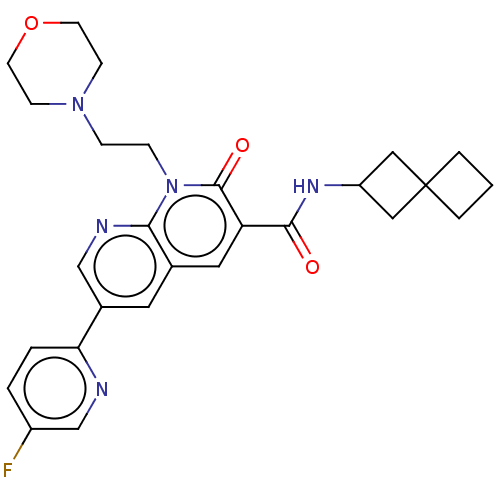
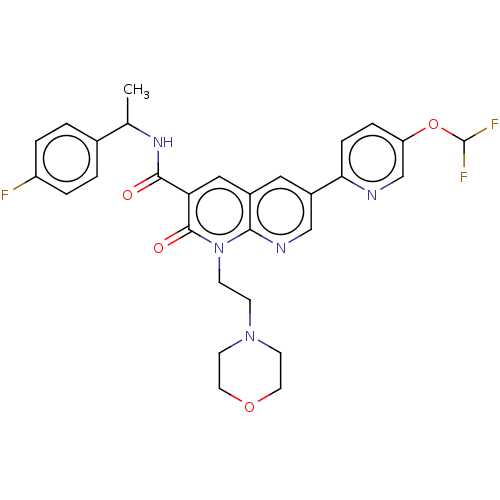
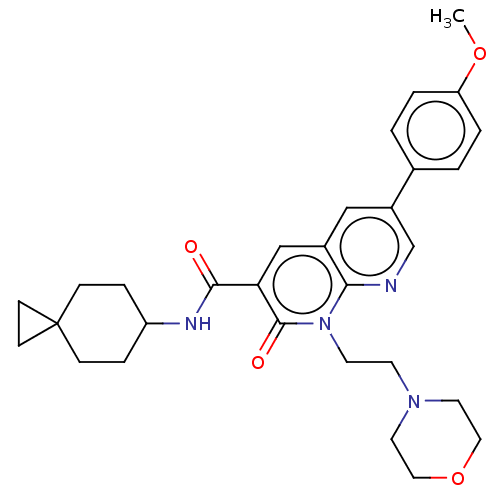
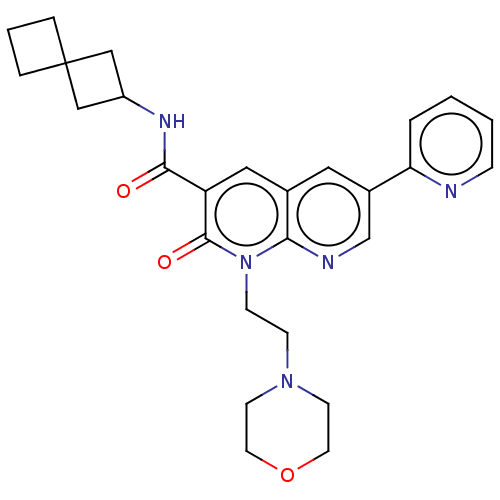
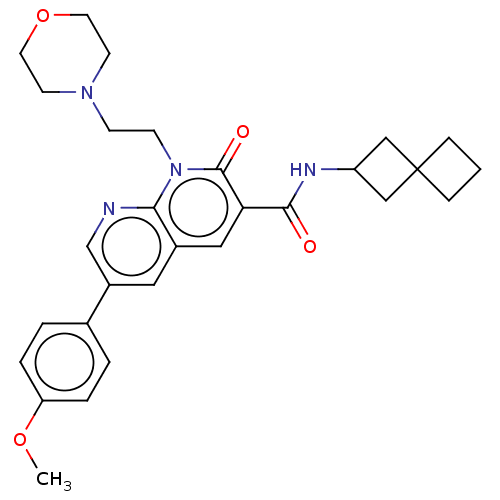
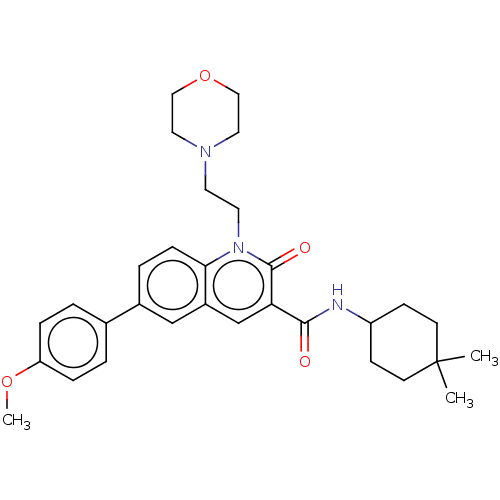
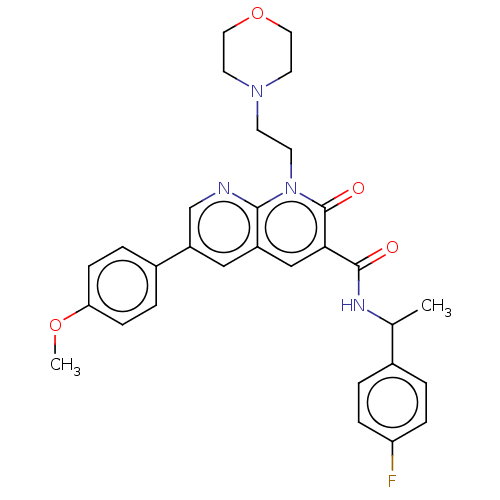
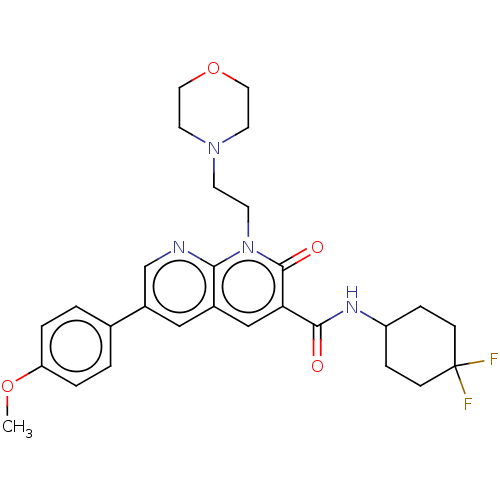
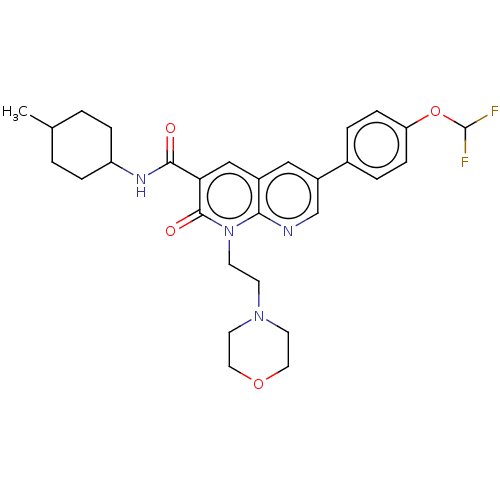
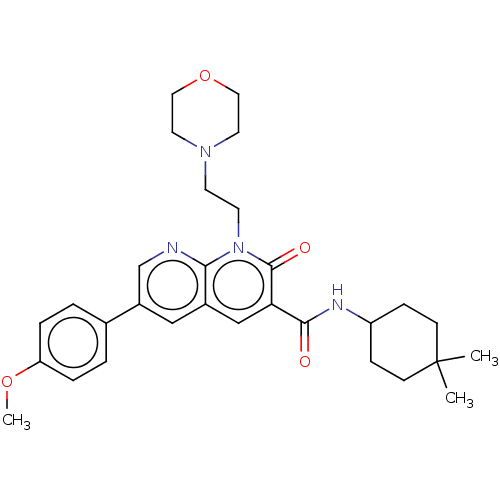
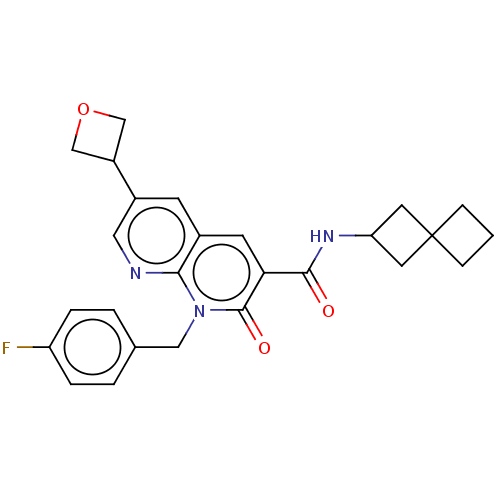
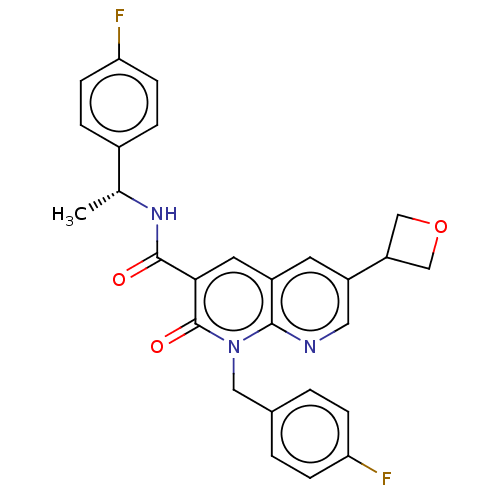
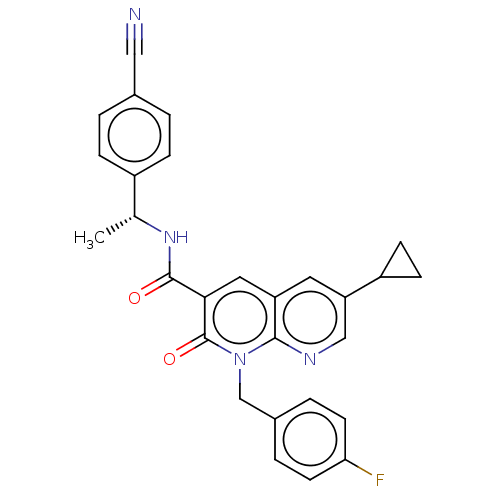
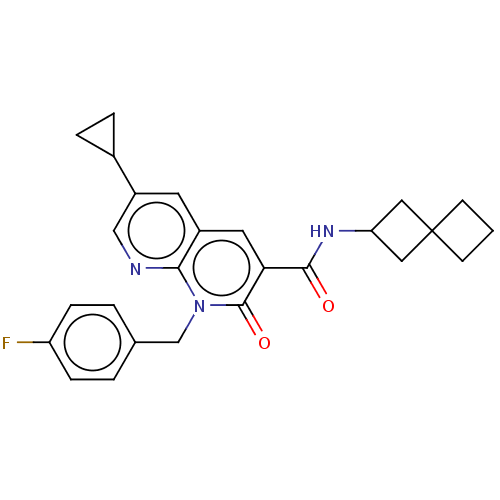
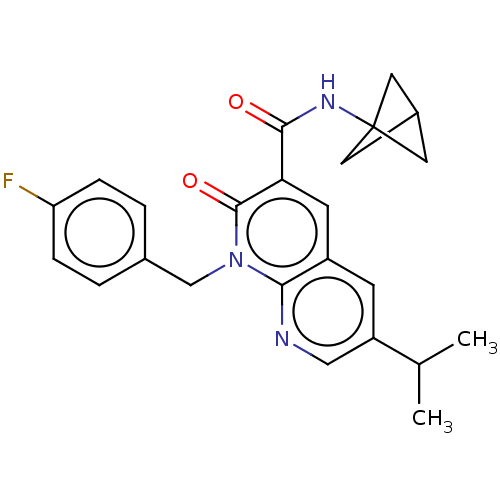
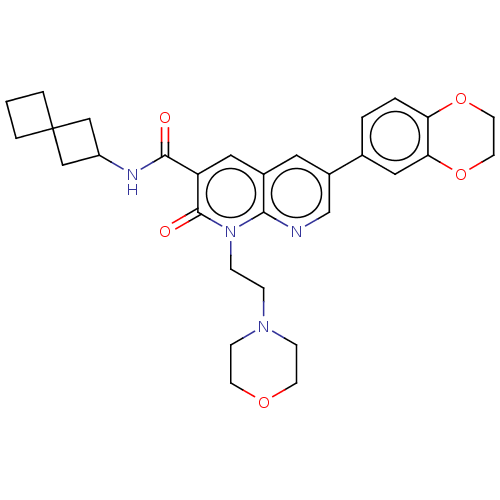
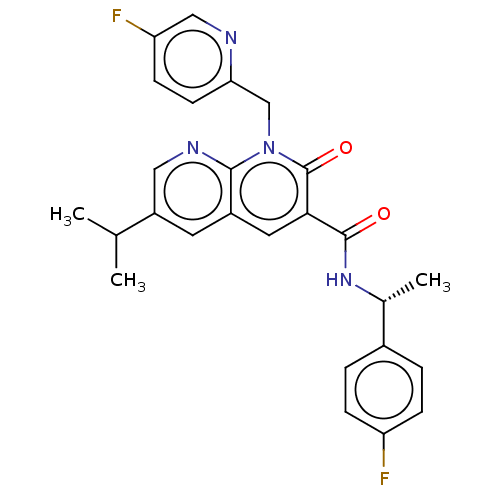
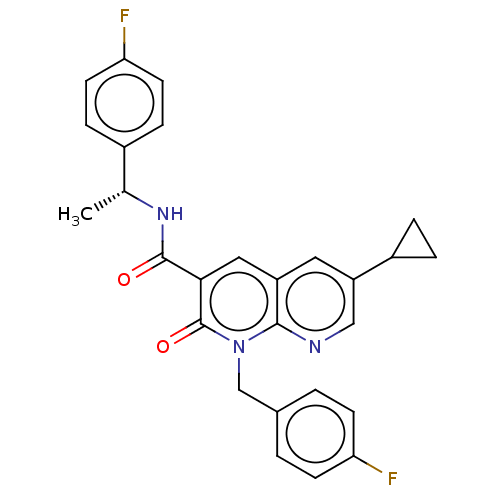
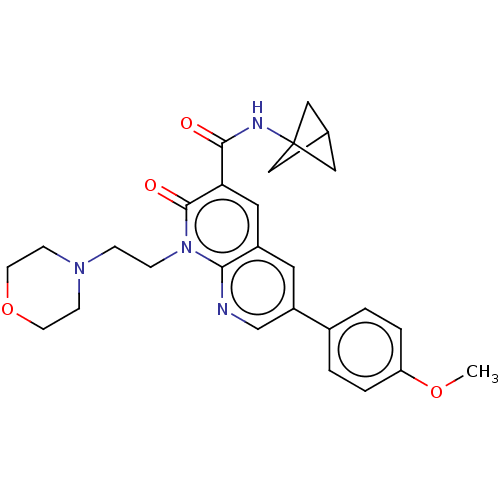
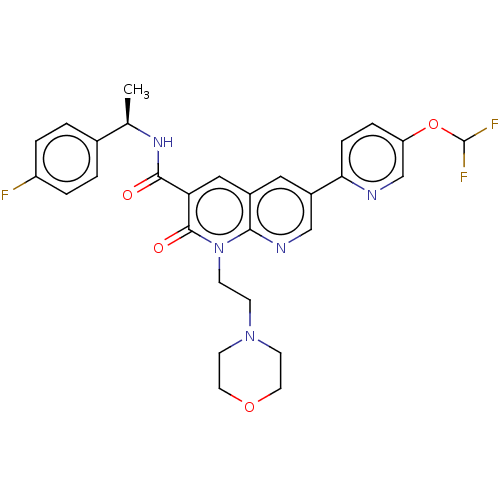
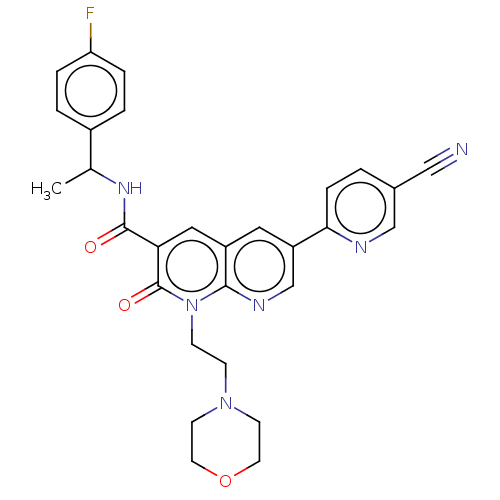
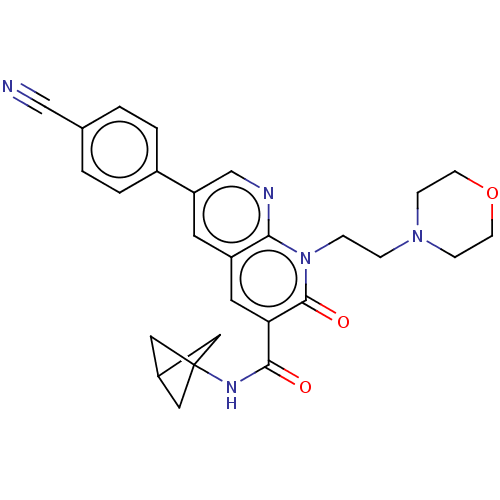
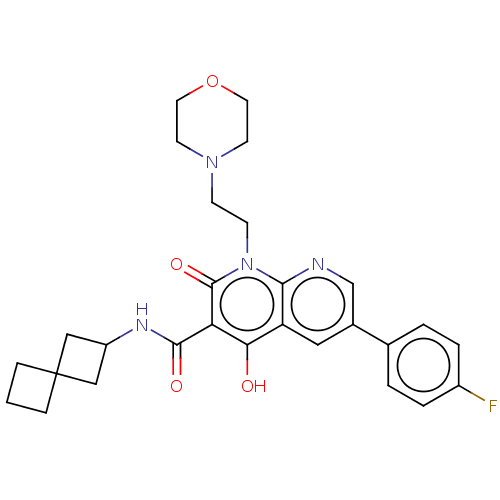
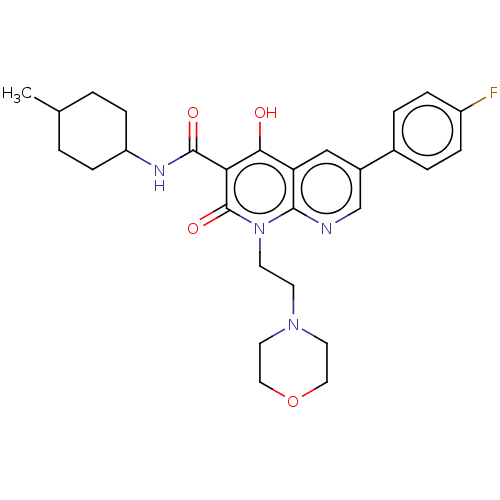
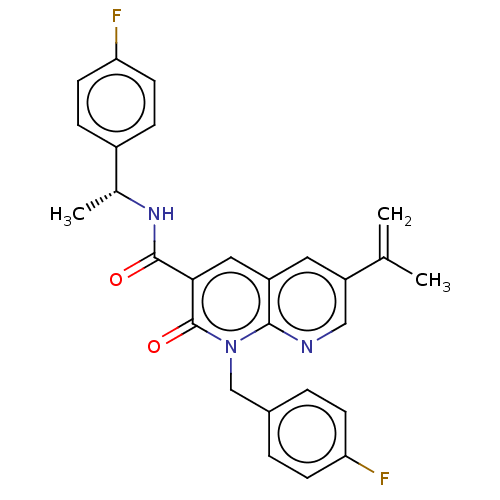
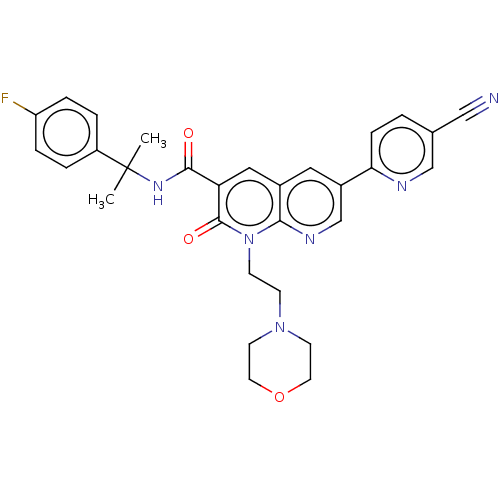
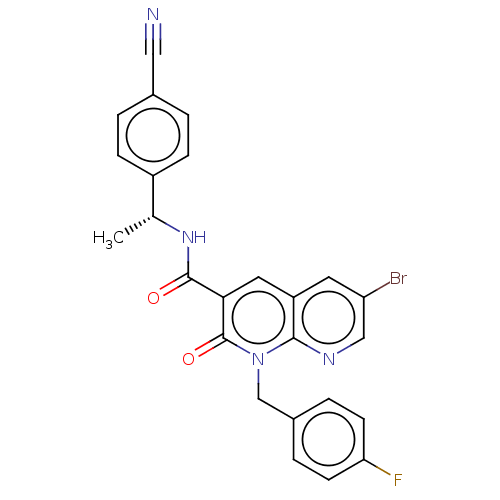
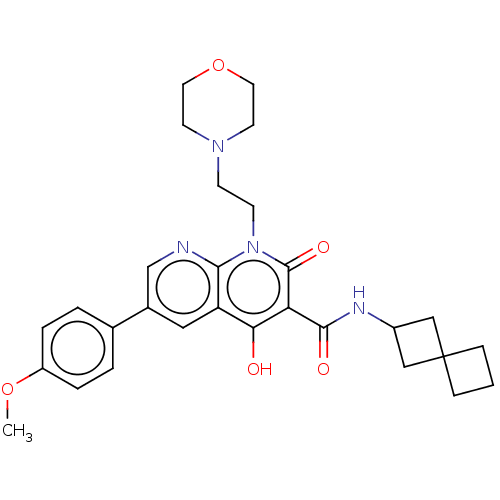
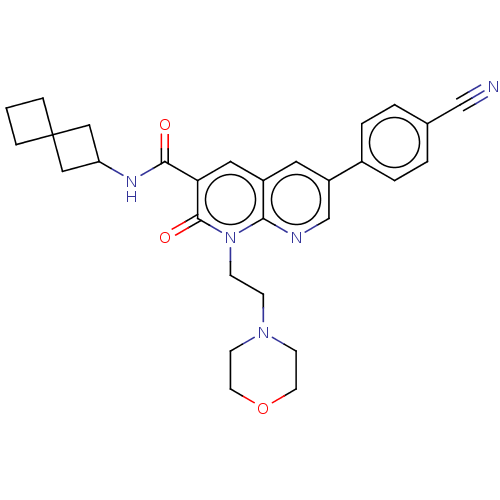
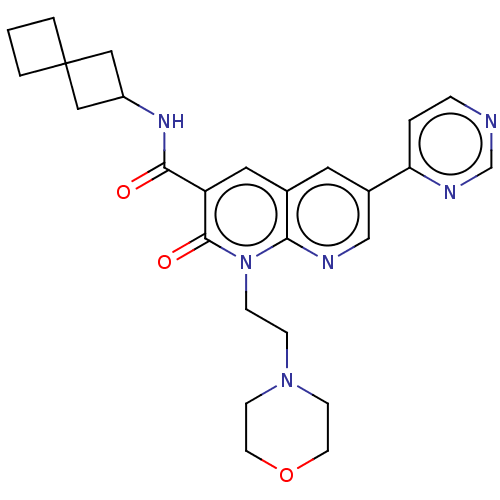
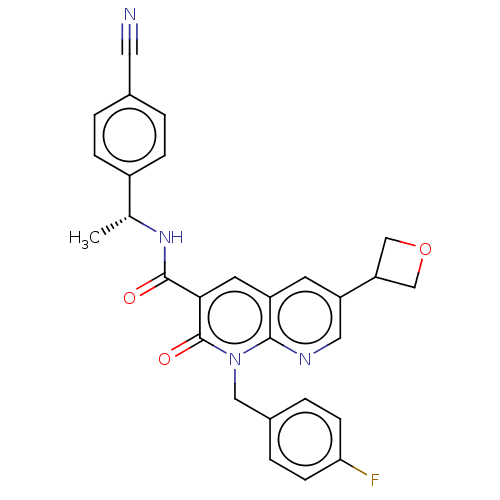
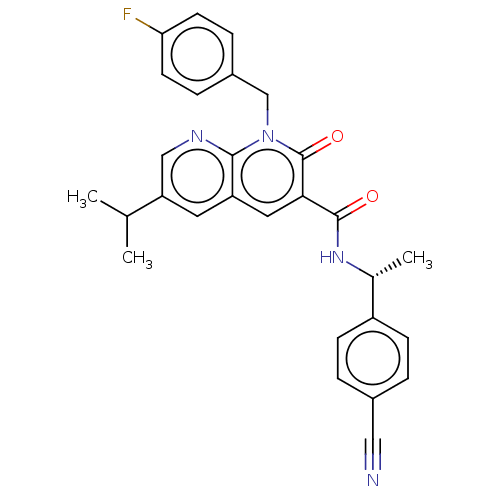
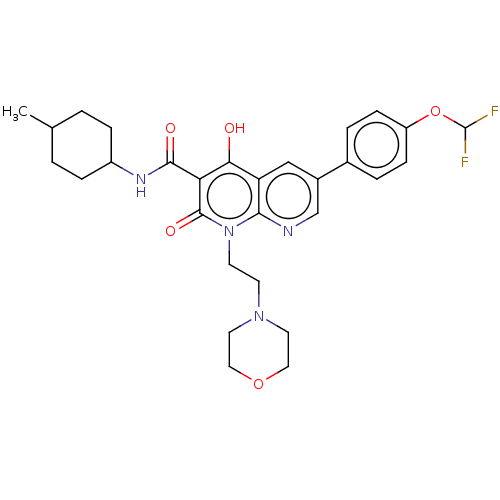
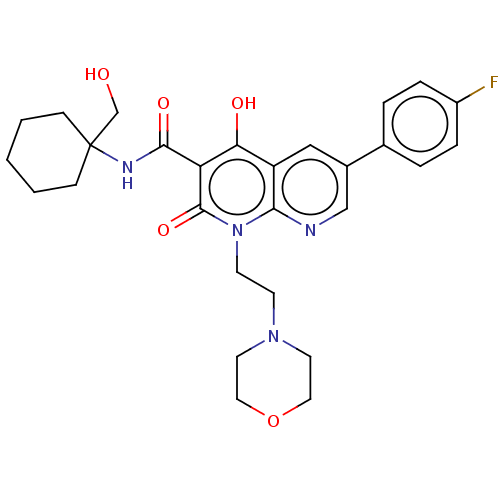
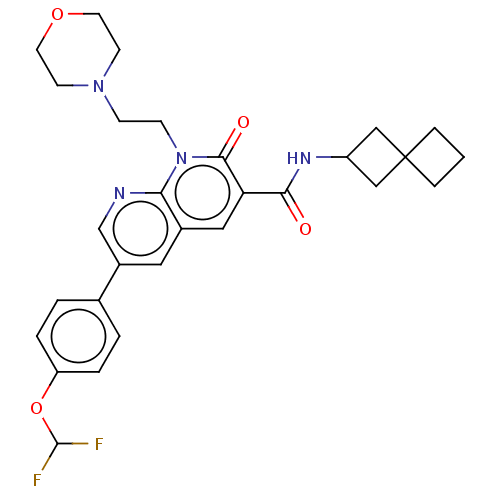
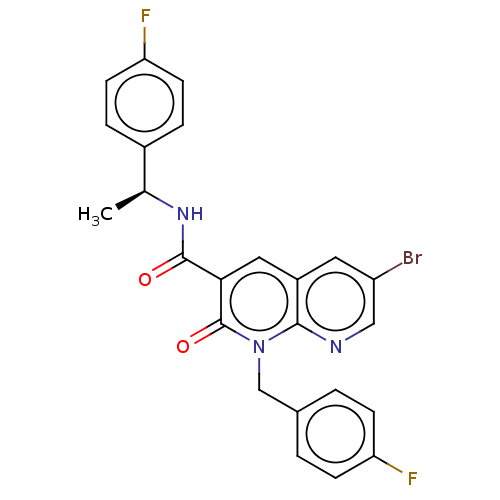
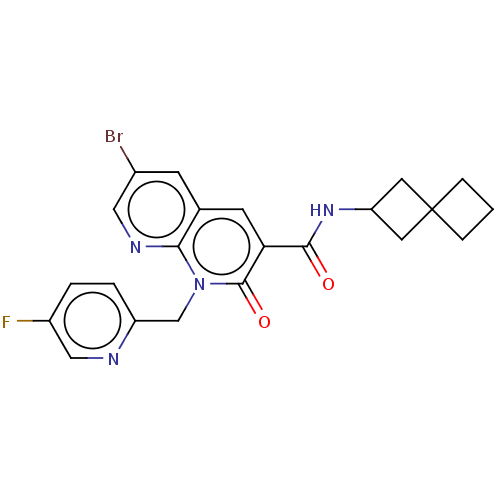
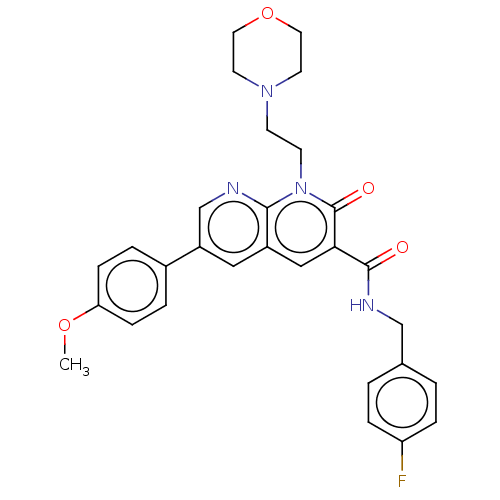
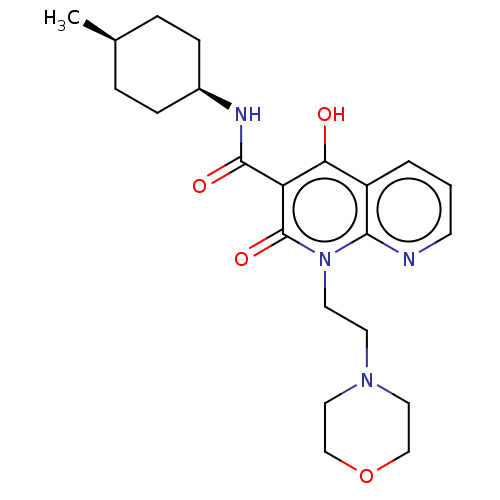
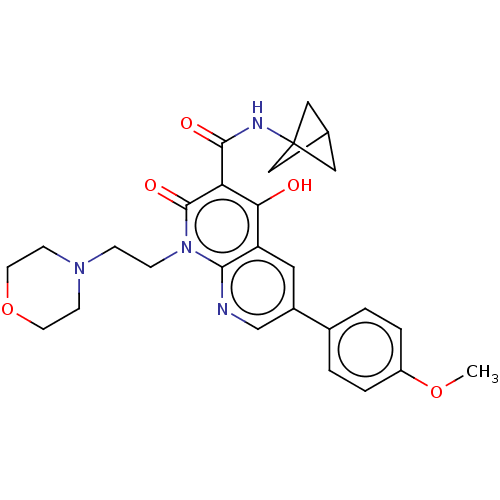
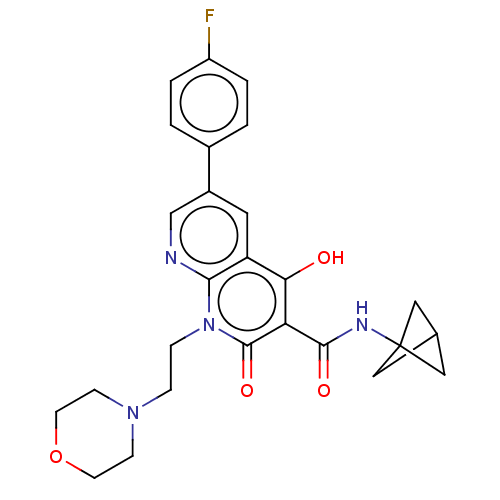
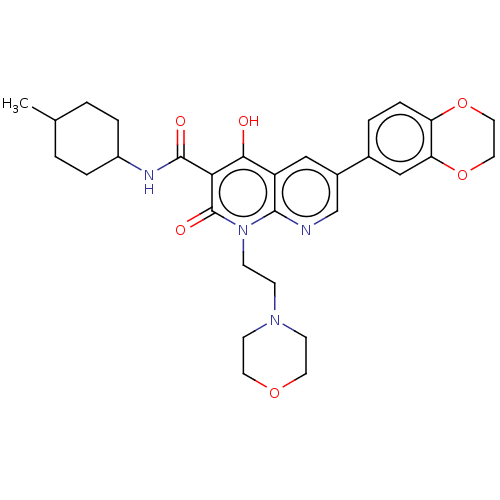
