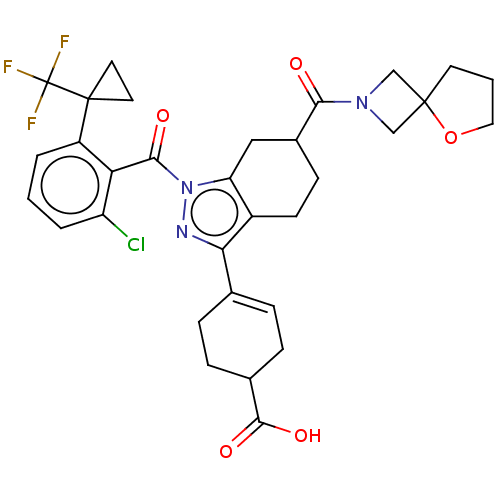
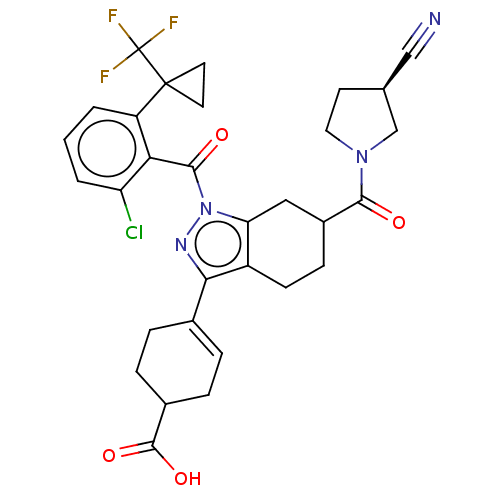
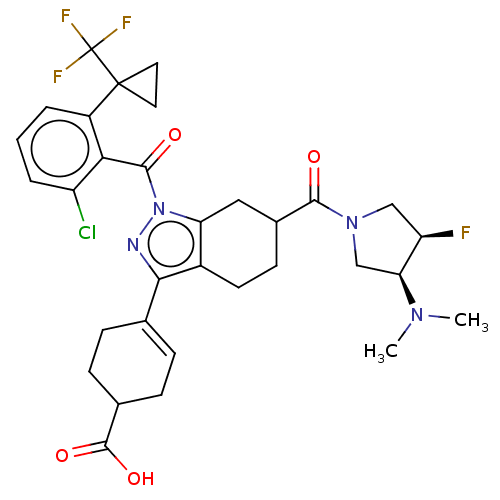
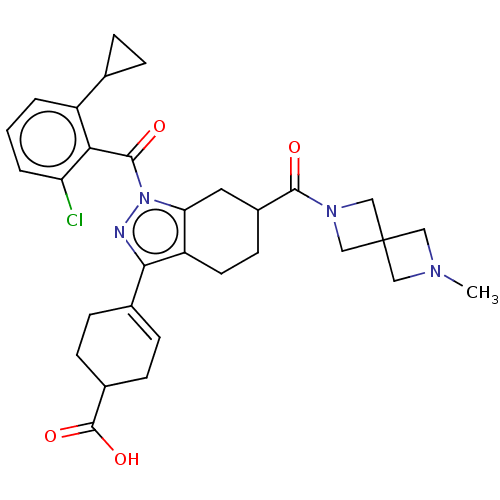
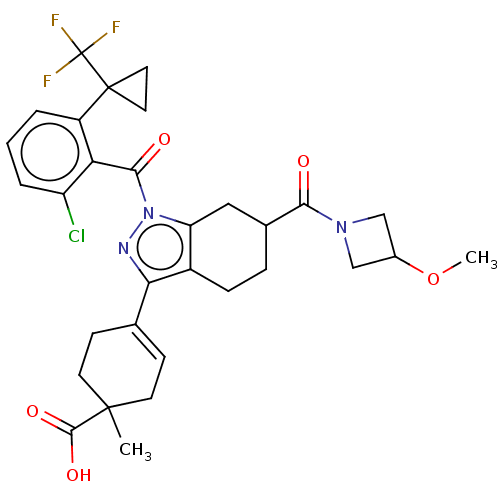
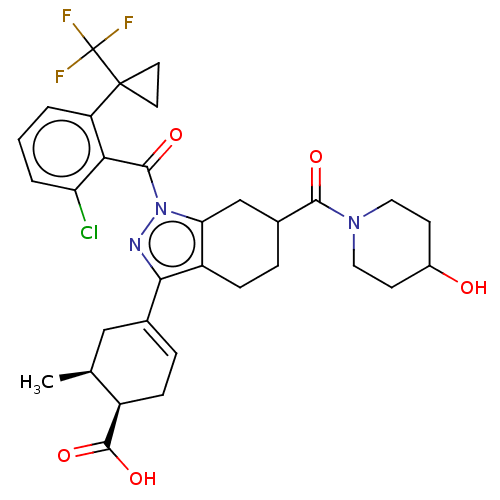
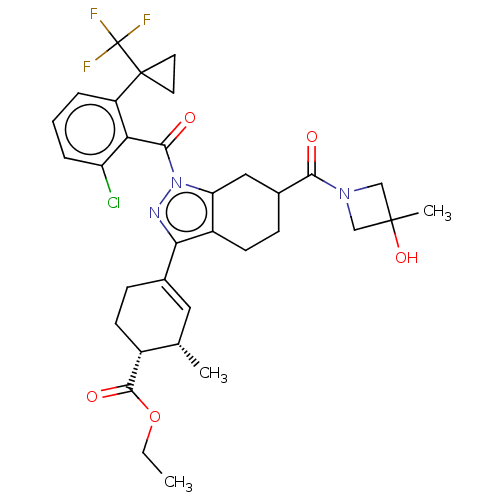
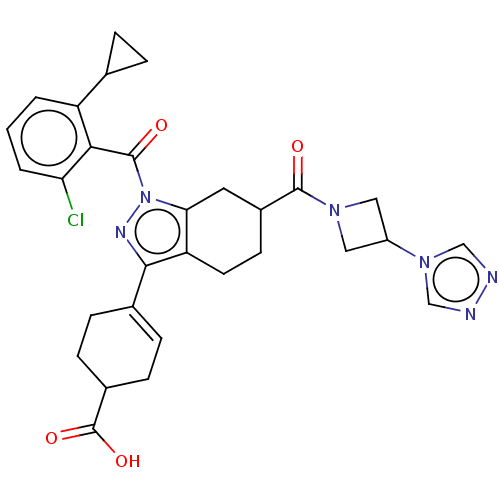
Affinity DataIC50: 1nMAssay Description:The compounds of the invention inhibit RORgammaT activity. Activation of RORgammaT activity can be measured using, e.g., biochemical TR-FRET assay. I...More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:The compounds of the invention inhibit RORgammaT activity. Activation of RORgammaT activity can be measured using, e.g., biochemical TR-FRET assay. I...More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:The compounds of the invention inhibit RORgammaT activity. Activation of RORgammaT activity can be measured using, e.g., biochemical TR-FRET assay. I...More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:The compounds of the invention inhibit RORgammaT activity. Activation of RORgammaT activity can be measured using, e.g., biochemical TR-FRET assay. I...More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:The compounds of the invention inhibit RORgammaT activity. Activation of RORgammaT activity can be measured using, e.g., biochemical TR-FRET assay. I...More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:The compounds of the invention inhibit RORgammaT activity. Activation of RORgammaT activity can be measured using, e.g., biochemical TR-FRET assay. I...More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:The compounds of the invention inhibit RORgammaT activity. Activation of RORgammaT activity can be measured using, e.g., biochemical TR-FRET assay. I...More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:The compounds of the invention inhibit RORgammaT activity. Activation of RORgammaT activity can be measured using, e.g., biochemical TR-FRET assay. I...More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:The compounds of the invention inhibit RORgammaT activity. Activation of RORgammaT activity can be measured using, e.g., biochemical TR-FRET assay. I...More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:The compounds of the invention inhibit RORgammaT activity. Activation of RORgammaT activity can be measured using, e.g., biochemical TR-FRET assay. I...More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:The compounds of the invention inhibit RORgammaT activity. Activation of RORgammaT activity can be measured using, e.g., biochemical TR-FRET assay. I...More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:The