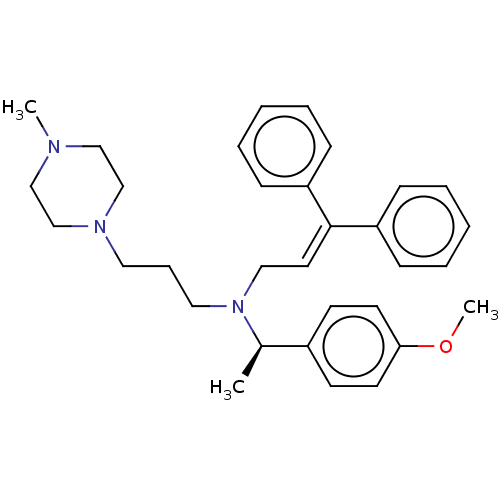
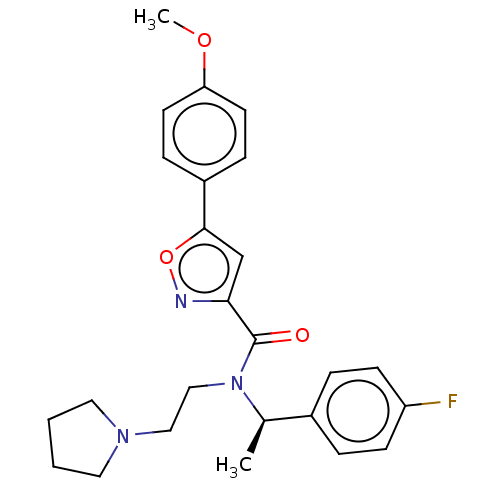
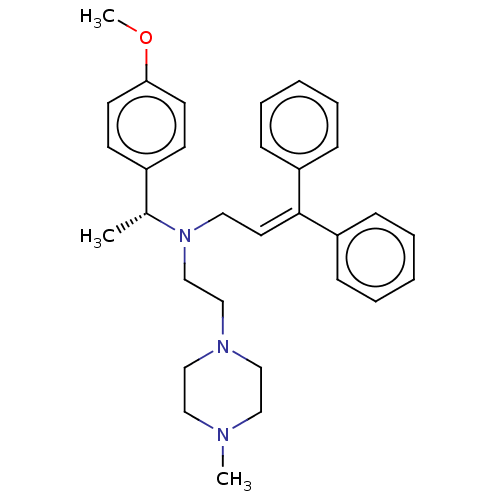
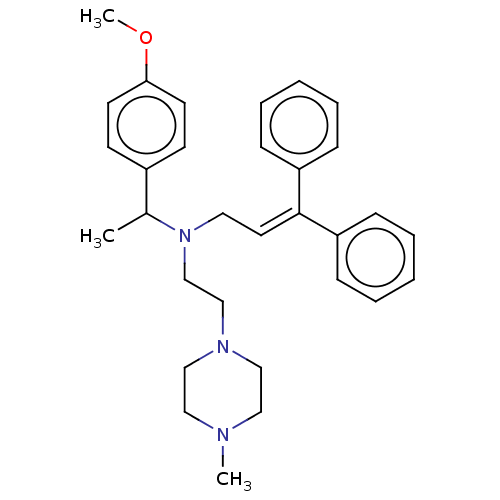
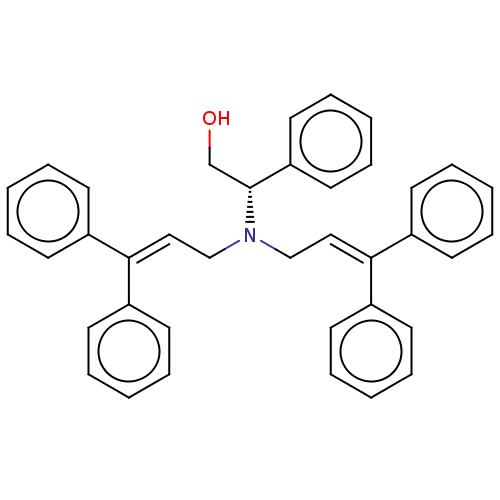
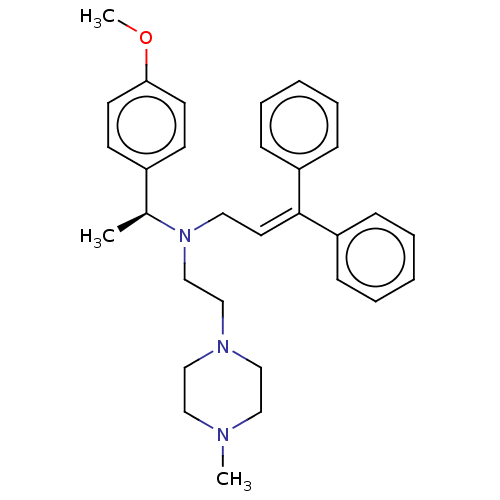
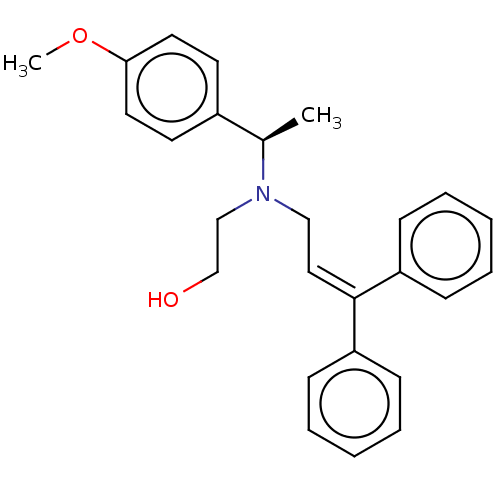
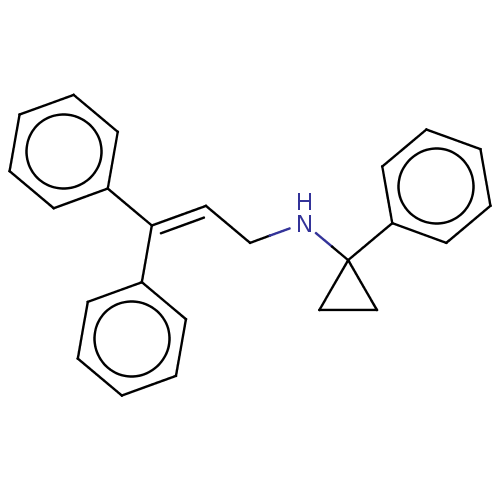
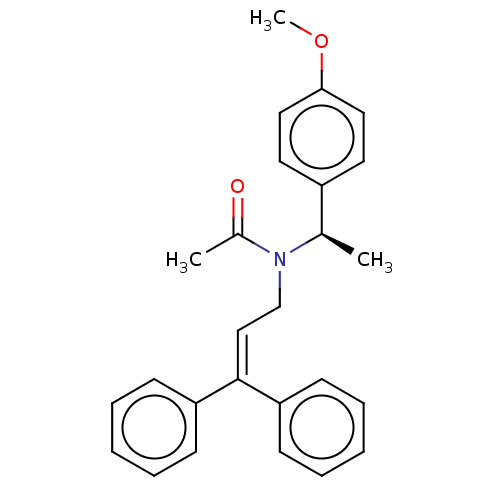
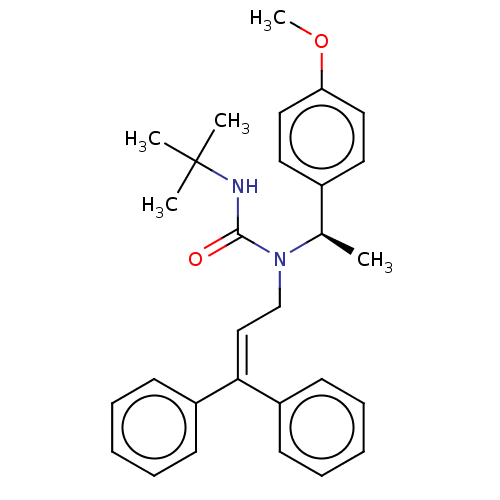
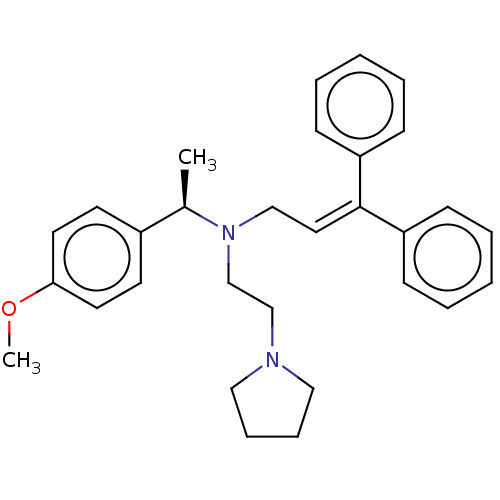
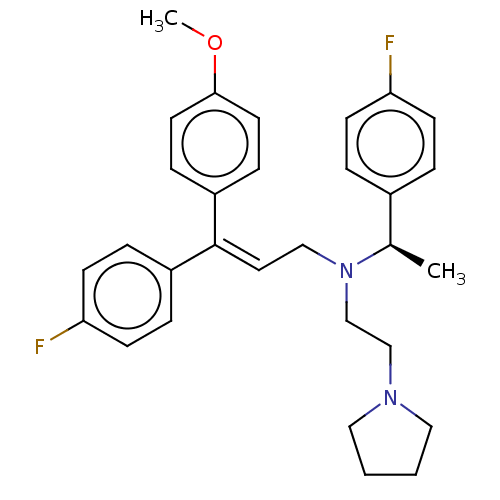
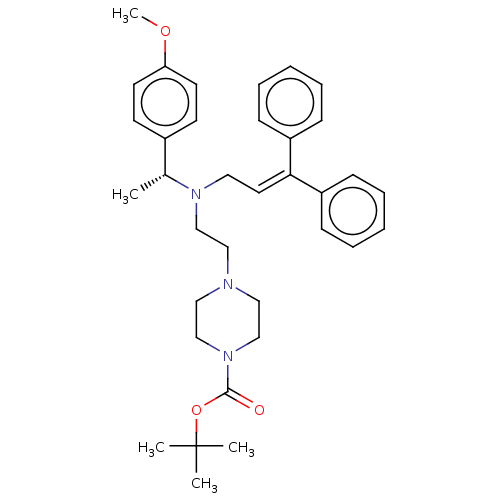
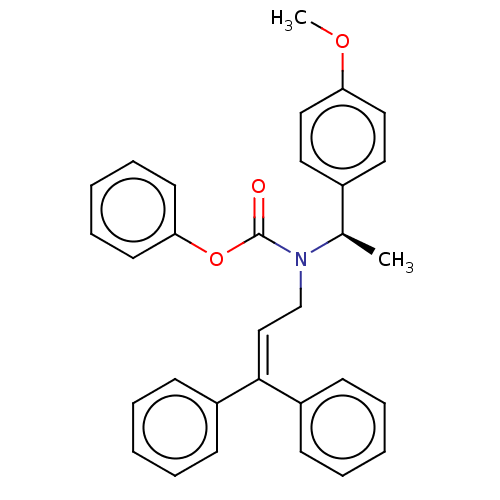
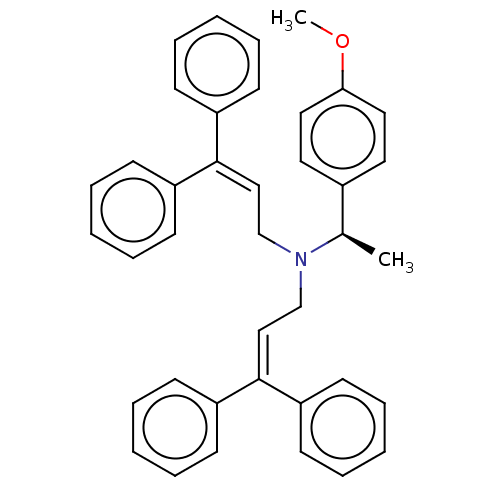
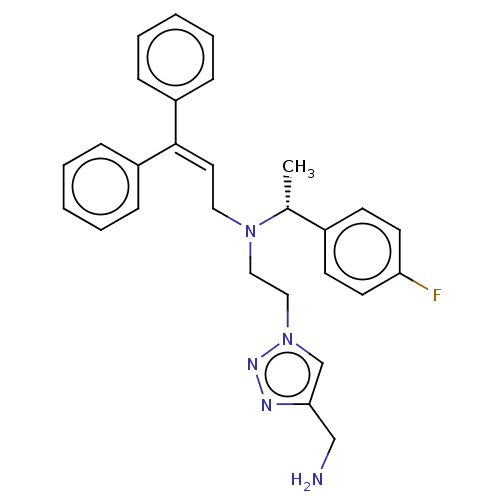
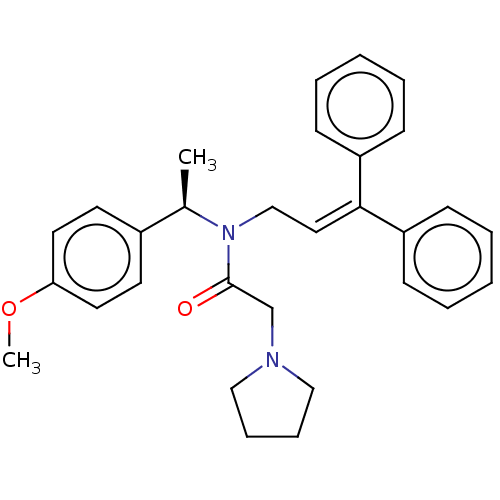
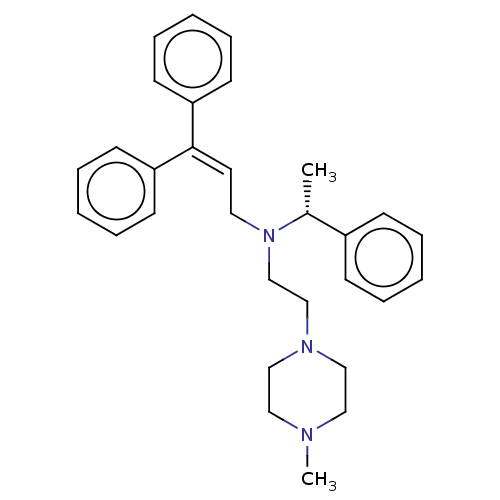
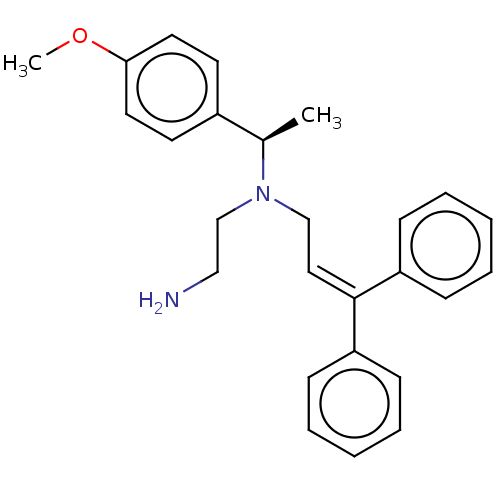
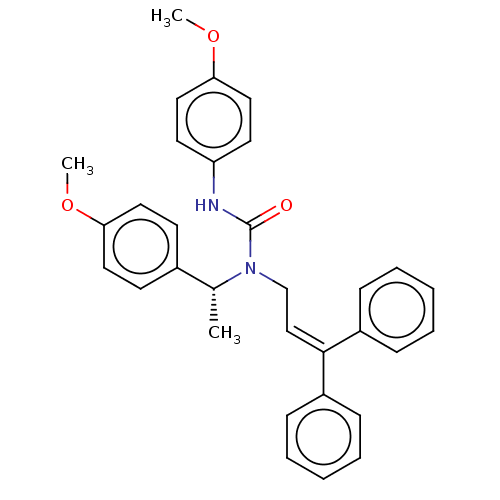
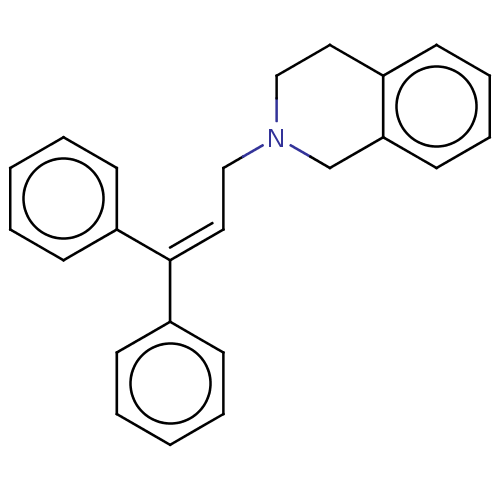
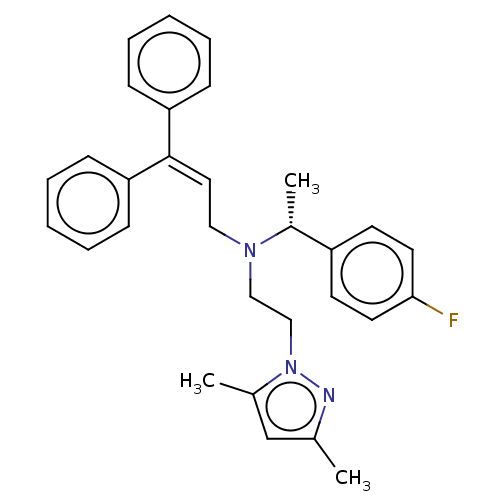
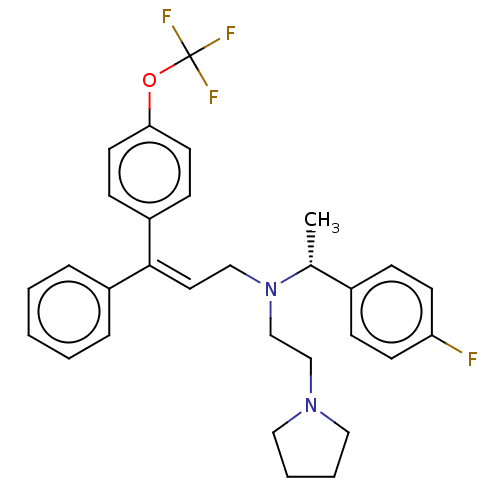
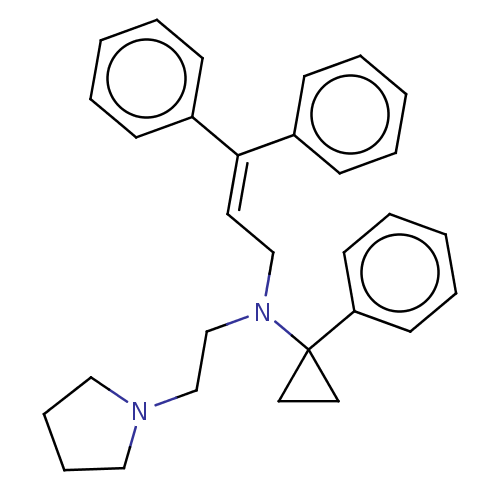
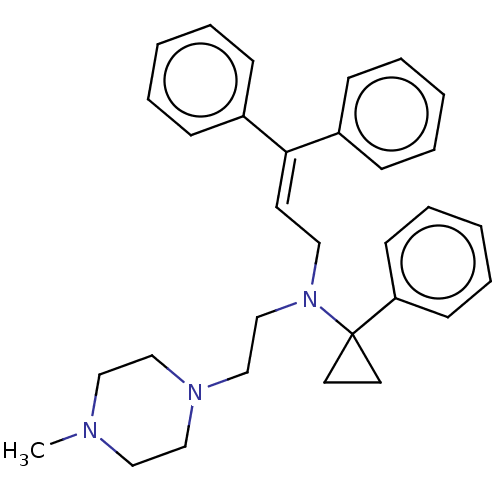
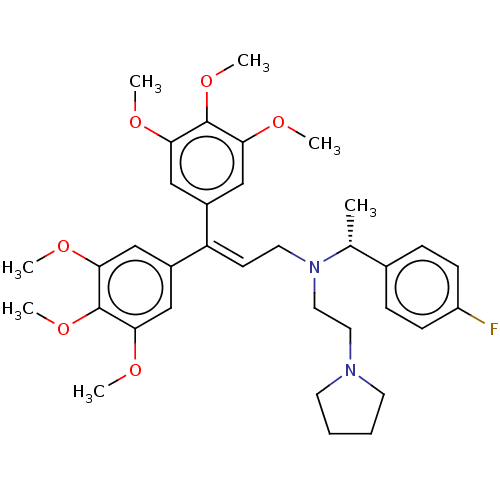
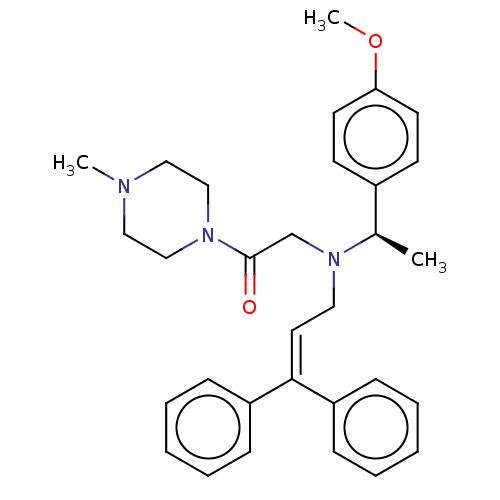
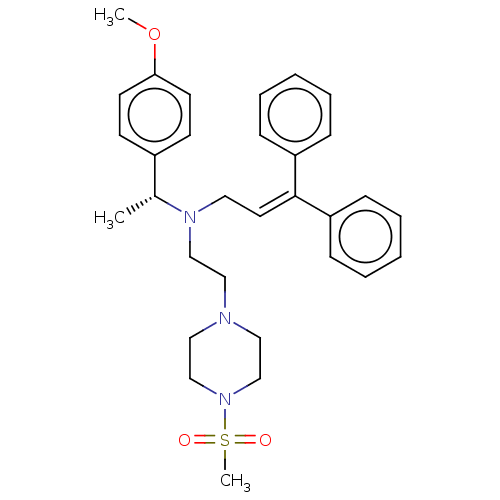
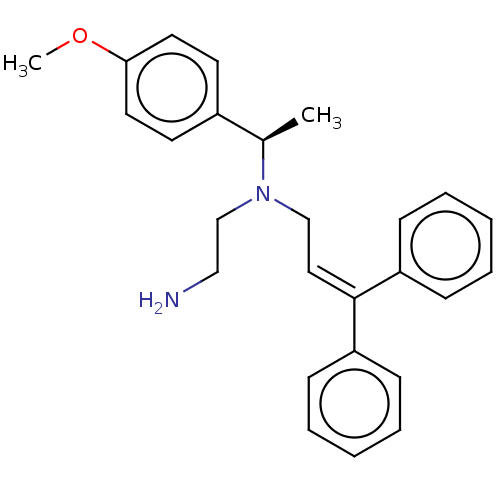
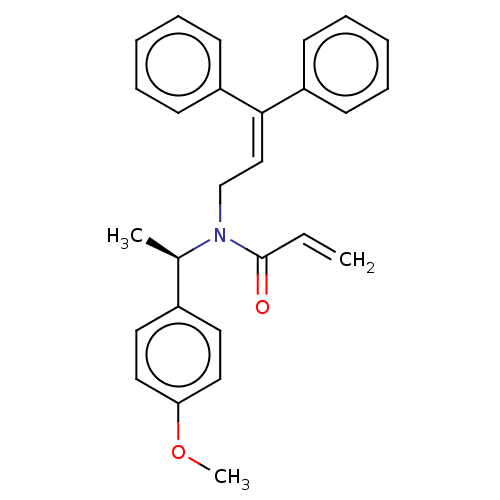
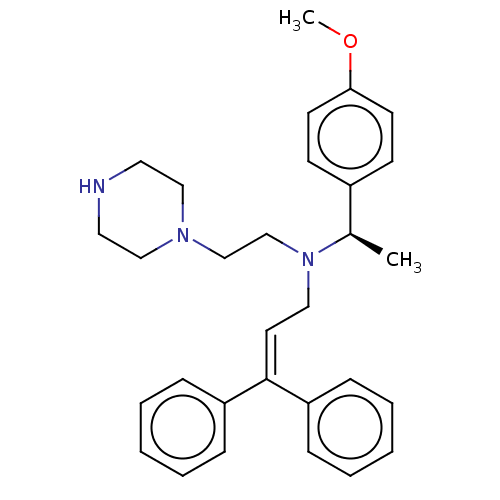
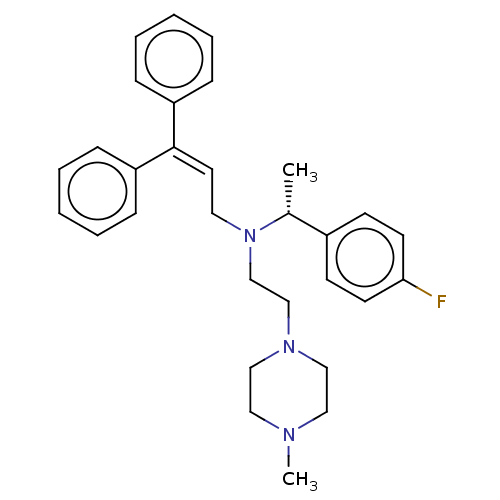
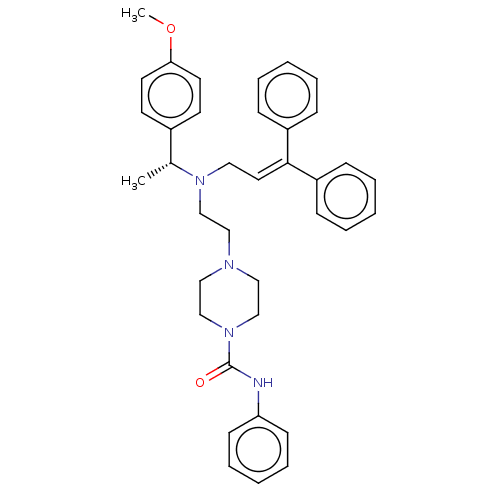
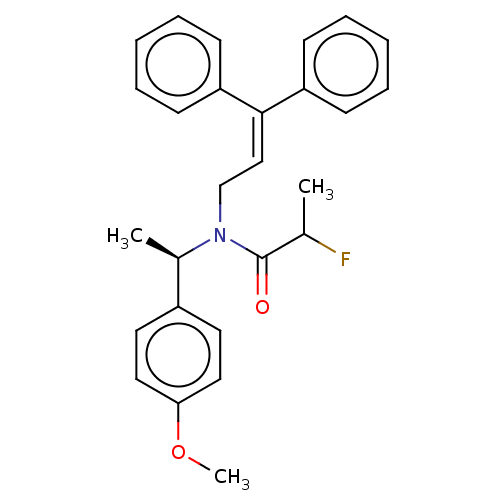
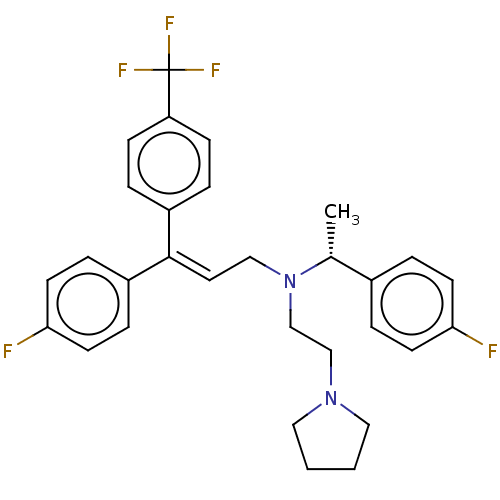
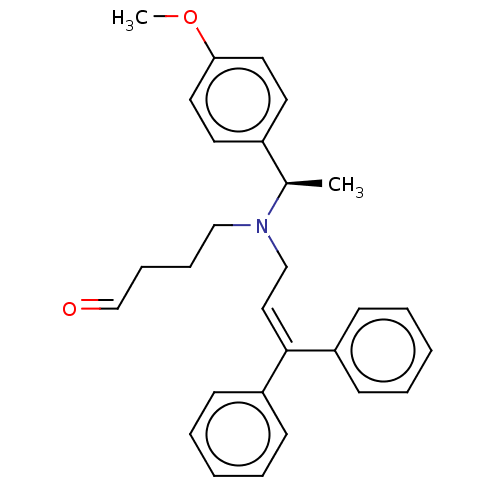
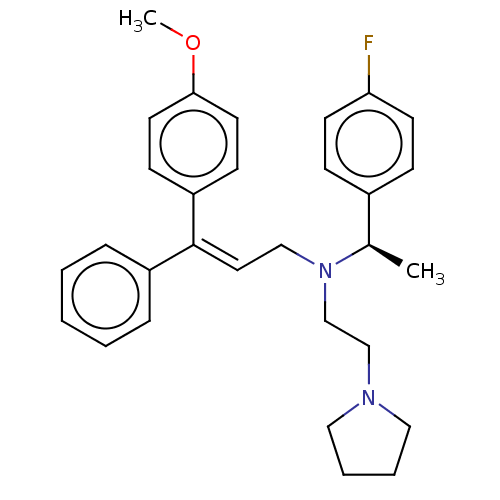
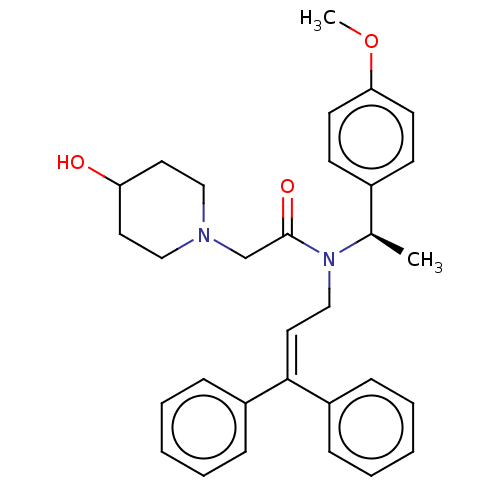
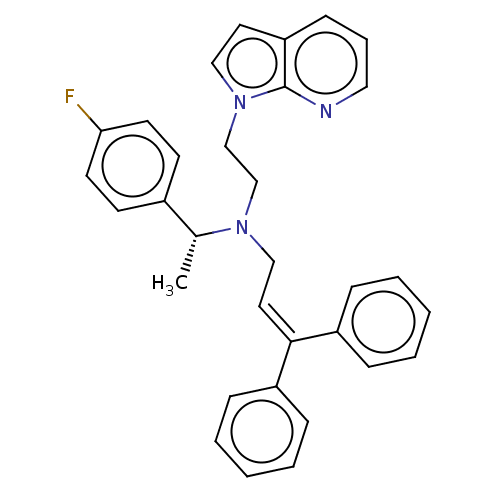
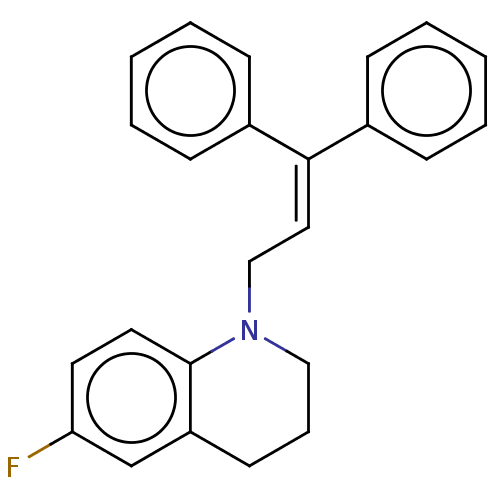