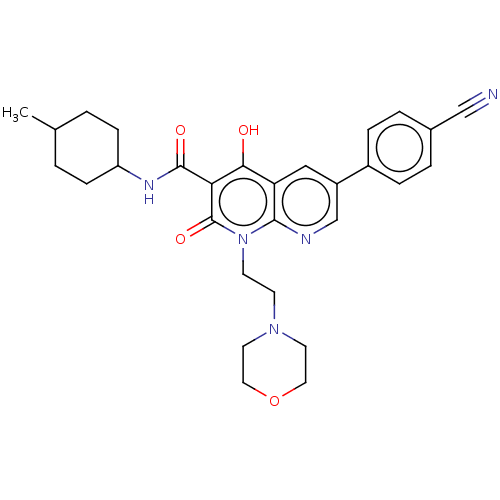
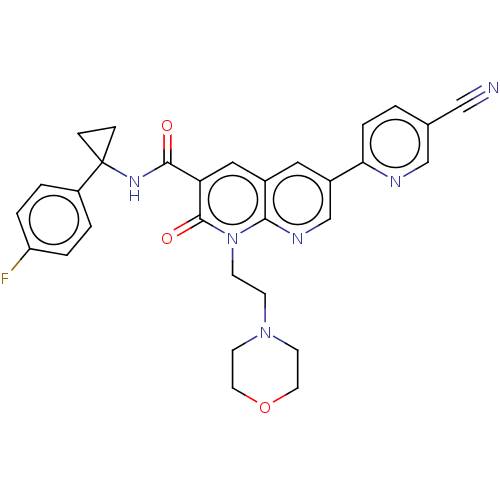
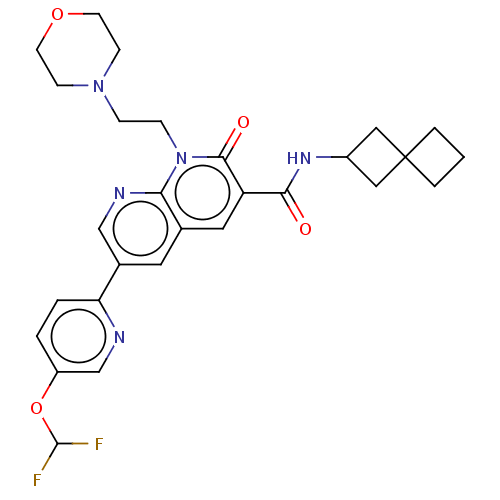
Report error Found 306 Enz. Inhib. hit(s) with all data for entry = 11106
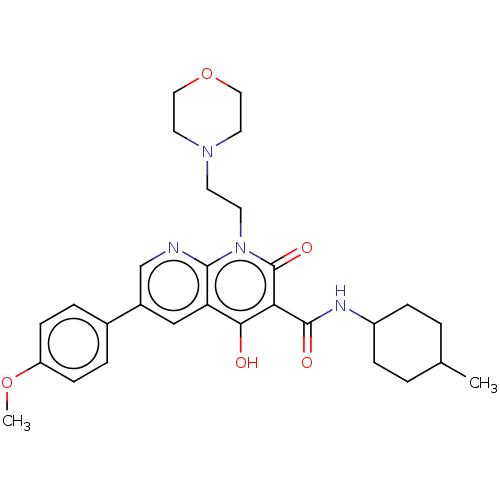
Affinity DataIC50: 0.600nMAssay Description:CB1R binding protocol involves the use of the same solution buffer used for both incubation and washing reaction (Tris-HCl, 50 mM; EDTA, 2.5 mM; MgCl...More data for this Ligand-Target Pair
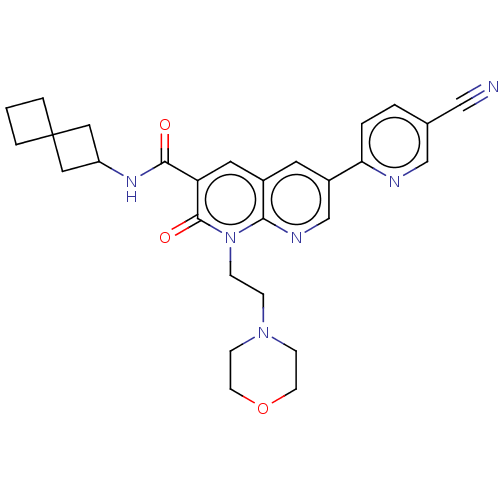
Affinity DataIC50: 0.600nMAssay Description:CB1R binding protocol involves the use of the same solution buffer used for both incubation and washing reaction (Tris-HCl, 50 mM; EDTA, 2.5 mM; MgCl...More data for this Ligand-Target Pair
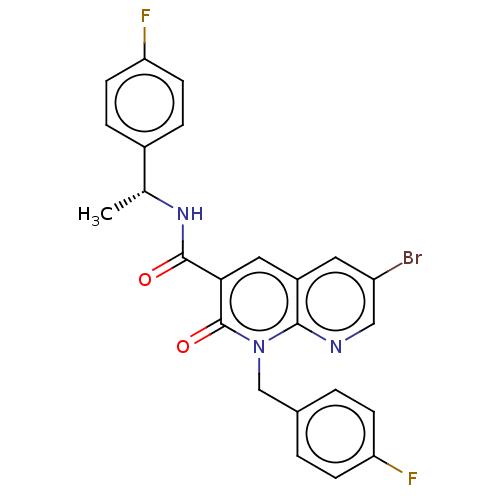
Affinity DataIC50: 0.700nMAssay Description:CB1R binding protocol involves the use of the same solution buffer used for both incubation and washing reaction (Tris-HCl, 50 mM; EDTA, 2.5 mM; MgCl...More data for this Ligand-Target Pair
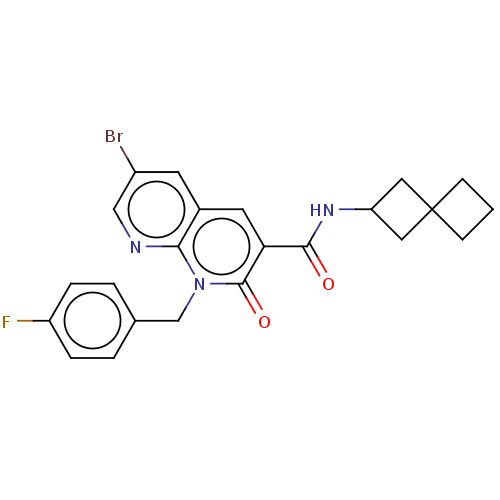
Affinity DataIC50: 0.800nMAssay Description:CB1R binding protocol involves the use of the same solution buffer used for both incubation and washing reaction (Tris-HCl, 50 mM; EDTA, 2.5 mM; MgCl...More data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:CB1R binding protocol involves the use of the same solution buffer used for both incubation and washing reaction (Tris-HCl, 50 mM; EDTA, 2.5 mM; MgCl...More data for this Ligand-Target Pair
Affinity DataIC50: 1.20nMAssay Description:CB1R binding protocol involves the use of the same solution buffer used for both incubation and washing reaction (Tris-HCl, 50 mM; EDTA, 2.5 mM; MgCl...More data for this Ligand-Target Pair