compounds of the invention inhibit RORgammaT activity. Activation of RORgammaT activity can be measured using, e.g., biochemical TR-FRET assay. I...More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:The compounds of the invention inhibit RORgammaT activity. Activation of RORgammaT activity can be measured using, e.g., biochemical TR-FRET assay. I...More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:The compounds of the invention inhibit RORgammaT activity. Activation of RORgammaT activity can be measured using, e.g., biochemical TR-FRET assay. I...More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:The compounds of the invention inhibit RORgammaT activity. Activation of RORgammaT activity can be measured using, e.g., biochemical TR-FRET assay. I...More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:The compounds of the invention inhibit RORgammaT activity. Activation of RORgammaT activity can be measured using, e.g., biochemical TR-FRET assay. I...More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:The compounds of the invention inhibit RORgammaT activity. Activation of RORgammaT activity can be measured using, e.g., biochemical TR-FRET assay. I...More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:The compounds of the invention inhibit RORgammaT activity. Activation of RORgammaT activity can be measured using, e.g., biochemical TR-FRET assay. I...More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:The compounds of the invention inhibit RORgammaT activity. Activation of RORgammaT activity can be measured using, e.g., biochemical TR-FRET assay. I...More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:The compounds of the invention inhibit RORgammaT activity. Activation of RORgammaT activity can be measured using, e.g., biochemical TR-FRET assay. I...More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:The compounds of the invention inhibit RORgammaT activity. Activation of RORgammaT activity can be measured using, e.g., biochemical TR-FRET assay. I...More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:The compounds of the invention inhibit RORgammaT activity. Activation of RORgammaT activity can be measured using, e.g., biochemical TR-FRET assay. I...More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:The compounds of the invention inhibit RORgammaT activity. Activation of RORgammaT activity can be measured using, e.g., biochemical TR-FRET assay. I...More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:The compounds of the invention inhibit RORgammaT activity. Activation of RORgammaT activity can be measured