Report error Found 110 Enz. Inhib. hit(s) with all data for entry = 12198
Affinity DataIC50: 20nMAssay Description:Table 1: Potency and efficacy of K-RAS inhibition and toxicity of batch1 and 2 compounds. MDCK cells co-expressing GFP-K-RASG12V and mCherry-CAAX wer...More data for this Ligand-Target Pair
Affinity DataIC50: 50nMAssay Description:Table 7: Potency and efficacy of K-RAS inhibition and toxicity ofbatch 5 compounds. MDCK cells co-expressing GFP-K-RASG12V andmCherry-CAAX were grown...More data for this Ligand-Target Pair
Affinity DataIC50: 100nMAssay Description:Table 10: IC50: 50% inhibitory concentration for KRASG12V mislocalization. Values were calculated from at least eight data points. In general, at lea...More data for this Ligand-Target Pair
Affinity DataIC50: 100nMAssay Description:Table 1: Potency and efficacy of K-RAS inhibition and toxicity of batch1 and 2 compounds. MDCK cells co-expressing GFP-K-RASG12V and mCherry-CAAX wer...More data for this Ligand-Target Pair
Affinity DataIC50: 100nMAssay Description:Table 9: IC50: 50% inhibitory concentration for KRASG12V mislocalization. Values were calculated from at least eight data points. In general, at leas...More data for this Ligand-Target Pair
Affinity DataIC50: 100nMAssay Description:Table 1: Potency and efficacy of K-RAS inhibition and toxicity of batch1 and 2 compounds. MDCK cells co-expressing GFP-K-RASG12V and mCherry-CAAX wer...More data for this Ligand-Target Pair
Affinity DataIC50: 160nMAssay Description:Table 1: Potency and efficacy of K-RAS inhibition and toxicity of batch1 and 2 compounds. MDCK cells co-expressing GFP-K-RASG12V and mCherry-CAAX wer...More data for this Ligand-Target Pair