Affinity DataIC50: 1.20nMAssay Description:CB1R binding protocol involves the use of the same solution buffer used for both incubation and washing reaction (Tris-HCl, 50 mM; EDTA, 2.5 mM; MgCl...More data for this Ligand-Target Pair
Affinity DataIC50: 1.20nMAssay Description:CB1R binding protocol involves the use of the same solution buffer used for both incubation and washing reaction (Tris-HCl, 50 mM; EDTA, 2.5 mM; MgCl...More data for this Ligand-Target Pair
Affinity DataIC50: 2nMAssay Description:CB1R binding protocol involves the use of the same solution buffer used for both incubation and washing reaction (Tris-HCl, 50 mM; EDTA, 2.5 mM; MgCl...More data for this Ligand-Target Pair
Affinity DataIC50: 2.10nMAssay Description:CB1R binding protocol involves the use of the same solution buffer used for both incubation and washing reaction (Tris-HCl, 50 mM; EDTA, 2.5 mM; MgCl...More data for this Ligand-Target Pair
Affinity DataIC50: 2.20nMAssay Description:CB1R binding protocol involves the use of the same solution buffer used for both incubation and washing reaction (Tris-HCl, 50 mM; EDTA, 2.5 mM; MgCl...More data for this Ligand-Target Pair
Affinity DataIC50: 2.30nMAssay Description:CB1R binding protocol involves the use of the same solution buffer used for both incubation and washing reaction (Tris-HCl, 50 mM; EDTA, 2.5 mM; MgCl...More data for this Ligand-Target Pair
Affinity DataIC50: 3nMAssay Description:CB1R binding protocol involves the use of the same solution buffer used for both incubation and washing reaction (Tris-HCl, 50 mM; EDTA, 2.5 mM; MgCl...More data for this Ligand-Target Pair