using, e.g., biochemical TR-FRET assay. I...More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:The compounds of the invention inhibit RORgammaT activity. Activation of RORgammaT activity can be measured using, e.g., biochemical TR-FRET assay. I...More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:The compounds of the invention inhibit RORgammaT activity. Activation of RORgammaT activity can be measured using, e.g., biochemical TR-FRET assay. I...More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:The compounds of the invention inhibit RORgammaT activity. Activation of RORgammaT activity can be measured using, e.g., biochemical TR-FRET assay. I...More data for this Ligand-Target Pair
Affinity DataIC50: 2nMAssay Description:The compounds of the invention inhibit RORgammaT activity. Activation of RORgammaT activity can be measured using, e.g., biochemical TR-FRET assay. I...More data for this Ligand-Target Pair
Affinity DataIC50: 2nMAssay Description:The compounds of the invention inhibit RORgammaT activity. Activation of RORgammaT activity can be measured using, e.g., biochemical TR-FRET assay. I...More data for this Ligand-Target Pair
Affinity DataIC50: 2nMAssay Description:The compounds of the invention inhibit RORgammaT activity. Activation of RORgammaT activity can be measured using, e.g., biochemical TR-FRET assay. I...More data for this Ligand-Target Pair
Affinity DataIC50: 2nMAssay Description:The compounds of the invention inhibit RORgammaT activity. Activation of RORgammaT activity can be measured using, e.g., biochemical TR-FRET assay. I...More data for this Ligand-Target Pair
Affinity DataIC50: 2nMAssay Description:The compounds of the invention inhibit RORgammaT activity. Activation of RORgammaT activity can be measured using, e.g., biochemical TR-FRET assay. I...More data for this Ligand-Target Pair
Affinity DataIC50: 2nMAssay Description:The compounds of the invention inhibit RORgammaT activity. Activation of RORgammaT activity can be measured using, e.g., biochemical TR-FRET assay. I...More data for this Ligand-Target Pair
Affinity DataIC50: 2nMAssay Description:The compounds of the invention inhibit RORgammaT activity. Activation of RORgammaT activity can be measured using, e.g., biochemical TR-FRET assay. I...More data for this Ligand-Target Pair
Affinity DataIC50: 2nMAssay Description:The compounds of the invention inhibit RORgammaT activity. Activation of RORgammaT activity can be measured using, e.g., biochemical TR-FRET assay. I...More data for this Ligand-Target Pair