Affinity DataIC50: 170nMAssay Description:Table 1: Potency and efficacy of K-RAS inhibition and toxicity of batch1 and 2 compounds. MDCK cells co-expressing GFP-K-RASG12V and mCherry-CAAX wer...More data for this Ligand-Target Pair
Affinity DataIC50: 170nMAssay Description:Table 1: Potency and efficacy of K-RAS inhibition and toxicity of batch1 and 2 compounds. MDCK cells co-expressing GFP-K-RASG12V and mCherry-CAAX wer...More data for this Ligand-Target Pair
Affinity DataIC50: 190nMAssay Description:Table 1: Potency and efficacy of K-RAS inhibition and toxicity of batch1 and 2 compounds. MDCK cells co-expressing GFP-K-RASG12V and mCherry-CAAX wer...More data for this Ligand-Target Pair
Affinity DataIC50: 200nMAssay Description:Table 1: Potency and efficacy of K-RAS inhibition and toxicity of batch1 and 2 compounds. MDCK cells co-expressing GFP-K-RASG12V and mCherry-CAAX wer...More data for this Ligand-Target Pair
Affinity DataIC50: 200nMAssay Description:Table 3: Potency and efficacy K-RAS inhibition and toxicity of batch 3 compounds. MDCK cells co-expressing GFP-K-RASG12V and mCherry-CAAX were grown ...More data for this Ligand-Target Pair
Affinity DataIC50: 250nMAssay Description:Table 1: Potency and efficacy of K-RAS inhibition and toxicity of batch1 and 2 compounds. MDCK cells co-expressing GFP-K-RASG12V and mCherry-CAAX wer...More data for this Ligand-Target Pair
Affinity DataIC50: 290nMAssay Description:Table 3: Potency and efficacy K-RAS inhibition and toxicity of batch 3 compounds. MDCK cells co-expressing GFP-K-RASG12V and mCherry-CAAX were grown ...More data for this Ligand-Target Pair
