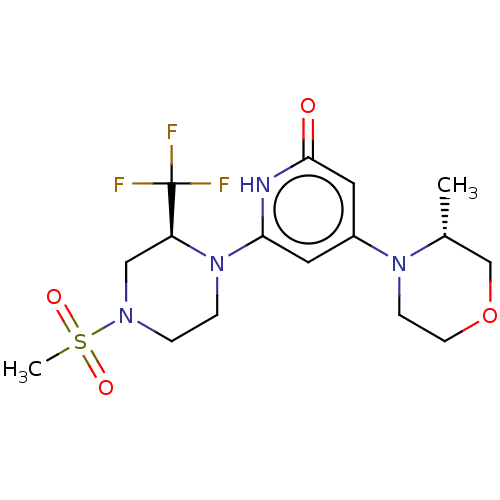
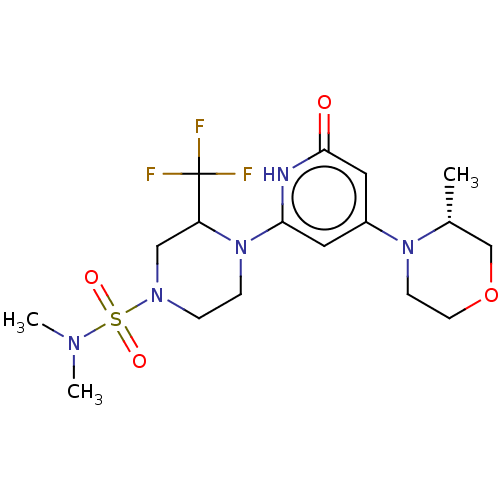
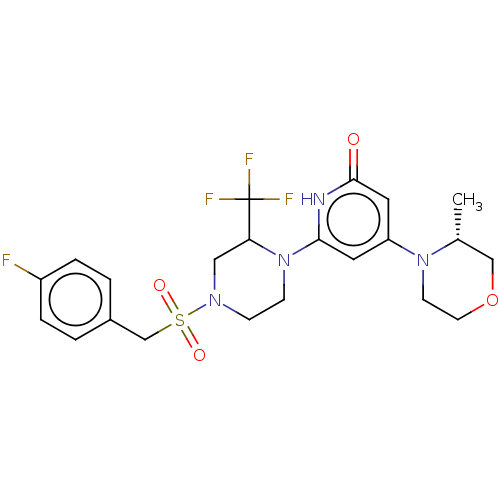
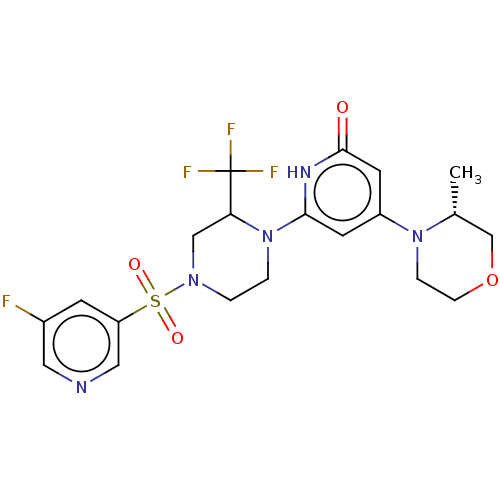
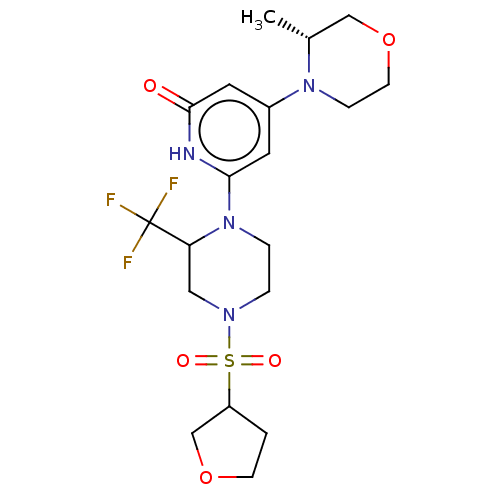
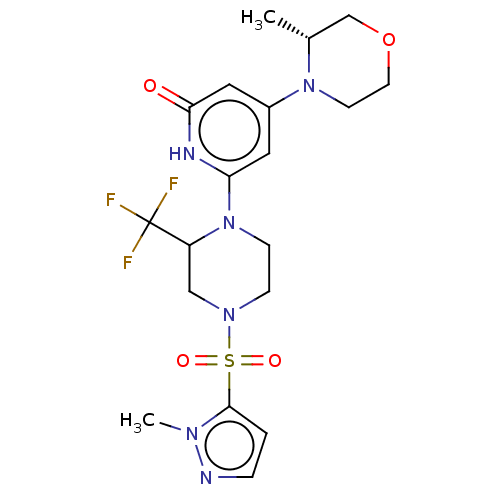
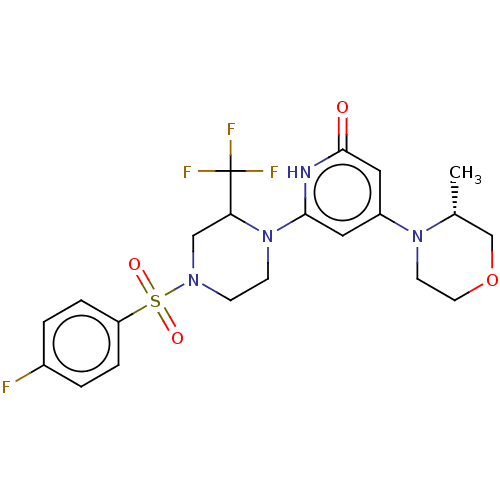
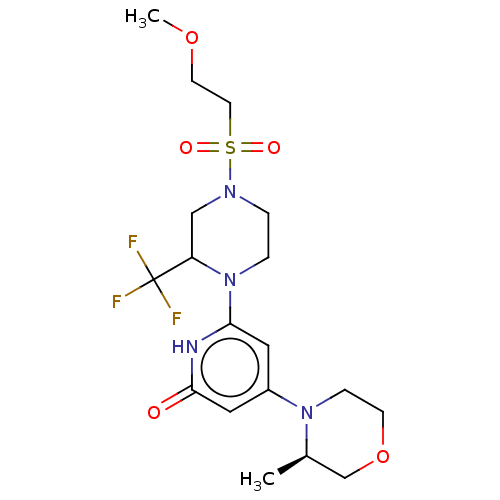
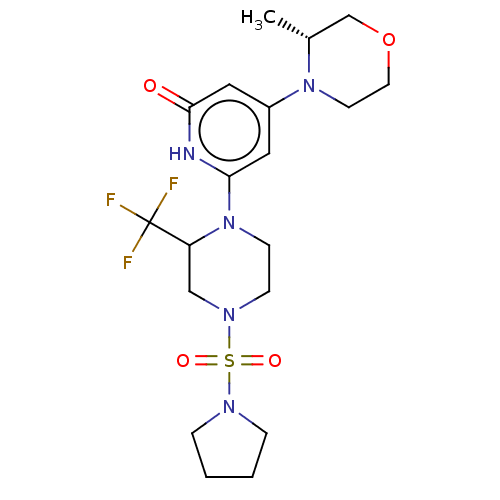
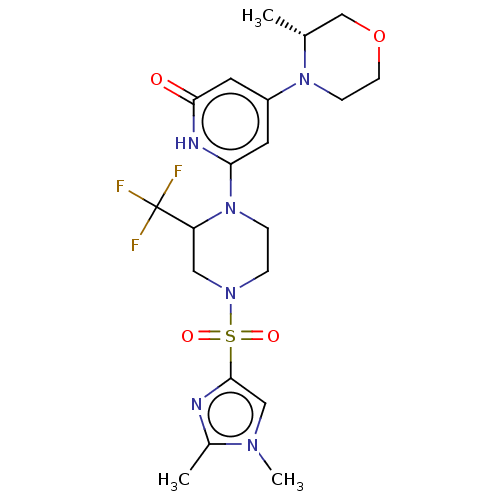
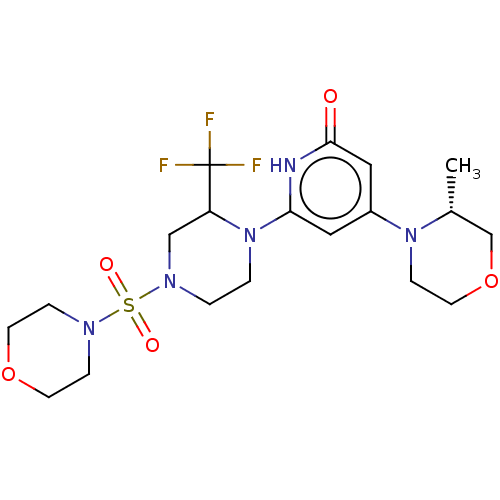
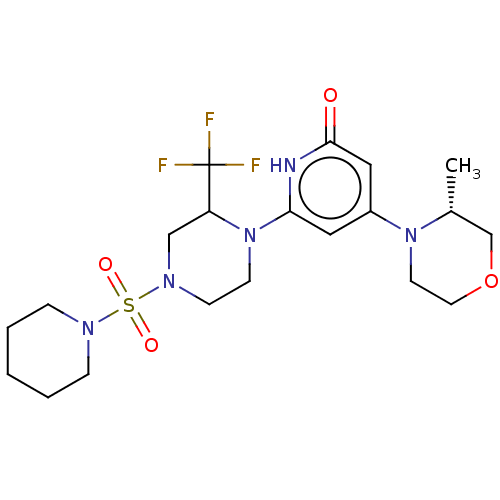
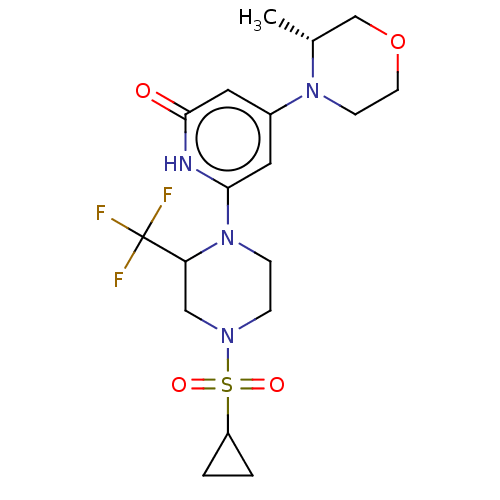
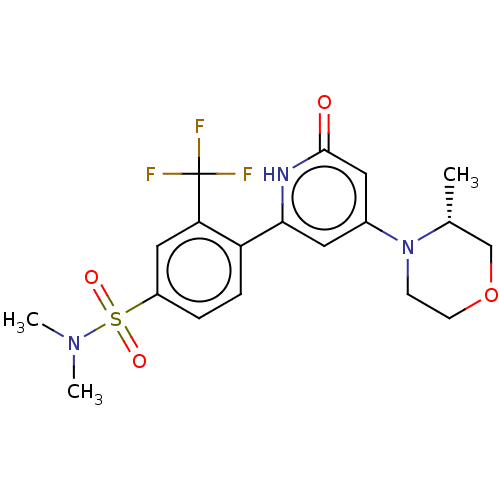
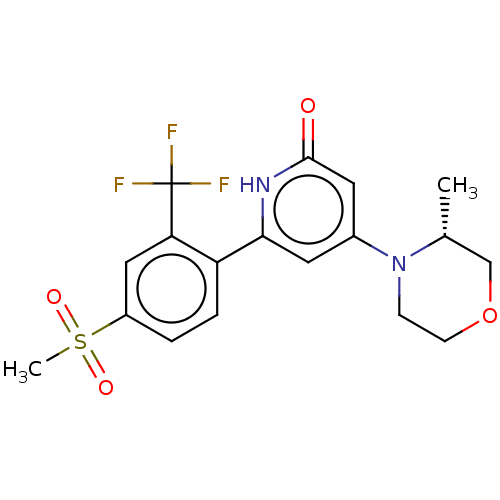
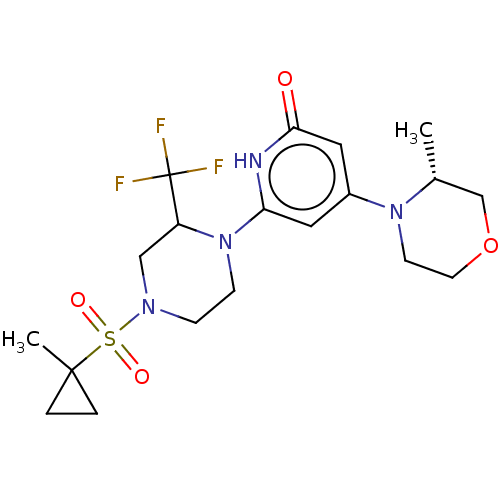
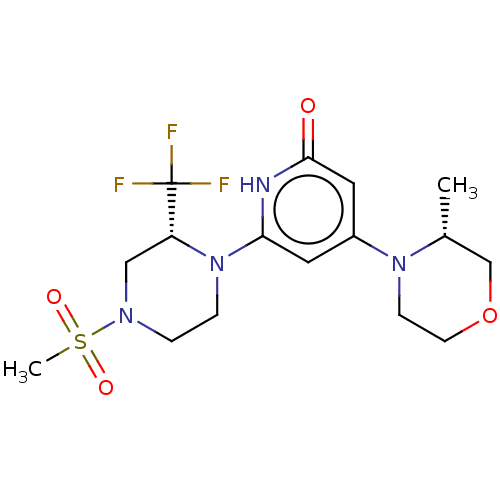
Report error Found 17 Enz. Inhib. hit(s) with all data for entry = 11100
Affinity DataIC50: 5nMAssay Description:Dilution series of compounds of the invention were prepared in DMSO at 100 times the final assay concentration (n1=n0/3 in 10 points). The compounds ...More data for this Ligand-Target Pair
Affinity DataIC50: 5nMAssay Description:Dilution series of compounds of the invention were prepared in DMSO at 100 times the final assay concentration (n1=n0/3 in 10 points). The compounds ...More data for this Ligand-Target Pair