Affinity DataIC50: 2nMAssay Description:The compounds of the invention inhibit RORgammaT activity. Activation of RORgammaT activity can be measured using, e.g., biochemical TR-FRET assay. I...More data for this Ligand-Target Pair
Affinity DataIC50: 2nMAssay Description:The compounds of the invention inhibit RORgammaT activity. Activation of RORgammaT activity can be measured using, e.g., biochemical TR-FRET assay. I...More data for this Ligand-Target Pair
Affinity DataIC50: 2nMAssay Description:The compounds of the invention inhibit RORgammaT activity. Activation of RORgammaT activity can be measured using, e.g., biochemical TR-FRET assay. I...More data for this Ligand-Target Pair
Affinity DataIC50: 2nMAssay Description:The compounds of the invention inhibit RORgammaT activity. Activation of RORgammaT activity can be measured using, e.g., biochemical TR-FRET assay. I...More data for this Ligand-Target Pair
Affinity DataIC50: 2nMAssay Description:The compounds of the invention inhibit RORgammaT activity. Activation of RORgammaT activity can be measured using, e.g., biochemical TR-FRET assay. I...More data for this Ligand-Target Pair
Affinity DataIC50: 2nMAssay Description:The compounds of the invention inhibit RORgammaT activity. Activation of RORgammaT activity can be measured using, e.g., biochemical TR-FRET assay. I...More data for this Ligand-Target Pair
Affinity DataIC50: 2nMAssay Description:The compounds of the invention inhibit RORgammaT activity. Activation of RORgammaT activity can be measured using, e.g., biochemical TR-FRET assay. I...More data for this Ligand-Target Pair
Affinity DataIC50: 2nMAssay Description:The compounds of the invention inhibit RORgammaT activity. Activation of RORgammaT activity can be measured using, e.g., biochemical TR-FRET assay. I...More data for this Ligand-Target Pair
Affinity DataIC50: 2nMAssay Description:The compounds of the invention inhibit RORgammaT activity. Activation of RORgammaT activity can be measured using, e.g., biochemical TR-FRET assay. I...More data for this Ligand-Target Pair
Affinity DataIC50: 2nMAssay Description:The compounds of the invention inhibit RORgammaT activity. Activation of RORgammaT activity can be measured using, e.g., biochemical TR-FRET assay. I...More data for this Ligand-Target Pair
Affinity DataIC50: 2nMAssay Description:The compounds of the invention inhibit RORgammaT activity. Activation of RORgammaT activity can be measured using, e.g., biochemical TR-FRET assay. I...More data for this Ligand-Target Pair
Affinity DataIC50: 2nMAssay Description:The compounds of the invention inhibit RORgammaT activity. Activation of RORgammaT activity can be measured using, e.g., biochemical TR-FRET assay. I...More data for this Ligand-Target Pair