Affinity DataIC50: 300nMAssay Description:Table 1: Potency and efficacy of K-RAS inhibition and toxicity of batch1 and 2 compounds. MDCK cells co-expressing GFP-K-RASG12V and mCherry-CAAX wer...More data for this Ligand-Target Pair
Affinity DataIC50: 319nMAssay Description:Table 7: Potency and efficacy of K-RAS inhibition and toxicity ofbatch 5 compounds. MDCK cells co-expressing GFP-K-RASG12V andmCherry-CAAX were grown...More data for this Ligand-Target Pair
Affinity DataIC50: 329nMAssay Description:Table 1: Potency and efficacy of K-RAS inhibition and toxicity of batch1 and 2 compounds. MDCK cells co-expressing GFP-K-RASG12V and mCherry-CAAX wer...More data for this Ligand-Target Pair
Affinity DataIC50: 420nMAssay Description:Table 1: Potency and efficacy of K-RAS inhibition and toxicity of batch1 and 2 compounds. MDCK cells co-expressing GFP-K-RASG12V and mCherry-CAAX wer...More data for this Ligand-Target Pair
Affinity DataIC50: 430nMAssay Description:Table 1: Potency and efficacy of K-RAS inhibition and toxicity of batch1 and 2 compounds. MDCK cells co-expressing GFP-K-RASG12V and mCherry-CAAX wer...More data for this Ligand-Target Pair
Affinity DataIC50: 500nMAssay Description:Table 10: IC50: 50% inhibitory concentration for KRASG12V mislocalization. Values were calculated from at least eight data points. In general, at lea...More data for this Ligand-Target Pair
Affinity DataIC50: 500nMAssay Description:Table 9: IC50: 50% inhibitory concentration for KRASG12V mislocalization. Values were calculated from at least eight data points. In general, at leas...More data for this Ligand-Target Pair
