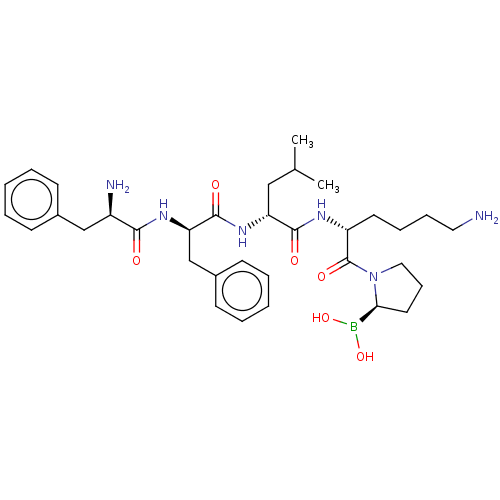
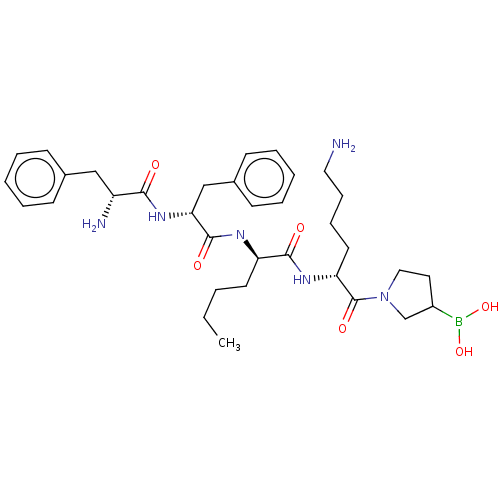
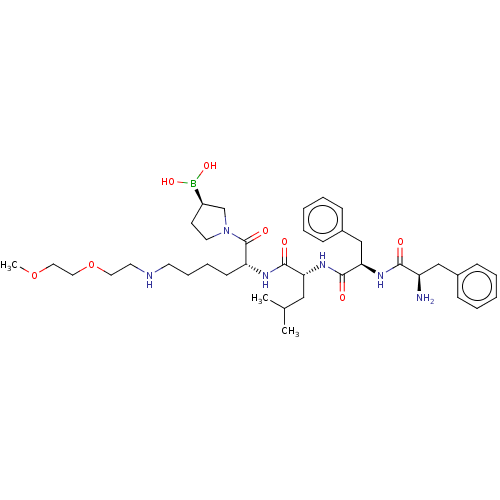
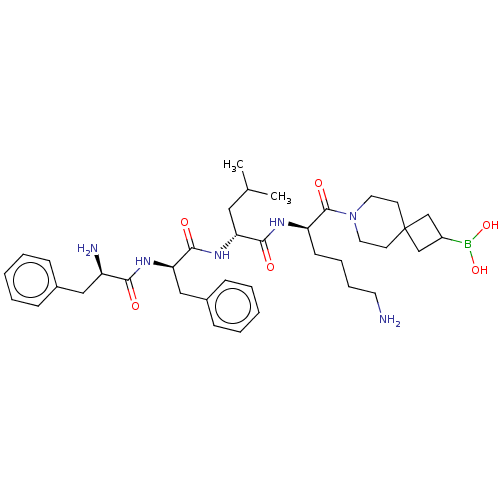
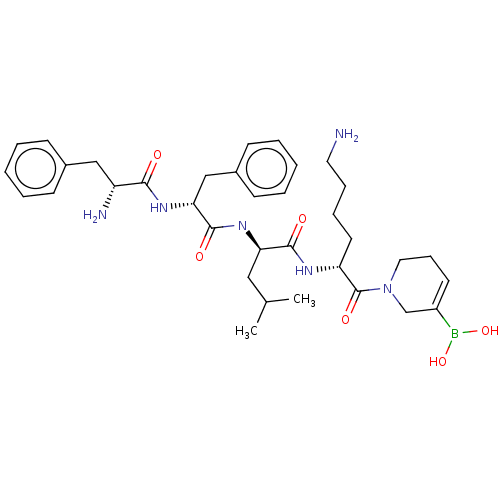
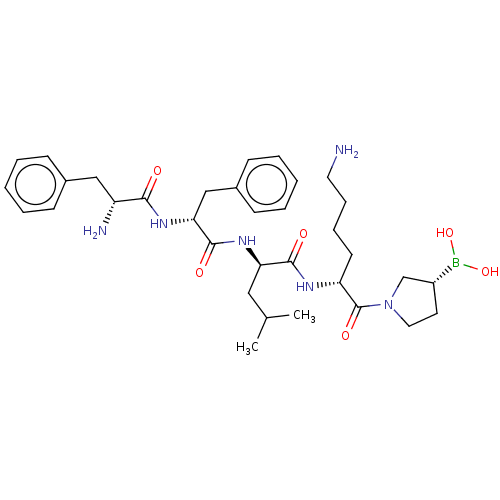
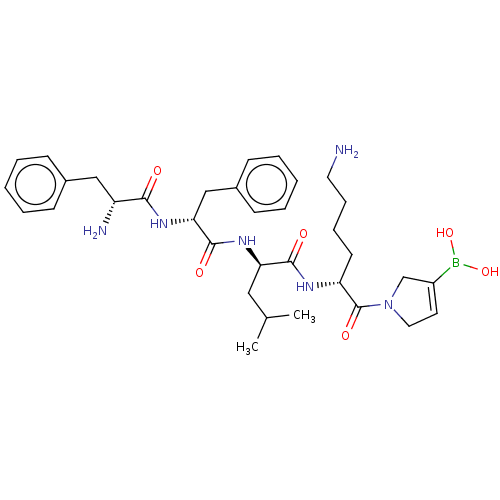
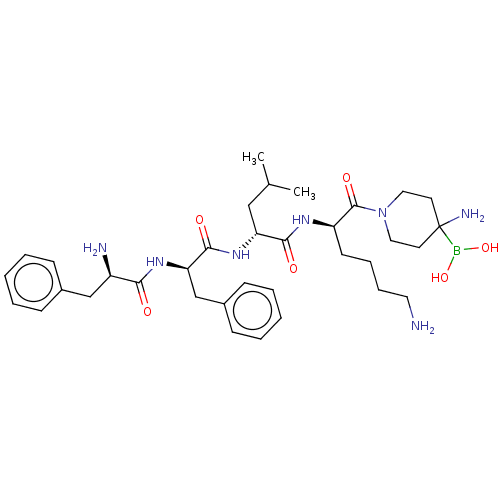
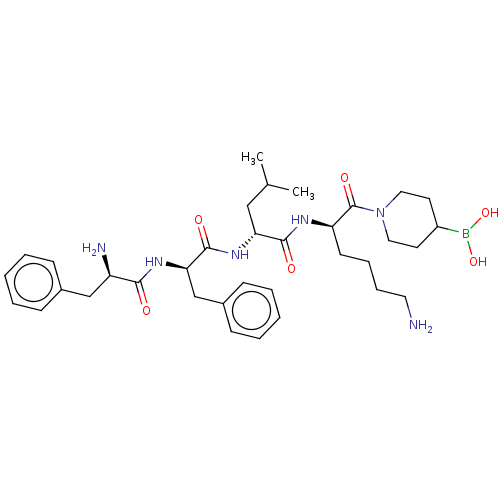
Report error Found 60 Enz. Inhib. hit(s) with all data for entry = 11243
Affinity DataEC50: 0.00100nMAssay Description:Forskolin can stimulate the release of cAMP from HEK293 cells with high expression of human κ (or μ, or δ)-opioid receptors. The κ...More data for this Ligand-Target Pair
Affinity DataEC50: 0.00800nMAssay Description:Forskolin can stimulate the release of cAMP from HEK293 cells with high expression of human κ (or μ, or δ)-opioid receptors. The κ...More data for this Ligand-Target Pair
Affinity DataEC50: 0.00900nMAssay Description:Forskolin can stimulate the release of cAMP from HEK293 cells with high expression of human κ (or μ, or δ)-opioid receptors. The κ...More data for this Ligand-Target Pair
Affinity DataEC50: 0.00900nMAssay Description:Forskolin can stimulate the release of cAMP from HEK293 cells with high expression of human κ (or μ, or δ)-opioid receptors. The κ...More data for this Ligand-Target Pair
Affinity DataEC50: 0.0270nMAssay Description:Forskolin can stimulate the release of cAMP from HEK293 cells with high expression of human κ (or μ, or δ)-opioid receptors. The κ...More data for this Ligand-Target Pair
Affinity DataEC50: 0.0330nMAssay Description:Forskolin can stimulate the release of cAMP from HEK293 cells with high expression of human κ (or μ, or δ)-opioid receptors. The κ...More data for this Ligand-Target Pair
Affinity DataEC50: 0.0550nMAssay Description:Forskolin can stimulate the release of cAMP from HEK293 cells with high expression of human κ (or μ, or δ)-opioid receptors. The κ...More data for this Ligand-Target Pair
Affinity DataEC50: 0.0850nMAssay Description:Forskolin can stimulate the release of cAMP from HEK293 cells with high expression of human κ (or μ, or δ)-opioid receptors. The κ...More data for this Ligand-Target Pair
Affinity DataEC50: 0.0950nMAssay Description:Forskolin can stimulate the release of cAMP from HEK293 cells with high expression of human κ (or μ, or δ)-opioid receptors. The κ...More data for this Ligand-Target Pair
