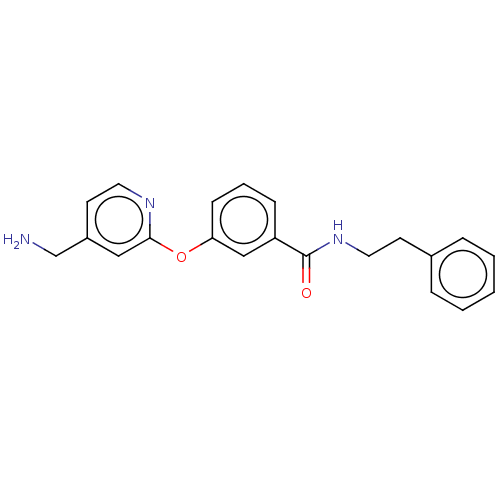
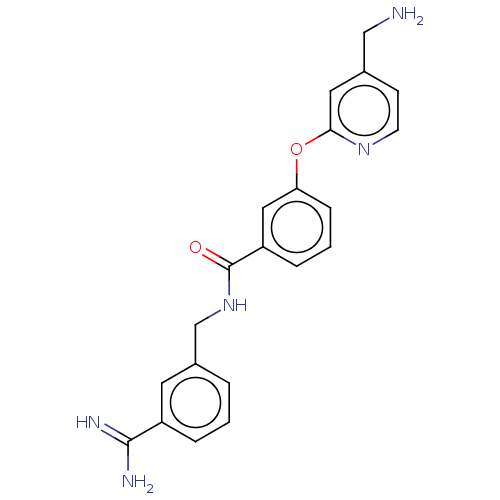
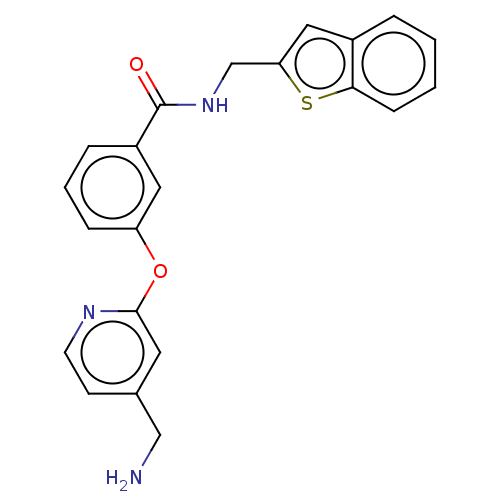
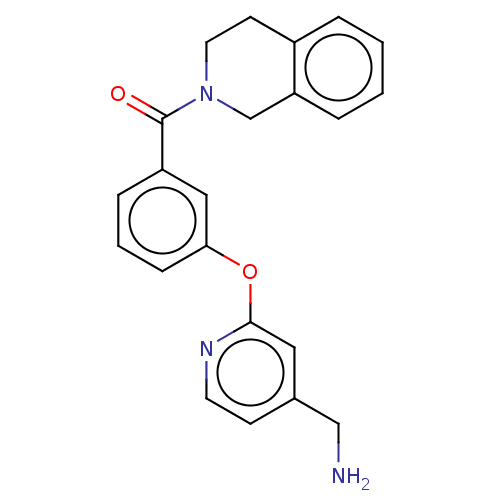
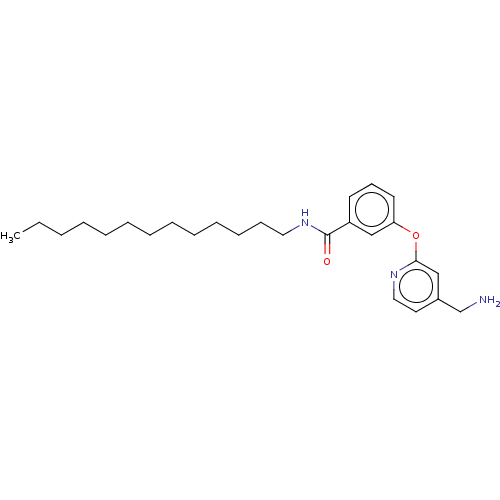
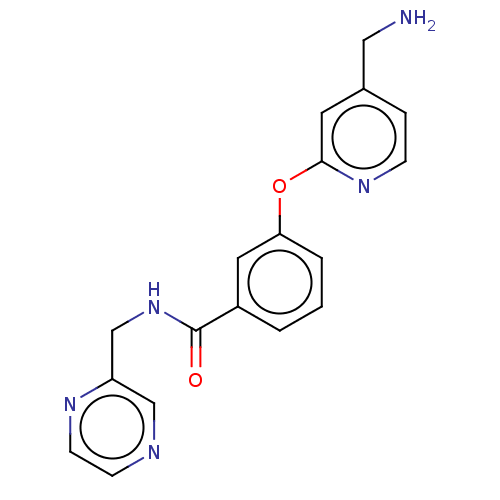
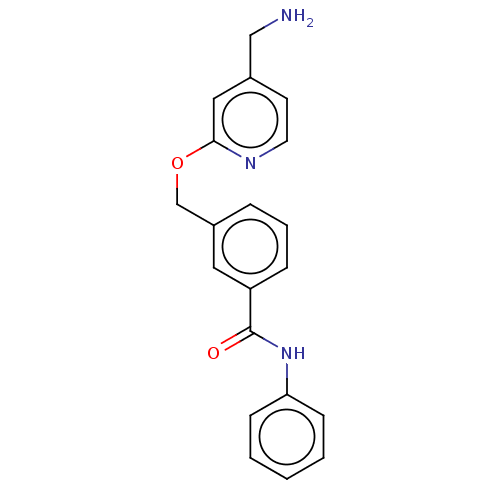
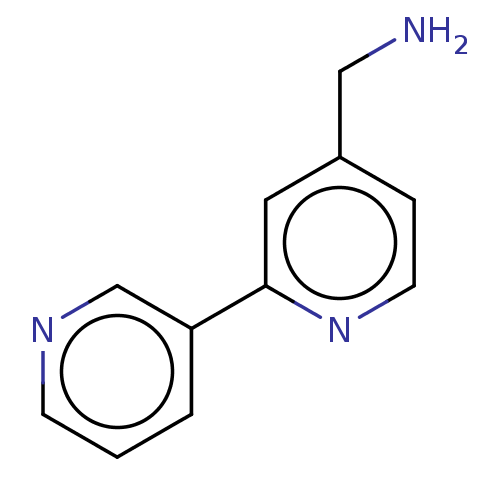
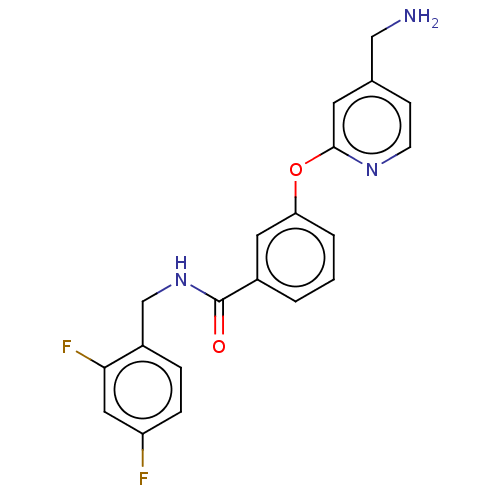
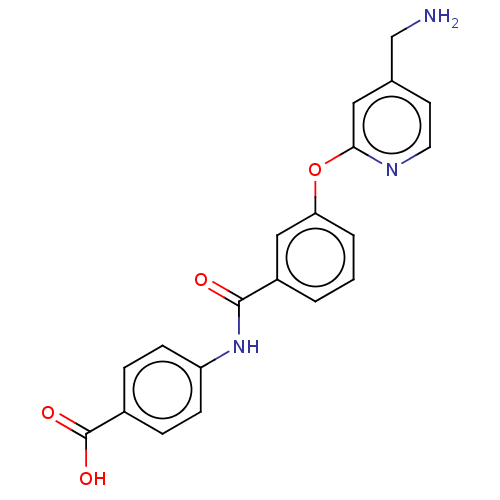
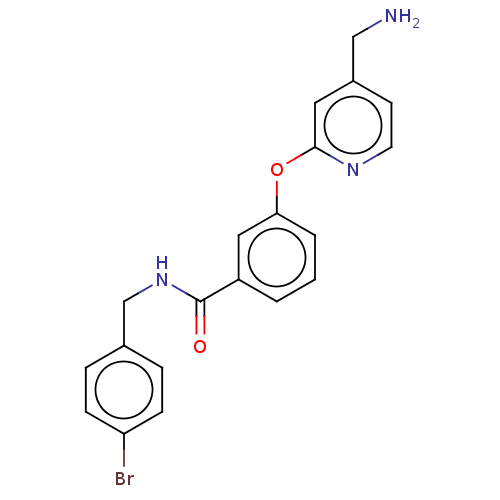
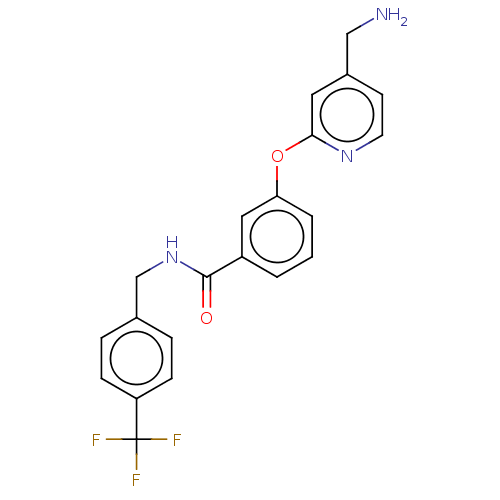
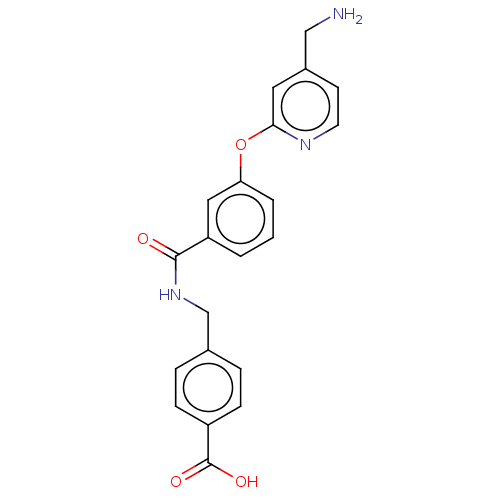
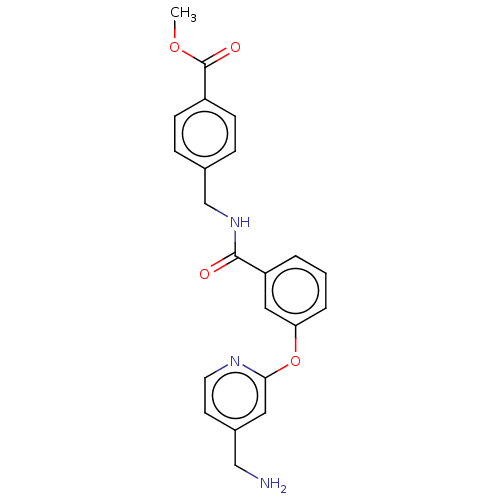
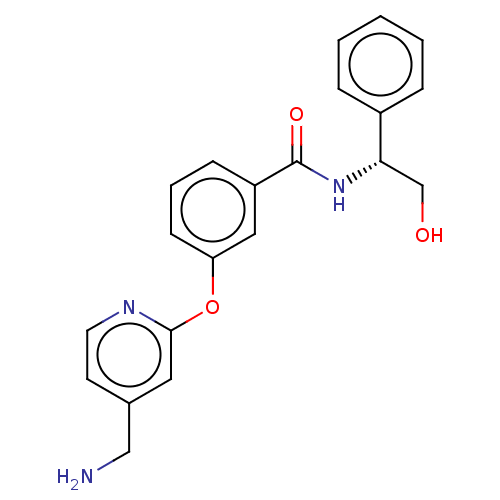
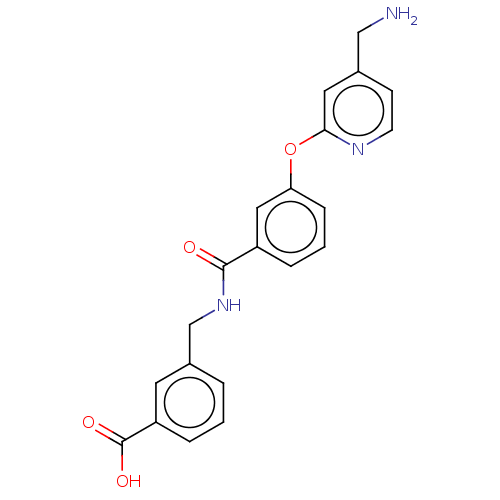
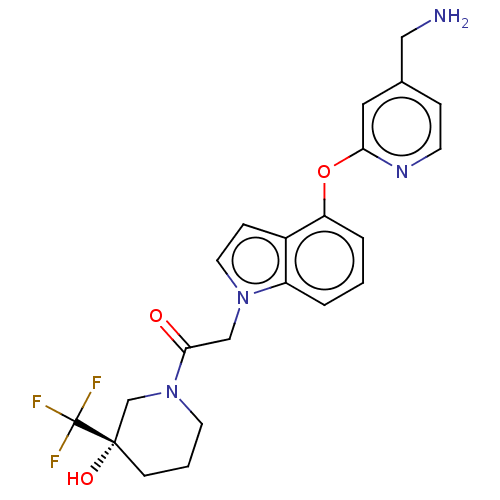
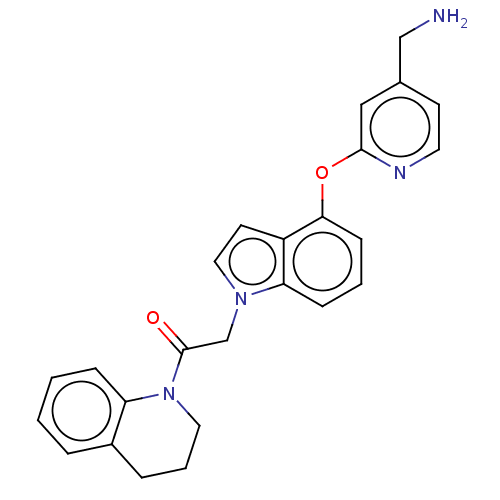
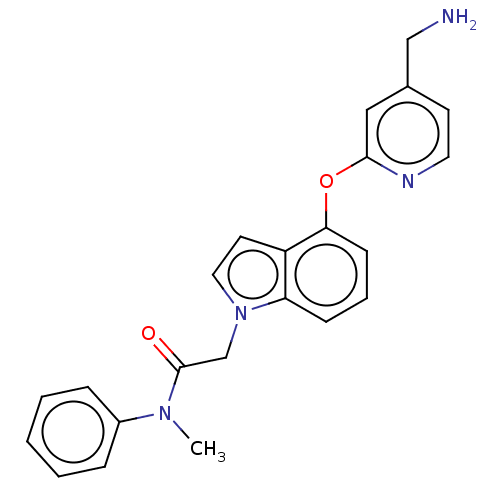
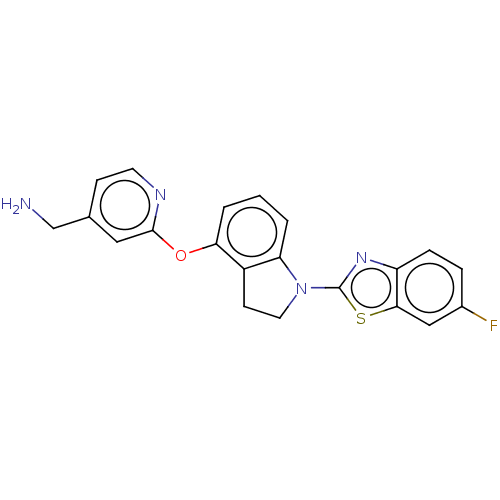
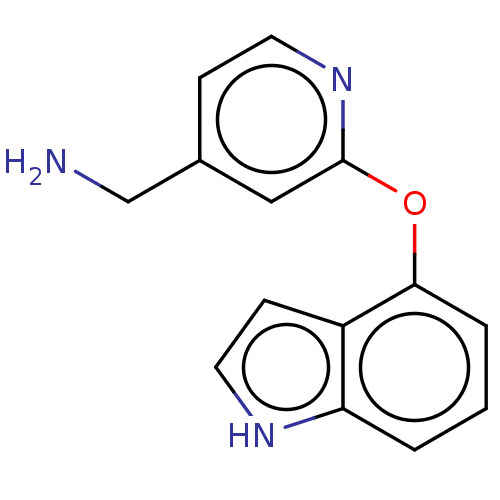
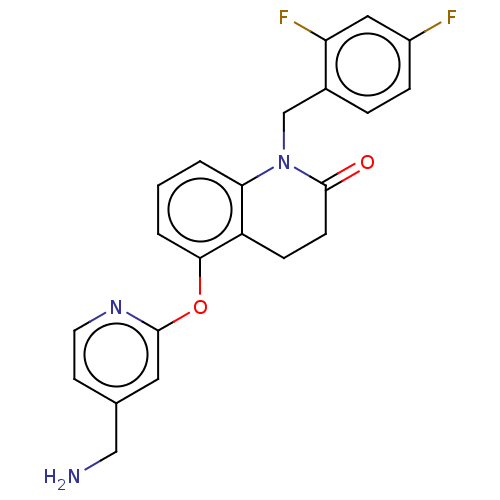
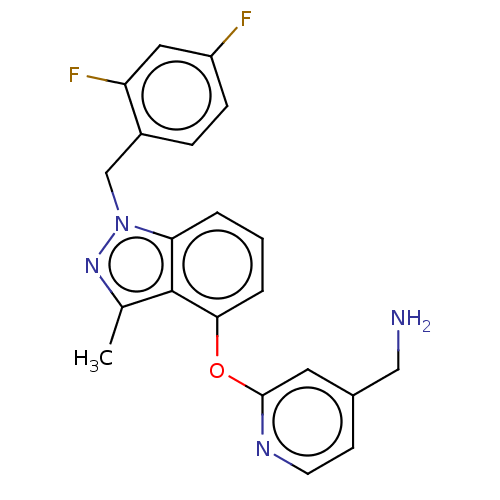
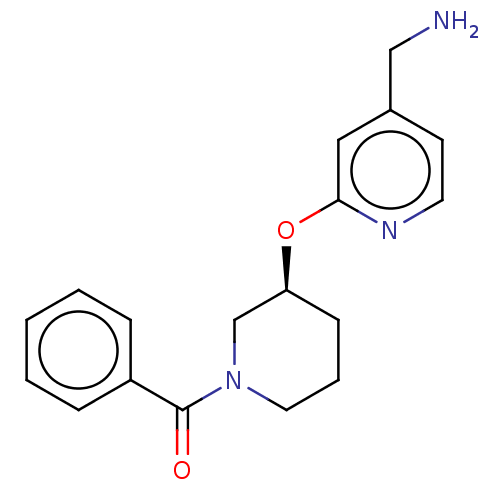
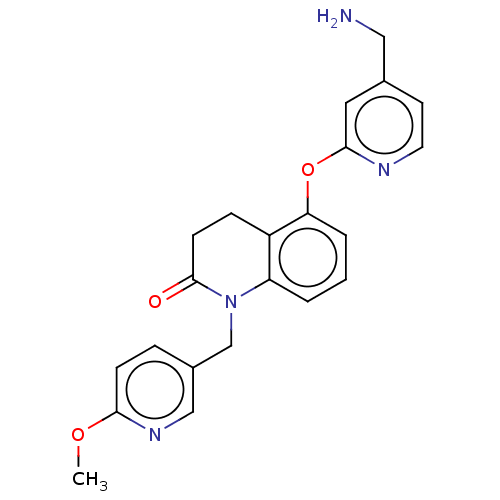
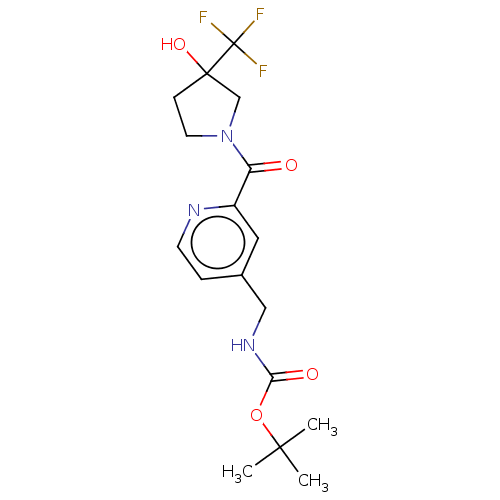
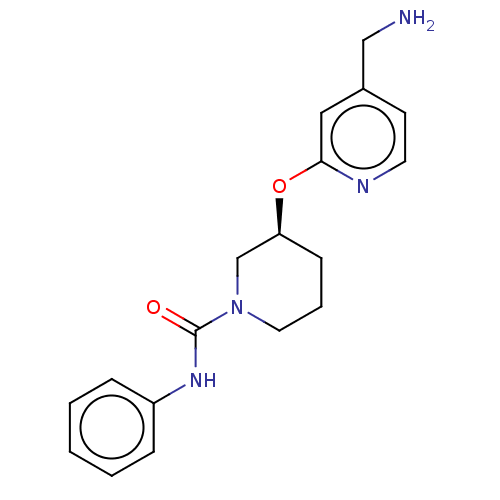
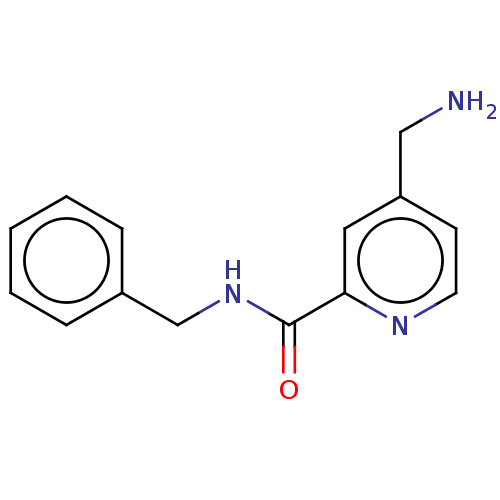
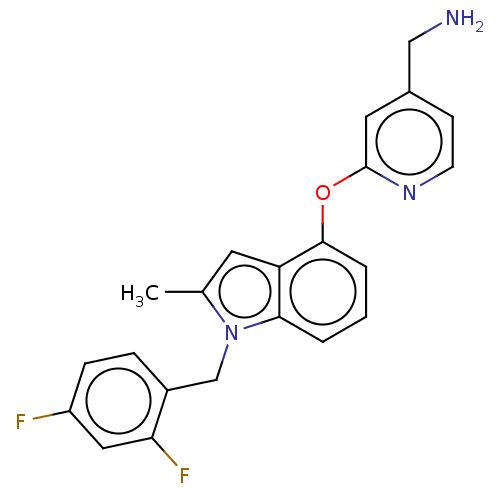
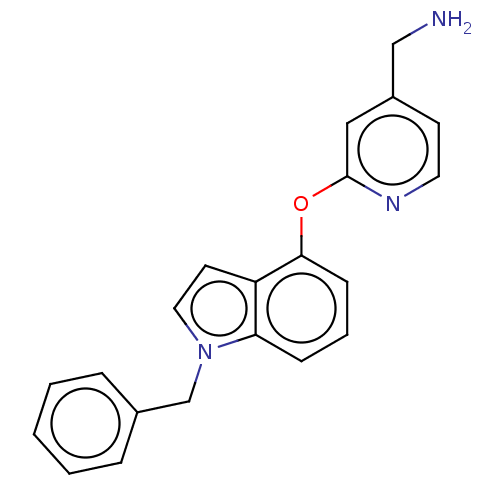
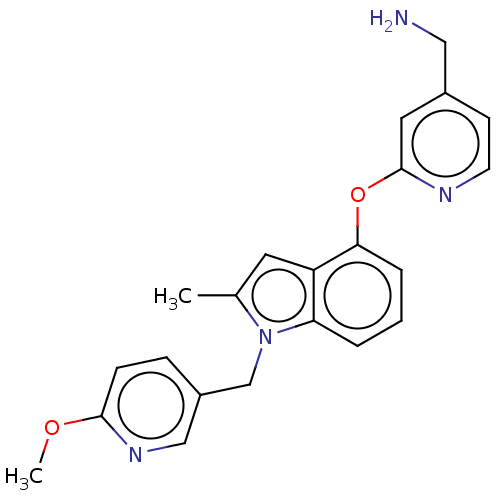
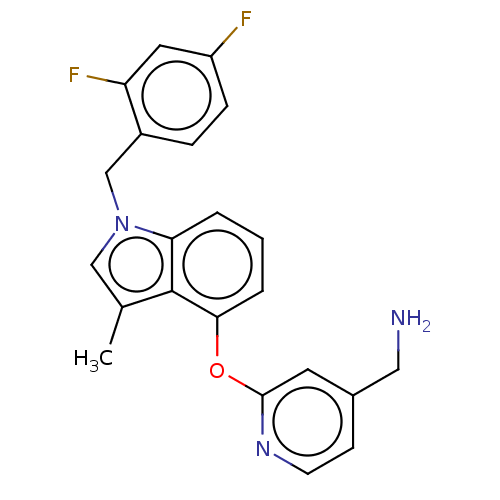
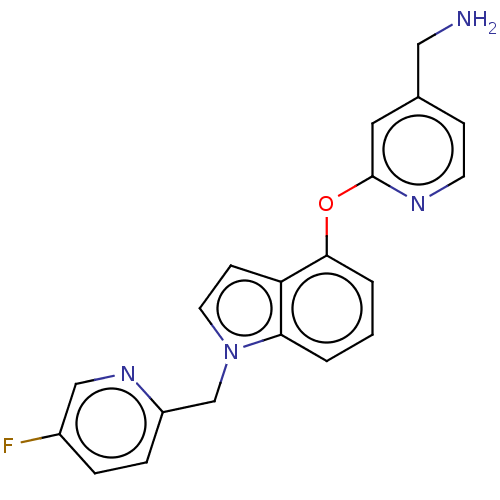
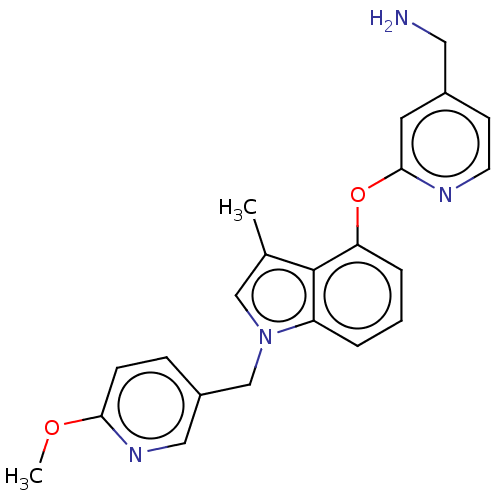
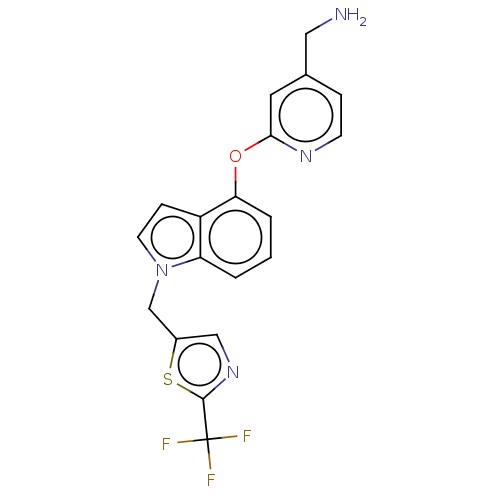
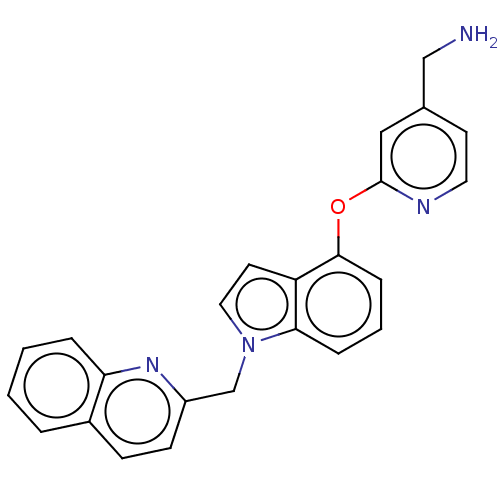
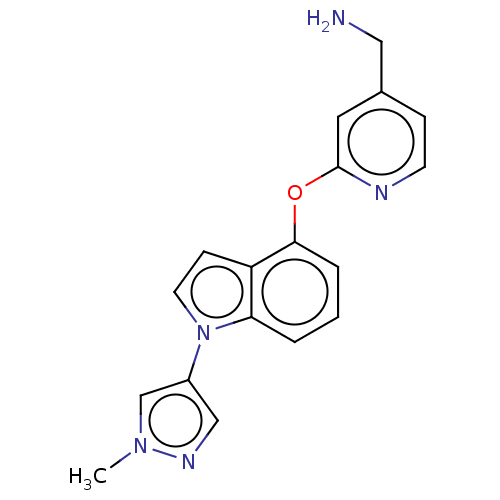
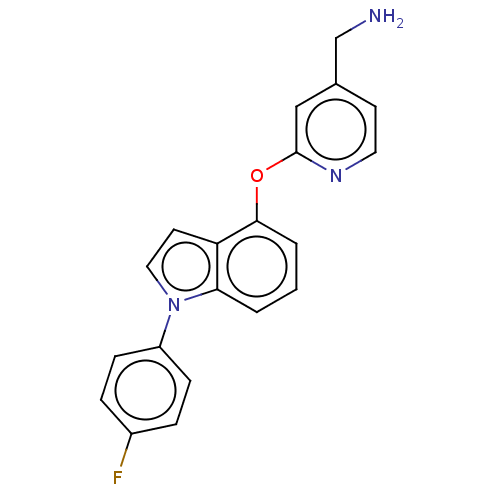
