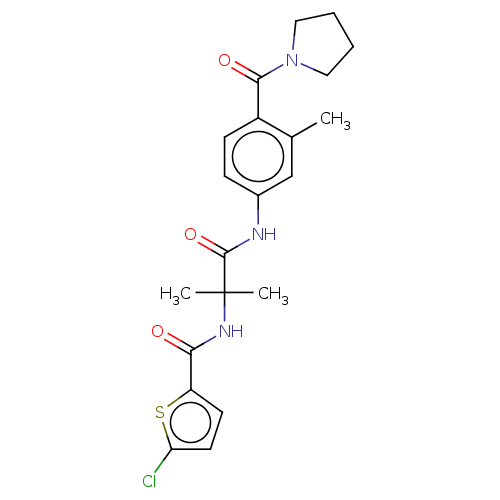
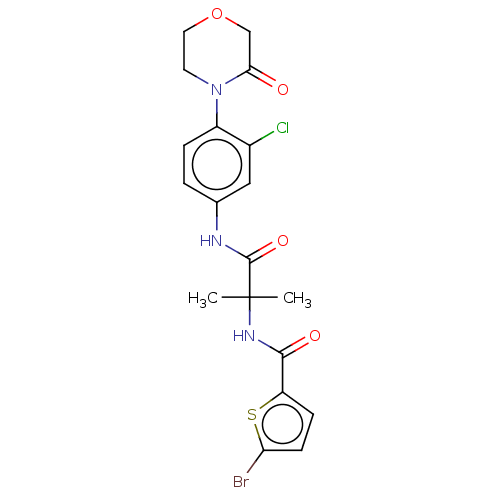
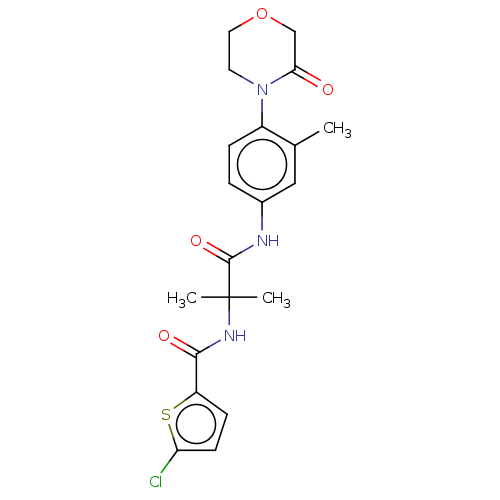
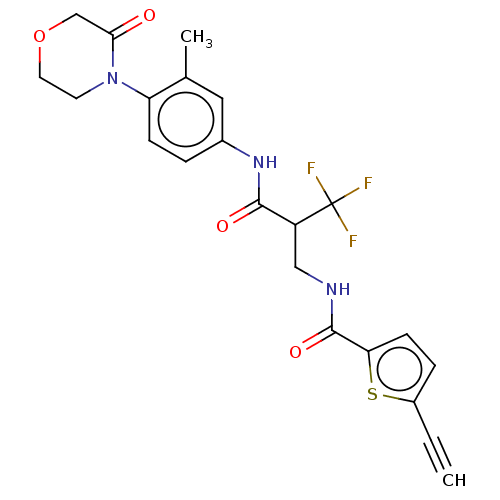
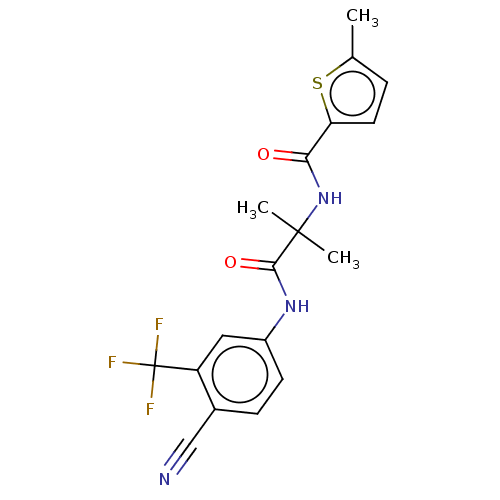
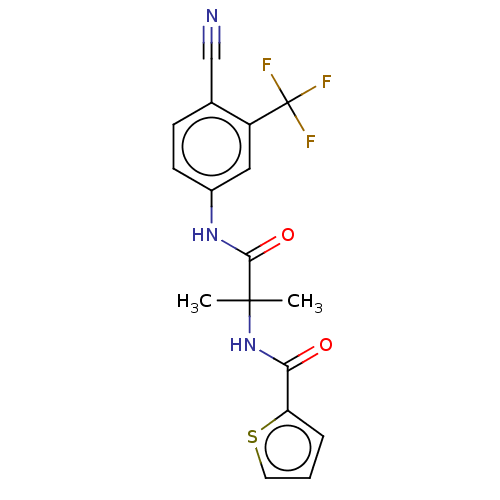
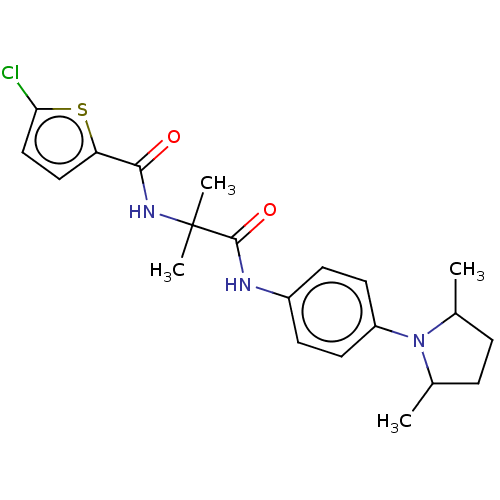
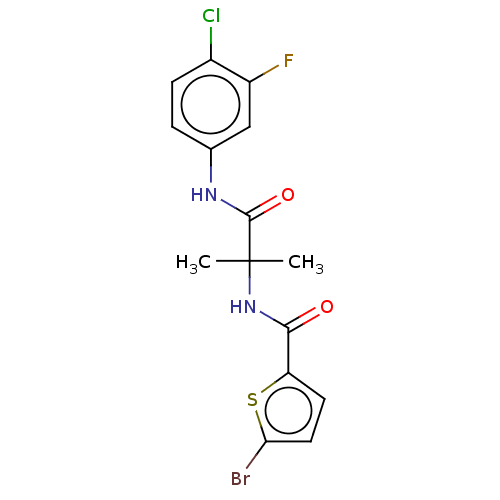
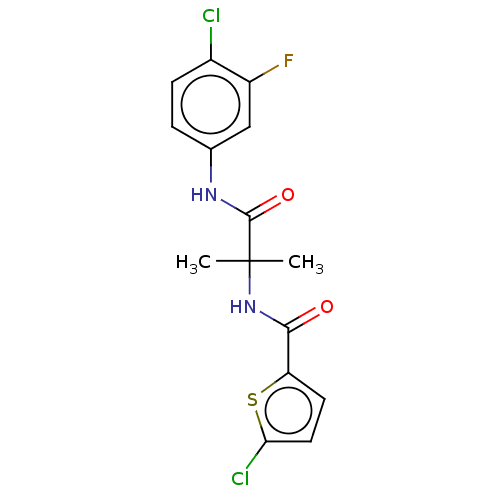
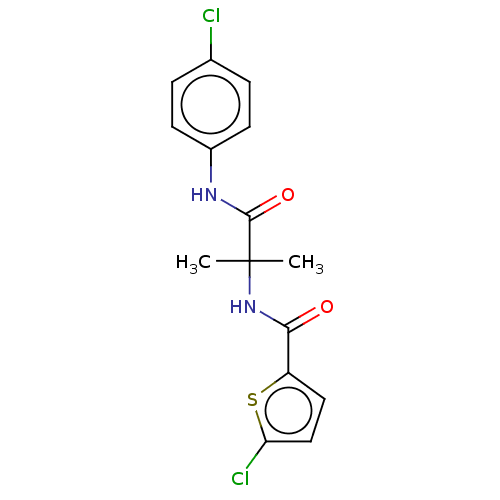
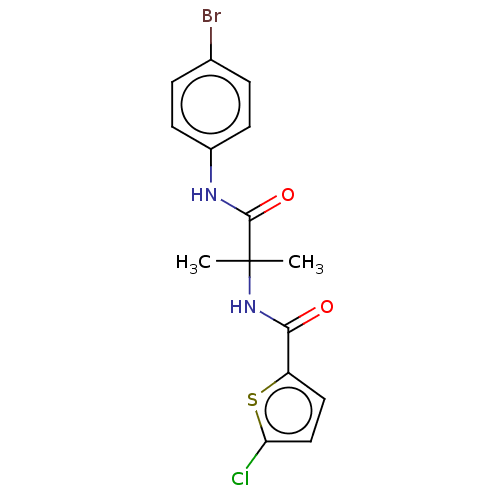
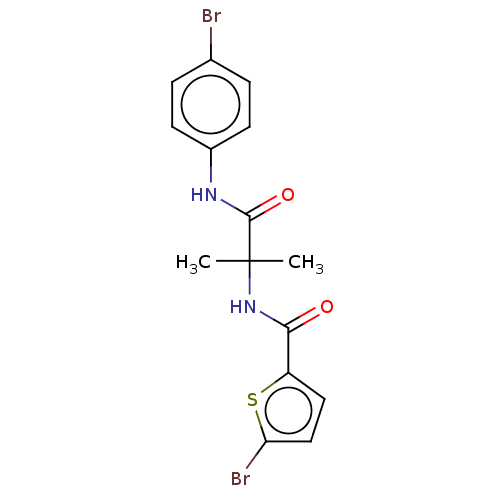
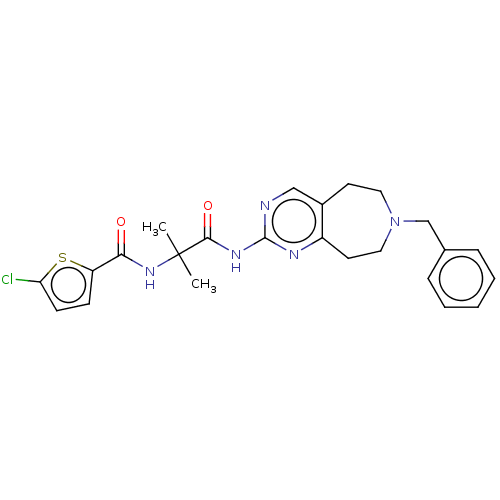
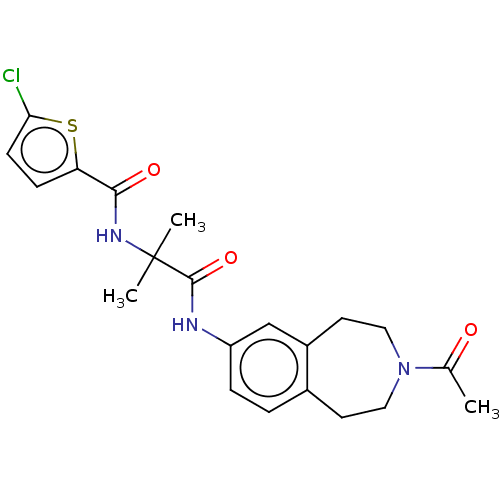
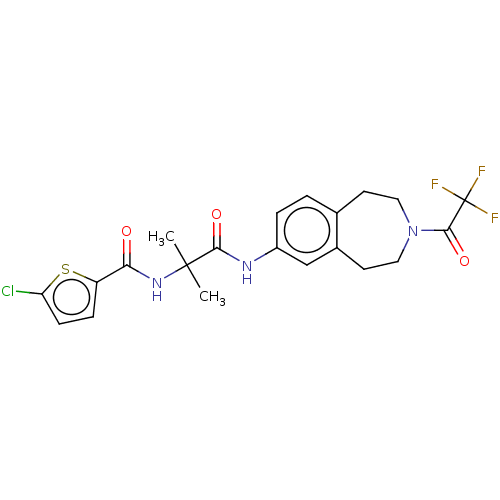
Report error Found 40 Enz. Inhib. hit(s) with all data for entry = 11325
Affinity DataIC50: 8nMAssay Description:Measurement of active human Factor Xa using a specific chromogenic substrate which is labeled with a p-nitro-anilino group (pNA). The cleavage in bet...More data for this Ligand-Target Pair
Affinity DataIC50: 26nMAssay Description:Measurement of active human Factor Xa using a specific chromogenic substrate which is labeled with a p-nitro-anilino group (pNA). The cleavage in bet...More data for this Ligand-Target Pair
Affinity DataIC50: 40nMAssay Description:Measurement of active human Factor Xa using a specific chromogenic substrate which is labeled with a p-nitro-anilino group (pNA). The cleavage in bet...More data for this Ligand-Target Pair
Affinity DataIC50: 48nMAssay Description:Measurement of active human Factor Xa using a specific chromogenic substrate which is labeled with a p-nitro-anilino group (pNA). The cleavage in bet...More data for this Ligand-Target Pair
Affinity DataIC50: 88nMAssay Description:Determination of In Vitro Pharmacological Activity. The human androgen receptor (AR) assay was run using Product #IB03001 from Indigo Biosciences. Th...More data for this Ligand-Target Pair