Affinity DataIC50: 507nMAssay Description:Table 7: Potency and efficacy of K-RAS inhibition and toxicity ofbatch 5 compounds. MDCK cells co-expressing GFP-K-RASG12V andmCherry-CAAX were grown...More data for this Ligand-Target Pair
Affinity DataIC50: 553nMAssay Description:Table 7: Potency and efficacy of K-RAS inhibition and toxicity ofbatch 5 compounds. MDCK cells co-expressing GFP-K-RASG12V andmCherry-CAAX were grown...More data for this Ligand-Target Pair
Affinity DataIC50: 600nMAssay Description:Table 9: IC50: 50% inhibitory concentration for KRASG12V mislocalization. Values were calculated from at least eight data points. In general, at leas...More data for this Ligand-Target Pair
Affinity DataIC50: 600nMAssay Description:Table 1: Potency and efficacy of K-RAS inhibition and toxicity of batch1 and 2 compounds. MDCK cells co-expressing GFP-K-RASG12V and mCherry-CAAX wer...More data for this Ligand-Target Pair
Affinity DataIC50: 640nMAssay Description:Table 1: Potency and efficacy of K-RAS inhibition and toxicity of batch1 and 2 compounds. MDCK cells co-expressing GFP-K-RASG12V and mCherry-CAAX wer...More data for this Ligand-Target Pair
Affinity DataIC50: 650nMAssay Description:Table 1: Potency and efficacy of K-RAS inhibition and toxicity of batch1 and 2 compounds. MDCK cells co-expressing GFP-K-RASG12V and mCherry-CAAX wer...More data for this Ligand-Target Pair
Affinity DataIC50: 654nMAssay Description:Table 7: Potency and efficacy of K-RAS inhibition and toxicity ofbatch 5 compounds. MDCK cells co-expressing GFP-K-RASG12V andmCherry-CAAX were grown...More data for this Ligand-Target Pair
Affinity DataIC50: 725nMAssay Description:Table 7: Potency and efficacy of K-RAS inhibition and toxicity ofbatch 5 compounds. MDCK cells co-expressing GFP-K-RASG12V andmCherry-CAAX were grown...More data for this Ligand-Target Pair