Report error Found 104 Enz. Inhib. hit(s) with all data for entry = 10654
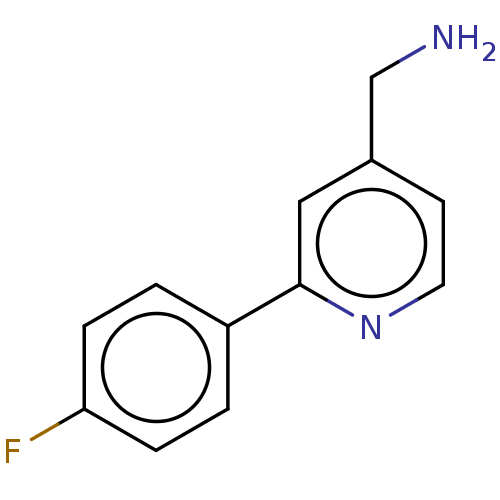
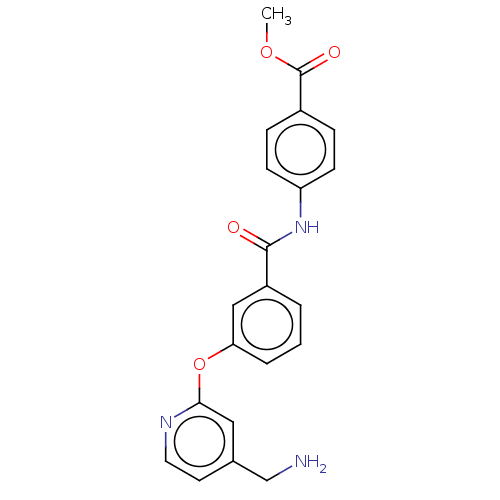
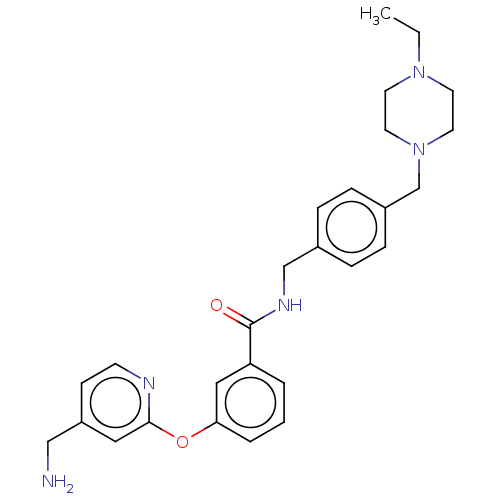
Affinity DataIC50: 300nMAssay Description:LOXL2 amine oxidase activity is evaluated by measuring Amplex Red fluorescence using 10-20× concentrated conditioned media from CHO cells stably expr...More data for this Ligand-Target Pair
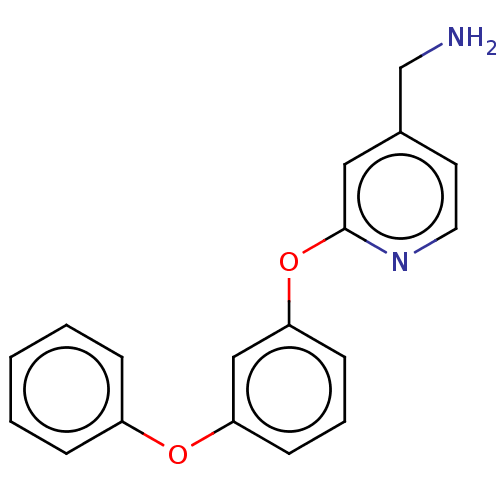
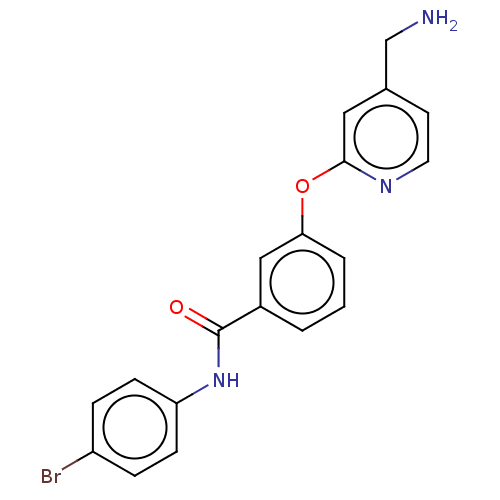
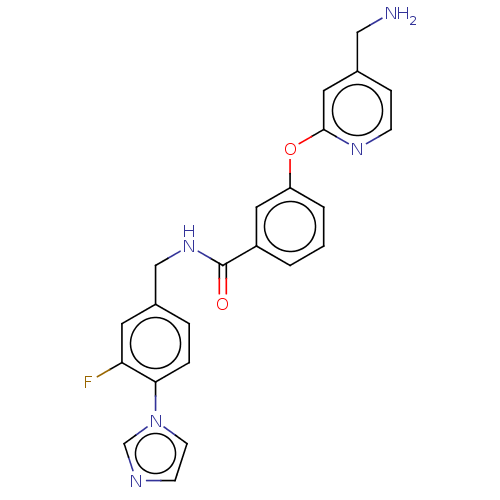
Affinity DataIC50: 300nMAssay Description:LOXL2 amine oxidase activity is evaluated by measuring Amplex Red fluorescence using 10-20× concentrated conditioned media from CHO cells stably expr...More data for this Ligand-Target Pair
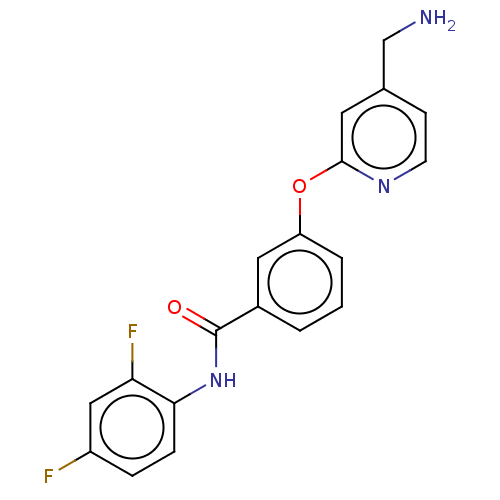
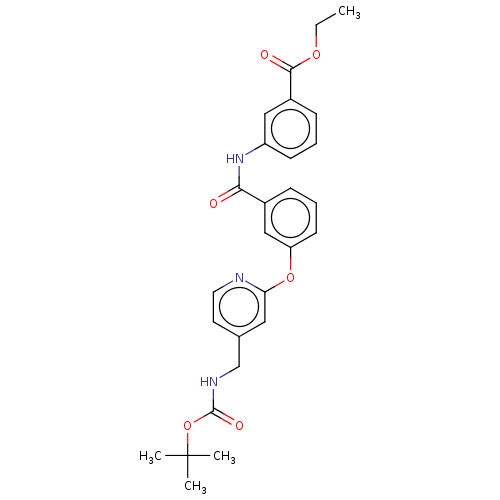
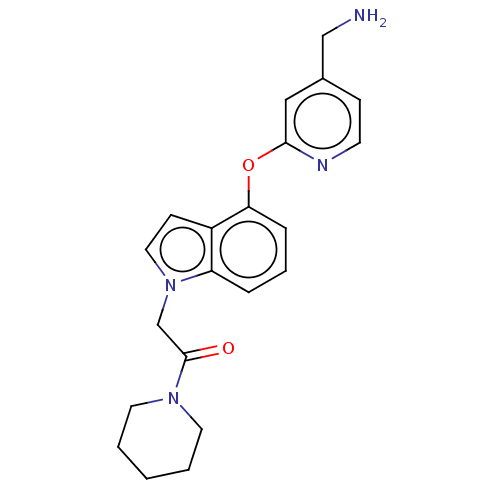
Affinity DataIC50: 300nMAssay Description:LOXL2 amine oxidase activity is evaluated by measuring Amplex Red fluorescence using 10-20× concentrated conditioned media from CHO cells stably expr...More data for this Ligand-Target Pair
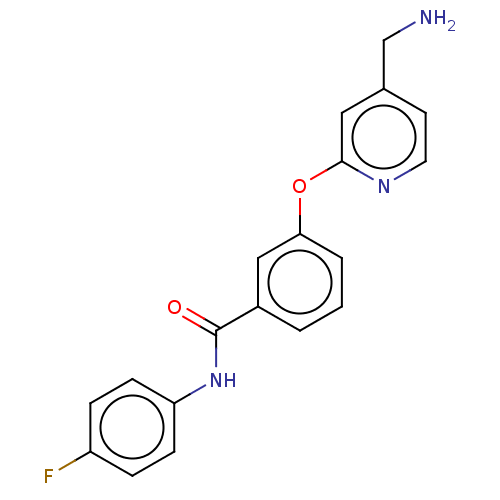
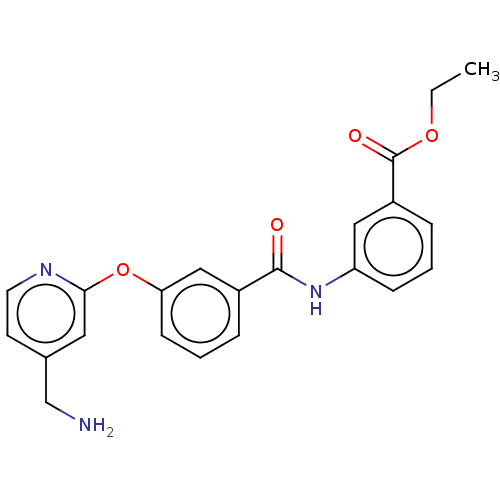
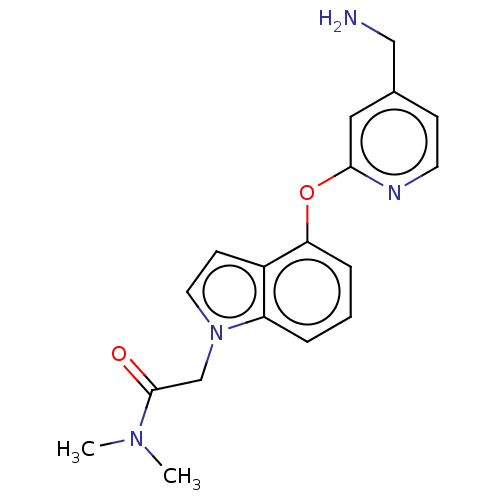
Affinity DataIC50: 300nMAssay Description:LOXL2 amine oxidase activity is evaluated by measuring Amplex Red fluorescence using 10-20× concentrated conditioned media from CHO cells stably expr...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:LOXL2 amine oxidase activity is evaluated by measuring Amplex Red fluorescence using 10-20× concentrated conditioned media from CHO cells stably expr...