Affinity DataIC50: 3.20nMAssay Description:CB1R binding protocol involves the use of the same solution buffer used for both incubation and washing reaction (Tris-HCl, 50 mM; EDTA, 2.5 mM; MgCl...More data for this Ligand-Target Pair
Affinity DataIC50: 3.60nMAssay Description:CB1R binding protocol involves the use of the same solution buffer used for both incubation and washing reaction (Tris-HCl, 50 mM; EDTA, 2.5 mM; MgCl...More data for this Ligand-Target Pair
Affinity DataIC50: 3.70nMAssay Description:CB1R binding protocol involves the use of the same solution buffer used for both incubation and washing reaction (Tris-HCl, 50 mM; EDTA, 2.5 mM; MgCl...More data for this Ligand-Target Pair
Affinity DataIC50: 4nMAssay Description:CB1R binding protocol involves the use of the same solution buffer used for both incubation and washing reaction (Tris-HCl, 50 mM; EDTA, 2.5 mM; MgCl...More data for this Ligand-Target Pair
Affinity DataIC50: 4nMAssay Description:CB1R binding protocol involves the use of the same solution buffer used for both incubation and washing reaction (Tris-HCl, 50 mM; EDTA, 2.5 mM; MgCl...More data for this Ligand-Target Pair
Affinity DataIC50: 4.10nMAssay Description:CB1R binding protocol involves the use of the same solution buffer used for both incubation and washing reaction (Tris-HCl, 50 mM; EDTA, 2.5 mM; MgCl...More data for this Ligand-Target Pair
Affinity DataIC50: 4.30nMAssay Description:CB1R binding protocol involves the use of the same solution buffer used for both incubation and washing reaction (Tris-HCl, 50 mM; EDTA, 2.5 mM; MgCl...More data for this Ligand-Target Pair