Affinity DataEC50: 0.0980nMAssay Description:Forskolin can stimulate the release of cAMP from HEK293 cells with high expression of human κ (or μ, or δ)-opioid receptors. The κ...More data for this Ligand-Target Pair
Affinity DataEC50: >1.00E+4nMAssay Description:Forskolin can stimulate the release of cAMP from HEK293 cells with high expression of human κ (or μ, or δ)-opioid receptors. The κ...More data for this Ligand-Target Pair
Affinity DataEC50: >1.00E+4nMAssay Description:Forskolin can stimulate the release of cAMP from HEK293 cells with high expression of human κ (or μ, or δ)-opioid receptors. The κ...More data for this Ligand-Target Pair
Affinity DataEC50: >1.00E+4nMAssay Description:Forskolin can stimulate the release of cAMP from HEK293 cells with high expression of human κ (or μ, or δ)-opioid receptors. The κ...More data for this Ligand-Target Pair
Affinity DataEC50: >1.00E+4nMAssay Description:Forskolin can stimulate the release of cAMP from HEK293 cells with high expression of human κ (or μ, or δ)-opioid receptors. The κ...More data for this Ligand-Target Pair
Affinity DataEC50: >1.00E+4nMAssay Description:Forskolin can stimulate the release of cAMP from HEK293 cells with high expression of human κ (or μ, or δ)-opioid receptors. The κ...More data for this Ligand-Target Pair
Affinity DataEC50: >1.00E+4nMAssay Description:Forskolin can stimulate the release of cAMP from HEK293 cells with high expression of human κ (or μ, or δ)-opioid receptors. The κ...More data for this Ligand-Target Pair
Affinity DataEC50: >1.00E+4nMAssay Description:Forskolin can stimulate the release of cAMP from HEK293 cells with high expression of human κ (or μ, or δ)-opioid receptors. The κ...More data for this Ligand-Target Pair
Affinity DataEC50: >1.00E+4nMAssay Description:Forskolin can stimulate the release of cAMP from HEK293 cells with high expression of human κ (or μ, or δ)-opioid receptors. The κ...More data for this Ligand-Target Pair
Affinity DataEC50: >1.00E+4nMAssay Description:Forskolin can stimulate the release of cAMP from HEK293 cells with high expression of human κ (or μ, or δ)-opioid receptors. The κ...More data for this Ligand-Target Pair