Affinity DataIC50: 353nMAssay Description:Determination of In Vitro Pharmacological Activity. The human androgen receptor (AR) assay was run using Product #IB03001 from Indigo Biosciences. Th...More data for this Ligand-Target Pair
Affinity DataIC50: 429nMAssay Description:Measurement of active human Factor Xa using a specific chromogenic substrate which is labeled with a p-nitro-anilino group (pNA). The cleavage in bet...More data for this Ligand-Target Pair
Affinity DataIC50: 657nMAssay Description:Determination of In Vitro Pharmacological Activity. The human androgen receptor (AR) assay was run using Product #IB03001 from Indigo Biosciences. Th...More data for this Ligand-Target Pair
Affinity DataIC50: 859nMAssay Description:Determination of In Vitro Pharmacological Activity. The human androgen receptor (AR) assay was run using Product #IB03001 from Indigo Biosciences. Th...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMAssay Description:Measurement of active human Factor Xa using a specific chromogenic substrate which is labeled with a p-nitro-anilino group (pNA). The cleavage in bet...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMAssay Description:Measurement of active human Factor Xa using a specific chromogenic substrate which is labeled with a p-nitro-anilino group (pNA). The cleavage in bet...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMAssay Description:Measurement of active human Factor Xa using a specific chromogenic substrate which is labeled with a p-nitro-anilino group (pNA). The cleavage in bet...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMAssay Description:Measurement of active human Factor Xa using a specific chromogenic substrate which is labeled with a p-nitro-anilino group (pNA). The cleavage in bet...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMAssay Description:Measurement of active human Factor Xa using a specific chromogenic substrate which is labeled with a p-nitro-anilino group (pNA). The cleavage in bet...