Affinity DataIC50: 4.5nMAssay Description:CB1R binding protocol involves the use of the same solution buffer used for both incubation and washing reaction (Tris-HCl, 50 mM; EDTA, 2.5 mM; MgCl...More data for this Ligand-Target Pair
Affinity DataIC50: 4.70nMAssay Description:CB1R binding protocol involves the use of the same solution buffer used for both incubation and washing reaction (Tris-HCl, 50 mM; EDTA, 2.5 mM; MgCl...More data for this Ligand-Target Pair
Affinity DataIC50: 5nMAssay Description:CB1R binding protocol involves the use of the same solution buffer used for both incubation and washing reaction (Tris-HCl, 50 mM; EDTA, 2.5 mM; MgCl...More data for this Ligand-Target Pair
Affinity DataIC50: 5nMAssay Description:CB1R binding protocol involves the use of the same solution buffer used for both incubation and washing reaction (Tris-HCl, 50 mM; EDTA, 2.5 mM; MgCl...More data for this Ligand-Target Pair
Affinity DataIC50: 5nMAssay Description:CB1R binding protocol involves the use of the same solution buffer used for both incubation and washing reaction (Tris-HCl, 50 mM; EDTA, 2.5 mM; MgCl...More data for this Ligand-Target Pair
Affinity DataIC50: 5.58nMAssay Description:CB1R binding protocol involves the use of the same solution buffer used for both incubation and washing reaction (Tris-HCl, 50 mM; EDTA, 2.5 mM; MgCl...More data for this Ligand-Target Pair
Affinity DataIC50: 6nMAssay Description:CB1R binding protocol involves the use of the same solution buffer used for both incubation and washing reaction (Tris-HCl, 50 mM; EDTA, 2.5 mM; MgCl...More data for this Ligand-Target Pair
Affinity DataIC50: 6.10nMAssay Description:CB1R binding protocol involves the use of the same solution buffer used for both incubation and washing reaction (Tris-HCl, 50 mM; EDTA, 2.5 mM; MgCl...More data for this Ligand-Target Pair