Affinity DataIC50: 730nMAssay Description:Table 3: Potency and efficacy K-RAS inhibition and toxicity of batch 3 compounds. MDCK cells co-expressing GFP-K-RASG12V and mCherry-CAAX were grown ...More data for this Ligand-Target Pair
Affinity DataIC50: 910nMAssay Description:Table 3: Potency and efficacy K-RAS inhibition and toxicity of batch 3 compounds. MDCK cells co-expressing GFP-K-RASG12V and mCherry-CAAX were grown ...More data for this Ligand-Target Pair
Affinity DataIC50: 1.02E+3nMAssay Description:Table 7: Potency and efficacy of K-RAS inhibition and toxicity ofbatch 5 compounds. MDCK cells co-expressing GFP-K-RASG12V andmCherry-CAAX were grown...More data for this Ligand-Target Pair
Affinity DataIC50: 1.10E+3nMAssay Description:Table 1: Potency and efficacy of K-RAS inhibition and toxicity of batch1 and 2 compounds. MDCK cells co-expressing GFP-K-RASG12V and mCherry-CAAX wer...More data for this Ligand-Target Pair
Affinity DataIC50: 1.11E+3nMAssay Description:Table 1: Potency and efficacy of K-RAS inhibition and toxicity of batch1 and 2 compounds. MDCK cells co-expressing GFP-K-RASG12V and mCherry-CAAX wer...More data for this Ligand-Target Pair
Affinity DataIC50: 1.15E+3nMAssay Description:Table 3: Potency and efficacy K-RAS inhibition and toxicity of batch 3 compounds. MDCK cells co-expressing GFP-K-RASG12V and mCherry-CAAX were grown ...More data for this Ligand-Target Pair
Affinity DataIC50: 1.20E+3nMAssay Description:Table 9: IC50: 50% inhibitory concentration for KRASG12V mislocalization. Values were calculated from at least eight data points. In general, at leas...More data for this Ligand-Target Pair