Affinity DataIC50: 5nMAssay Description:Dilution series of compounds of the invention were prepared in DMSO at 100 times the final assay concentration (n1=n0/3 in 10 points). The compounds ...More data for this Ligand-Target Pair
Affinity DataIC50: 5nMAssay Description:Dilution series of compounds of the invention were prepared in DMSO at 100 times the final assay concentration (n1=n0/3 in 10 points). The compounds ...More data for this Ligand-Target Pair
Affinity DataIC50: 5nMAssay Description:Dilution series of compounds of the invention were prepared in DMSO at 100 times the final assay concentration (n1=n0/3 in 10 points). The compounds ...More data for this Ligand-Target Pair
Affinity DataIC50: 5nMAssay Description:Dilution series of compounds of the invention were prepared in DMSO at 100 times the final assay concentration (n1=n0/3 in 10 points). The compounds ...More data for this Ligand-Target Pair
Affinity DataIC50: 5nMAssay Description:Dilution series of compounds of the invention were prepared in DMSO at 100 times the final assay concentration (n1=n0/3 in 10 points). The compounds ...More data for this Ligand-Target Pair
Affinity DataIC50: 5nMAssay Description:Dilution series of compounds of the invention were prepared in DMSO at 100 times the final assay concentration (n1=n0/3 in 10 points). The compounds ...More data for this Ligand-Target Pair
Affinity DataIC50: 5nMAssay Description:Dilution series of compounds of the invention were prepared in DMSO at 100 times the final assay concentration (n1=n0/3 in 10 points). The compounds ...More data for this Ligand-Target Pair
Affinity DataIC50: 5nMAssay Description:Dilution series of compounds of the invention were prepared in DMSO at 100 times the final assay concentration (n1=n0/3 in 10 points). The compounds ...More data for this Ligand-Target Pair
Affinity DataIC50: 5nMAssay Description:Dilution series of compounds of the invention were prepared in DMSO at 100 times the final assay concentration (n1=n0/3 in 10 points). The compounds ...More data for this Ligand-Target Pair
Affinity DataIC50: 5nMAssay Description:Dilution series of compounds of the invention were prepared in DMSO at 100 times the final assay concentration (n1=n0/3 in 10 points). The compounds ...More data for this Ligand-Target Pair
Affinity DataIC50: 5nMAssay Description:Dilution series of compounds of the invention were prepared in DMSO at 100 times the final assay concentration (n1=n0/3 in 10 points). The compounds ...More data for this Ligand-Target Pair
Affinity DataIC50: 5nMAssay Description:Dilution series of compounds of the invention were prepared in DMSO at 100 times the final assay concentration (n1=n0/3 in 10 points). The compounds ...More data for this Ligand-Target Pair
Affinity DataIC50: 5nMAssay Description:Dilution series of compounds of the invention were prepared in DMSO at 100 times the final assay concentration (n1=n0/3 in 10 points). The compounds ...More data for this Ligand-Target Pair
Affinity DataIC50: 5nMAssay Description:Dilution series of compounds of the invention were prepared in DMSO at 100 times the final assay concentration (n1=n0/3 in 10 points). The compounds ...More data for this Ligand-Target Pair
Affinity DataIC50: 15nMAssay Description:Dilution series of compounds of the invention were prepared in DMSO at 100 times the final assay concentration (n1=n0/3 in 10 points). The compounds ...More data for this Ligand-Target Pair