Affinity DataEC50: >1.00E+4nMAssay Description:Forskolin can stimulate the release of cAMP from HEK293 cells with high expression of human κ (or μ, or δ)-opioid receptors. The κ...More data for this Ligand-Target Pair
Affinity DataEC50: >1.00E+4nMAssay Description:Forskolin can stimulate the release of cAMP from HEK293 cells with high expression of human κ (or μ, or δ)-opioid receptors. The κ...More data for this Ligand-Target Pair
Affinity DataEC50: >1.00E+4nMAssay Description:Forskolin can stimulate the release of cAMP from HEK293 cells with high expression of human κ (or μ, or δ)-opioid receptors. The κ...More data for this Ligand-Target Pair
Affinity DataEC50: >1.00E+4nMAssay Description:Forskolin can stimulate the release of cAMP from HEK293 cells with high expression of human κ (or μ, or δ)-opioid receptors. The κ...More data for this Ligand-Target Pair
Affinity DataEC50: >1.00E+4nMAssay Description:Forskolin can stimulate the release of cAMP from HEK293 cells with high expression of human κ (or μ, or δ)-opioid receptors. The κ...More data for this Ligand-Target Pair
Affinity DataEC50: >1.00E+4nMAssay Description:Forskolin can stimulate the release of cAMP from HEK293 cells with high expression of human κ (or μ, or δ)-opioid receptors. The κ...More data for this Ligand-Target Pair
Affinity DataEC50: >1.00E+4nMAssay Description:Forskolin can stimulate the release of cAMP from HEK293 cells with high expression of human κ (or μ, or δ)-opioid receptors. The κ...More data for this Ligand-Target Pair
Affinity DataEC50: >1.00E+4nMAssay Description:Forskolin can stimulate the release of cAMP from HEK293 cells with high expression of human κ (or μ, or δ)-opioid receptors. The κ...More data for this Ligand-Target Pair
Affinity DataEC50: >1.00E+4nMAssay Description:Forskolin can stimulate the release of cAMP from HEK293 cells with high expression of human κ (or μ, or δ)-opioid receptors. The κ...More data for this Ligand-Target Pair
Affinity DataEC50: >1.00E+4nMAssay Description:Forskolin can stimulate the release of cAMP from HEK293 cells with high expression of human κ (or μ, or δ)-opioid receptors. The κ...More data for this Ligand-Target Pair
Affinity DataEC50: >1.00E+4nMAssay Description:Forskolin can stimulate the release of cAMP from HEK293 cells with high expression of human κ (or μ, or δ)-opioid receptors. The κ...More data for this Ligand-Target Pair