Affinity DataIC50: 6.5nMAssay Description:CB1R binding protocol involves the use of the same solution buffer used for both incubation and washing reaction (Tris-HCl, 50 mM; EDTA, 2.5 mM; MgCl...More data for this Ligand-Target Pair
Affinity DataIC50: 6.70nMAssay Description:CB1R binding protocol involves the use of the same solution buffer used for both incubation and washing reaction (Tris-HCl, 50 mM; EDTA, 2.5 mM; MgCl...More data for this Ligand-Target Pair
Affinity DataIC50: 7nMAssay Description:CB1R binding protocol involves the use of the same solution buffer used for both incubation and washing reaction (Tris-HCl, 50 mM; EDTA, 2.5 mM; MgCl...More data for this Ligand-Target Pair
Affinity DataIC50: 7nMAssay Description:CB1R binding protocol involves the use of the same solution buffer used for both incubation and washing reaction (Tris-HCl, 50 mM; EDTA, 2.5 mM; MgCl...More data for this Ligand-Target Pair
Affinity DataIC50: 7nMAssay Description:CB1R binding protocol involves the use of the same solution buffer used for both incubation and washing reaction (Tris-HCl, 50 mM; EDTA, 2.5 mM; MgCl...More data for this Ligand-Target Pair
Affinity DataIC50: 7.20nMAssay Description:CB1R binding protocol involves the use of the same solution buffer used for both incubation and washing reaction (Tris-HCl, 50 mM; EDTA, 2.5 mM; MgCl...More data for this Ligand-Target Pair
Affinity DataIC50: 7.5nMAssay Description:CB1R binding protocol involves the use of the same solution buffer used for both incubation and washing reaction (Tris-HCl, 50 mM; EDTA, 2.5 mM; MgCl...More data for this Ligand-Target Pair