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMAssay Description:Measurement of active human Factor Xa using a specific chromogenic substrate which is labeled with a p-nitro-anilino group (pNA). The cleavage in bet...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMAssay Description:Measurement of active human Factor Xa using a specific chromogenic substrate which is labeled with a p-nitro-anilino group (pNA). The cleavage in bet...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMAssay Description:Measurement of active human Factor Xa using a specific chromogenic substrate which is labeled with a p-nitro-anilino group (pNA). The cleavage in bet...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMAssay Description:Measurement of active human Factor Xa using a specific chromogenic substrate which is labeled with a p-nitro-anilino group (pNA). The cleavage in bet...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMAssay Description:Measurement of active human Factor Xa using a specific chromogenic substrate which is labeled with a p-nitro-anilino group (pNA). The cleavage in bet...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMAssay Description:Measurement of active human Factor Xa using a specific chromogenic substrate which is labeled with a p-nitro-anilino group (pNA). The cleavage in bet...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMAssay Description:Measurement of active human Factor Xa using a specific chromogenic substrate which is labeled with a p-nitro-anilino group (pNA). The cleavage in bet...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMAssay Description:Measurement of active human Factor Xa using a specific chromogenic substrate which is labeled with a p-nitro-anilino group (pNA). The cleavage in bet...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMAssay Description:Measurement of active human Factor Xa using a specific chromogenic substrate which is labeled with a p-nitro-anilino group (pNA). The cleavage in bet...