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:LOXL2 amine oxidase activity is evaluated by measuring Amplex Red fluorescence using 10-20× concentrated conditioned media from CHO cells stably expr...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:LOXL2 amine oxidase activity is evaluated by measuring Amplex Red fluorescence using 10-20× concentrated conditioned media from CHO cells stably expr...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:LOXL2 amine oxidase activity is evaluated by measuring Amplex Red fluorescence using 10-20× concentrated conditioned media from CHO cells stably expr...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:LOXL2 amine oxidase activity is evaluated by measuring Amplex Red fluorescence using 10-20× concentrated conditioned media from CHO cells stably expr...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:LOXL2 amine oxidase activity is evaluated by measuring Amplex Red fluorescence using 10-20× concentrated conditioned media from CHO cells stably expr...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:LOXL2 amine oxidase activity is evaluated by measuring Amplex Red fluorescence using 10-20× concentrated conditioned media from CHO cells stably expr...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:LOXL2 amine oxidase activity is evaluated by measuring Amplex Red fluorescence using 10-20× concentrated conditioned media from CHO cells stably expr...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:LOXL2 amine oxidase activity is evaluated by measuring Amplex Red fluorescence using 10-20× concentrated conditioned media from CHO cells stably expr...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:LOXL2 amine oxidase activity is evaluated by measuring Amplex Red fluorescence using 10-20× concentrated conditioned media from CHO cells stably expr...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:LOXL2 amine oxidase activity is evaluated by measuring Amplex Red fluorescence using 10-20× concentrated conditioned media from CHO cells stably expr...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:LOXL2 amine oxidase activity is evaluated by measuring Amplex Red fluorescence using 10-20× concentrated conditioned media from CHO cells stably expr...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:LOXL2 amine oxidase activity is evaluated by measuring Amplex Red fluorescence using 10-20× concentrated conditioned media from CHO cells stably expr...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:LOXL2 amine oxidase activity is evaluated by measuring Amplex Red fluorescence using 10-20× concentrated conditioned media from CHO cells stably expr...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:LOXL2 amine oxidase activity is evaluated by measuring Amplex Red fluorescence using 10-20× concentrated conditioned media from CHO cells stably expr...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:LOXL2 amine oxidase activity is evaluated by measuring Amplex Red fluorescence using 10-20× concentrated conditioned media from CHO cells stably expr...