Affinity DataIC50: 1.21E+3nMAssay Description:Table 1: Potency and efficacy of K-RAS inhibition and toxicity of batch1 and 2 compounds. MDCK cells co-expressing GFP-K-RASG12V and mCherry-CAAX wer...More data for this Ligand-Target Pair
Affinity DataIC50: 1.24E+3nMAssay Description:Table 3: Potency and efficacy K-RAS inhibition and toxicity of batch 3 compounds. MDCK cells co-expressing GFP-K-RASG12V and mCherry-CAAX were grown ...More data for this Ligand-Target Pair
Affinity DataIC50: 1.30E+3nMAssay Description:Table 1: Potency and efficacy of K-RAS inhibition and toxicity of batch1 and 2 compounds. MDCK cells co-expressing GFP-K-RASG12V and mCherry-CAAX wer...More data for this Ligand-Target Pair
Affinity DataIC50: 1.46E+3nMAssay Description:Table 3: Potency and efficacy K-RAS inhibition and toxicity of batch 3 compounds. MDCK cells co-expressing GFP-K-RASG12V and mCherry-CAAX were grown ...More data for this Ligand-Target Pair
Affinity DataIC50: 1.50E+3nMAssay Description:Table 9: IC50: 50% inhibitory concentration for KRASG12V mislocalization. Values were calculated from at least eight data points. In general, at leas...More data for this Ligand-Target Pair
Affinity DataIC50: 1.68E+3nMAssay Description:Table 1: Potency and efficacy of K-RAS inhibition and toxicity of batch1 and 2 compounds. MDCK cells co-expressing GFP-K-RASG12V and mCherry-CAAX wer...More data for this Ligand-Target Pair
Affinity DataIC50: 1.69E+3nMAssay Description:Table 1: Potency and efficacy of K-RAS inhibition and toxicity of batch1 and 2 compounds. MDCK cells co-expressing GFP-K-RASG12V and mCherry-CAAX wer...More data for this Ligand-Target Pair