Affinity DataIC50: 5.00E+4nMAssay Description:The human liver microsomes (0.253 mg/mL protein) containing cytochrome P450, test compounds (0.05-50 μM), CYPs substrates (10 μM p-acetamin...More data for this Ligand-Target Pair
Affinity DataIC50: 5.00E+4nMAssay Description:The human liver microsomes (0.253 mg/mL protein) containing cytochrome P450, test compounds (0.05-50 μM), CYPs substrates (10 μM p-acetamin...More data for this Ligand-Target Pair
Affinity DataIC50: 5.00E+4nMAssay Description:The human liver microsomes (0.253 mg/mL protein) containing cytochrome P450, test compounds (0.05-50 μM), CYPs substrates (10 μM p-acetamin...More data for this Ligand-Target Pair
Affinity DataIC50: 5.00E+4nMAssay Description:The human liver microsomes (0.253 mg/mL protein) containing cytochrome P450, test compounds (0.05-50 μM), CYPs substrates (10 μM p-acetamin...More data for this Ligand-Target Pair
Affinity DataIC50: 5.00E+4nMAssay Description:The human liver microsomes (0.253 mg/mL protein) containing cytochrome P450, test compounds (0.05-50 μM), CYPs substrates (10 μM p-acetamin...More data for this Ligand-Target Pair
Affinity DataIC50: 5.00E+4nMAssay Description:The human liver microsomes (0.253 mg/mL protein) containing cytochrome P450, test compounds (0.05-50 μM), CYPs substrates (10 μM p-acetamin...More data for this Ligand-Target Pair
Affinity DataIC50: 5.00E+4nMAssay Description:The human liver microsomes (0.253 mg/mL protein) containing cytochrome P450, test compounds (0.05-50 μM), CYPs substrates (10 μM p-acetamin...More data for this Ligand-Target Pair
Affinity DataIC50: 5.00E+4nMAssay Description:The human liver microsomes (0.253 mg/mL protein) containing cytochrome P450, test compounds (0.05-50 μM), CYPs substrates (10 μM p-acetamin...More data for this Ligand-Target Pair
Affinity DataIC50: 5.00E+4nMAssay Description:The human liver microsomes (0.253 mg/mL protein) containing cytochrome P450, test compounds (0.05-50 μM), CYPs substrates (10 μM p-acetamin...More data for this Ligand-Target Pair
Affinity DataIC50: 5.00E+4nMAssay Description:The human liver microsomes (0.253 mg/mL protein) containing cytochrome P450, test compounds (0.05-50 μM), CYPs substrates (10 μM p-acetamin...More data for this Ligand-Target Pair