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:LOXL2 amine oxidase activity is evaluated by measuring Amplex Red fluorescence using 10-20× concentrated conditioned media from CHO cells stably expr...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:LOXL2 amine oxidase activity is evaluated by measuring Amplex Red fluorescence using 10-20× concentrated conditioned media from CHO cells stably expr...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:LOXL2 amine oxidase activity is evaluated by measuring Amplex Red fluorescence using 10-20× concentrated conditioned media from CHO cells stably expr...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:LOXL2 amine oxidase activity is evaluated by measuring Amplex Red fluorescence using 10-20× concentrated conditioned media from CHO cells stably expr...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:LOXL2 amine oxidase activity is evaluated by measuring Amplex Red fluorescence using 10-20× concentrated conditioned media from CHO cells stably expr...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:LOXL2 amine oxidase activity is evaluated by measuring Amplex Red fluorescence using 10-20× concentrated conditioned media from CHO cells stably expr...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:LOXL2 amine oxidase activity is evaluated by measuring Amplex Red fluorescence using 10-20× concentrated conditioned media from CHO cells stably expr...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:LOXL2 amine oxidase activity is evaluated by measuring Amplex Red fluorescence using 10-20× concentrated conditioned media from CHO cells stably expr...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:LOXL2 amine oxidase activity is evaluated by measuring Amplex Red fluorescence using 10-20× concentrated conditioned media from CHO cells stably expr...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:LOXL2 amine oxidase activity is evaluated by measuring Amplex Red fluorescence using 10-20× concentrated conditioned media from CHO cells stably expr...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:LOXL2 amine oxidase activity is evaluated by measuring Amplex Red fluorescence using 10-20× concentrated conditioned media from CHO cells stably expr...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:LOXL2 amine oxidase activity is evaluated by measuring Amplex Red fluorescence using 10-20× concentrated conditioned media from CHO cells stably expr...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:LOXL2 amine oxidase activity is evaluated by measuring Amplex Red fluorescence using 10-20× concentrated conditioned media from CHO cells stably expr...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:LOXL2 amine oxidase activity is evaluated by measuring Amplex Red fluorescence using 10-20× concentrated conditioned media from CHO cells stably expr...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:LOXL2 amine oxidase activity is evaluated by measuring Amplex Red fluorescence using 10-20× concentrated conditioned media from CHO cells stably expr...