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMAssay Description:Measurement of active human Factor Xa using a specific chromogenic substrate which is labeled with a p-nitro-anilino group (pNA). The cleavage in bet...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMAssay Description:Measurement of active human Factor Xa using a specific chromogenic substrate which is labeled with a p-nitro-anilino group (pNA). The cleavage in bet...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMAssay Description:Measurement of active human Factor Xa using a specific chromogenic substrate which is labeled with a p-nitro-anilino group (pNA). The cleavage in bet...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMAssay Description:Measurement of active human Factor Xa using a specific chromogenic substrate which is labeled with a p-nitro-anilino group (pNA). The cleavage in bet...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMAssay Description:Measurement of active human Factor Xa using a specific chromogenic substrate which is labeled with a p-nitro-anilino group (pNA). The cleavage in bet...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMAssay Description:Measurement of active human Factor Xa using a specific chromogenic substrate which is labeled with a p-nitro-anilino group (pNA). The cleavage in bet...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMAssay Description:Measurement of active human Factor Xa using a specific chromogenic substrate which is labeled with a p-nitro-anilino group (pNA). The cleavage in bet...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMAssay Description:Measurement of active human Factor Xa using a specific chromogenic substrate which is labeled with a p-nitro-anilino group (pNA). The cleavage in bet...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMAssay Description:Measurement of active human Factor Xa using a specific chromogenic substrate which is labeled with a p-nitro-anilino group (pNA). The cleavage in bet...