Affinity DataIC50: 7.60nMAssay Description:CB1R binding protocol involves the use of the same solution buffer used for both incubation and washing reaction (Tris-HCl, 50 mM; EDTA, 2.5 mM; MgCl...More data for this Ligand-Target Pair
Affinity DataIC50: 7.90nMAssay Description:CB1R binding protocol involves the use of the same solution buffer used for both incubation and washing reaction (Tris-HCl, 50 mM; EDTA, 2.5 mM; MgCl...More data for this Ligand-Target Pair
Affinity DataIC50: 8.80nMAssay Description:CB1R binding protocol involves the use of the same solution buffer used for both incubation and washing reaction (Tris-HCl, 50 mM; EDTA, 2.5 mM; MgCl...More data for this Ligand-Target Pair
Affinity DataIC50: 9nMAssay Description:CB1R binding protocol involves the use of the same solution buffer used for both incubation and washing reaction (Tris-HCl, 50 mM; EDTA, 2.5 mM; MgCl...More data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:CB1R binding protocol involves the use of the same solution buffer used for both incubation and washing reaction (Tris-HCl, 50 mM; EDTA, 2.5 mM; MgCl...More data for this Ligand-Target Pair
Affinity DataIC50: 11nMAssay Description:CB1R binding protocol involves the use of the same solution buffer used for both incubation and washing reaction (Tris-HCl, 50 mM; EDTA, 2.5 mM; MgCl...More data for this Ligand-Target Pair
Affinity DataIC50: 12nMAssay Description:CB1R binding protocol involves the use of the same solution buffer used for both incubation and washing reaction (Tris-HCl, 50 mM; EDTA, 2.5 mM; MgCl...More data for this Ligand-Target Pair
Affinity DataIC50: 14nMAssay Description:CB1R binding protocol involves the use of the same solution buffer used for both incubation and washing reaction (Tris-HCl, 50 mM; EDTA, 2.5 mM; MgCl...More data for this Ligand-Target Pair