Affinity DataIC50: 1.76E+3nMAssay Description:Table 1: Potency and efficacy of K-RAS inhibition and toxicity of batch1 and 2 compounds. MDCK cells co-expressing GFP-K-RASG12V and mCherry-CAAX wer...More data for this Ligand-Target Pair
Affinity DataIC50: 1.86E+3nMAssay Description:Table 7: Potency and efficacy of K-RAS inhibition and toxicity ofbatch 5 compounds. MDCK cells co-expressing GFP-K-RASG12V andmCherry-CAAX were grown...More data for this Ligand-Target Pair
Affinity DataIC50: 2.02E+3nMAssay Description:Table 3: Potency and efficacy K-RAS inhibition and toxicity of batch 3 compounds. MDCK cells co-expressing GFP-K-RASG12V and mCherry-CAAX were grown ...More data for this Ligand-Target Pair
Affinity DataIC50: 2.02E+3nMAssay Description:Table 7: Potency and efficacy of K-RAS inhibition and toxicity ofbatch 5 compounds. MDCK cells co-expressing GFP-K-RASG12V andmCherry-CAAX were grown...More data for this Ligand-Target Pair
Affinity DataIC50: 2.19E+3nMAssay Description:Table 7: Potency and efficacy of K-RAS inhibition and toxicity ofbatch 5 compounds. MDCK cells co-expressing GFP-K-RASG12V andmCherry-CAAX were grown...More data for this Ligand-Target Pair
Affinity DataIC50: 2.27E+3nMAssay Description:Table 7: Potency and efficacy of K-RAS inhibition and toxicity ofbatch 5 compounds. MDCK cells co-expressing GFP-K-RASG12V andmCherry-CAAX were grown...More data for this Ligand-Target Pair
Affinity DataIC50: 2.30E+3nMAssay Description:Table 1: Potency and efficacy of K-RAS inhibition and toxicity of batch1 and 2 compounds. MDCK cells co-expressing GFP-K-RASG12V and mCherry-CAAX wer...More data for this Ligand-Target Pair