Affinity DataIC50: 5.00E+4nMAssay Description:The human liver microsomes (0.253 mg/mL protein) containing cytochrome P450, test compounds (0.05-50 μM), CYPs substrates (10 μM p-acetamin...More data for this Ligand-Target Pair
Affinity DataIC50: 5.00E+4nMAssay Description:The human liver microsomes (0.253 mg/mL protein) containing cytochrome P450, test compounds (0.05-50 μM), CYPs substrates (10 μM p-acetamin...More data for this Ligand-Target Pair
Affinity DataIC50: 5.00E+4nMAssay Description:The human liver microsomes (0.253 mg/mL protein) containing cytochrome P450, test compounds (0.05-50 μM), CYPs substrates (10 μM p-acetamin...More data for this Ligand-Target Pair
Affinity DataIC50: 5.00E+4nMAssay Description:The human liver microsomes (0.253 mg/mL protein) containing cytochrome P450, test compounds (0.05-50 μM), CYPs substrates (10 μM p-acetamin...More data for this Ligand-Target Pair
Affinity DataIC50: 5.00E+4nMAssay Description:The human liver microsomes (0.253 mg/mL protein) containing cytochrome P450, test compounds (0.05-50 μM), CYPs substrates (10 μM p-acetamin...More data for this Ligand-Target Pair
Affinity DataIC50: 5.00E+4nMAssay Description:The human liver microsomes (0.253 mg/mL protein) containing cytochrome P450, test compounds (0.05-50 μM), CYPs substrates (10 μM p-acetamin...More data for this Ligand-Target Pair
Affinity DataIC50: 5.00E+4nMAssay Description:The human liver microsomes (0.253 mg/mL protein) containing cytochrome P450, test compounds (0.05-50 μM), CYPs substrates (10 μM p-acetamin...More data for this Ligand-Target Pair
Affinity DataIC50: 5.00E+4nMAssay Description:The human liver microsomes (0.253 mg/mL protein) containing cytochrome P450, test compounds (0.05-50 μM), CYPs substrates (10 μM p-acetamin...More data for this Ligand-Target Pair
Affinity DataIC50: 5.00E+4nMAssay Description:The human liver microsomes (0.253 mg/mL protein) containing cytochrome P450, test compounds (0.05-50 μM), CYPs substrates (10 μM p-acetamin...More data for this Ligand-Target Pair
Affinity DataIC50: 5.00E+4nMAssay Description:The human liver microsomes (0.253 mg/mL protein) containing cytochrome P450, test compounds (0.05-50 μM), CYPs substrates (10 μM p-acetamin...More data for this Ligand-Target Pair