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:LOXL2 amine oxidase activity is evaluated by measuring Amplex Red fluorescence using 10-20× concentrated conditioned media from CHO cells stably expr...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:LOXL2 amine oxidase activity is evaluated by measuring Amplex Red fluorescence using 10-20× concentrated conditioned media from CHO cells stably expr...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:LOXL2 amine oxidase activity is evaluated by measuring Amplex Red fluorescence using 10-20× concentrated conditioned media from CHO cells stably expr...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:LOXL2 amine oxidase activity is evaluated by measuring Amplex Red fluorescence using 10-20× concentrated conditioned media from CHO cells stably expr...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:LOXL2 amine oxidase activity is evaluated by measuring Amplex Red fluorescence using 10-20× concentrated conditioned media from CHO cells stably expr...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:LOXL2 amine oxidase activity is evaluated by measuring Amplex Red fluorescence using 10-20× concentrated conditioned media from CHO cells stably expr...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:LOXL2 amine oxidase activity is evaluated by measuring Amplex Red fluorescence using 10-20× concentrated conditioned media from CHO cells stably expr...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:LOXL2 amine oxidase activity is evaluated by measuring Amplex Red fluorescence using 10-20× concentrated conditioned media from CHO cells stably expr...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:LOXL2 amine oxidase activity is evaluated by measuring Amplex Red fluorescence using 10-20× concentrated conditioned media from CHO cells stably expr...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:LOXL2 amine oxidase activity is evaluated by measuring Amplex Red fluorescence using 10-20× concentrated conditioned media from CHO cells stably expr...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:LOXL2 amine oxidase activity is evaluated by measuring Amplex Red fluorescence using 10-20× concentrated conditioned media from CHO cells stably expr...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:LOXL2 amine oxidase activity is evaluated by measuring Amplex Red fluorescence using 10-20× concentrated conditioned media from CHO cells stably expr...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:LOXL2 amine oxidase activity is evaluated by measuring Amplex Red fluorescence using 10-20× concentrated conditioned media from CHO cells stably expr...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:LOXL2 amine oxidase activity is evaluated by measuring Amplex Red fluorescence using 10-20× concentrated conditioned media from CHO cells stably expr...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:LOXL2 amine oxidase activity is evaluated by measuring Amplex Red fluorescence using 10-20× concentrated conditioned media from CHO cells stably expr...More data for this Ligand-Target Pair