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMAssay Description:Measurement of active human Factor Xa using a specific chromogenic substrate which is labeled with a p-nitro-anilino group (pNA). The cleavage in bet...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMAssay Description:Determination of In Vitro Pharmacological Activity. The human androgen receptor (AR) assay was run using Product #IB03001 from Indigo Biosciences. Th...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMAssay Description:Determination of In Vitro Pharmacological Activity. The human androgen receptor (AR) assay was run using Product #IB03001 from Indigo Biosciences. Th...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMAssay Description:Determination of In Vitro Pharmacological Activity. The human androgen receptor (AR) assay was run using Product #IB03001 from Indigo Biosciences. Th...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMAssay Description:Measurement of active human Factor Xa using a specific chromogenic substrate which is labeled with a p-nitro-anilino group (pNA). The cleavage in bet...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMAssay Description:Measurement of active human Factor Xa using a specific chromogenic substrate which is labeled with a p-nitro-anilino group (pNA). The cleavage in bet...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMAssay Description:Measurement of active human Factor Xa using a specific chromogenic substrate which is labeled with a p-nitro-anilino group (pNA). The cleavage in bet...More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMAssay Description:Measurement of active human Factor Xa using a specific chromogenic substrate which is labeled with a p-nitro-anilino group (pNA). The cleavage in bet...More data for this Ligand-Target Pair