Affinity DataIC50: 15nMAssay Description:CB1R binding protocol involves the use of the same solution buffer used for both incubation and washing reaction (Tris-HCl, 50 mM; EDTA, 2.5 mM; MgCl...More data for this Ligand-Target Pair
Affinity DataIC50: 17nMAssay Description:CB1R binding protocol involves the use of the same solution buffer used for both incubation and washing reaction (Tris-HCl, 50 mM; EDTA, 2.5 mM; MgCl...More data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:CB1R binding protocol involves the use of the same solution buffer used for both incubation and washing reaction (Tris-HCl, 50 mM; EDTA, 2.5 mM; MgCl...More data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:CB1R binding protocol involves the use of the same solution buffer used for both incubation and washing reaction (Tris-HCl, 50 mM; EDTA, 2.5 mM; MgCl...More data for this Ligand-Target Pair
Affinity DataIC50: 22.6nMAssay Description:CB1R binding protocol involves the use of the same solution buffer used for both incubation and washing reaction (Tris-HCl, 50 mM; EDTA, 2.5 mM; MgCl...More data for this Ligand-Target Pair
Affinity DataIC50: 23nMAssay Description:CB1R binding protocol involves the use of the same solution buffer used for both incubation and washing reaction (Tris-HCl, 50 mM; EDTA, 2.5 mM; MgCl...More data for this Ligand-Target Pair
Affinity DataIC50: 24.9nMAssay Description:CB1R binding protocol involves the use of the same solution buffer used for both incubation and washing reaction (Tris-HCl, 50 mM; EDTA, 2.5 mM; MgCl...More data for this Ligand-Target Pair