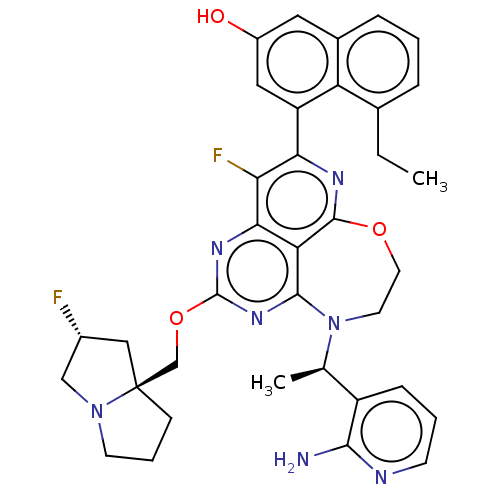
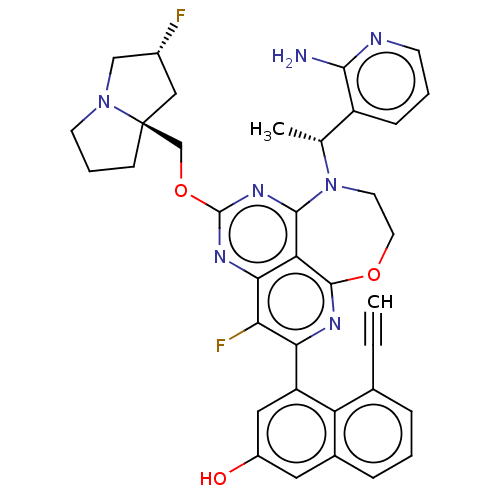
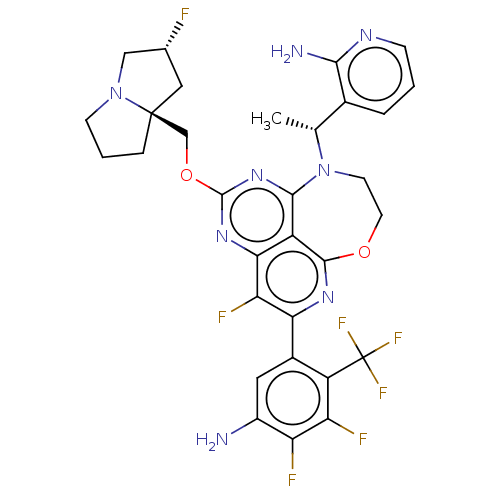
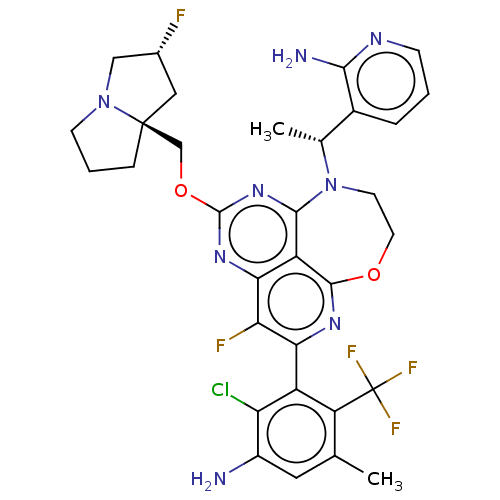
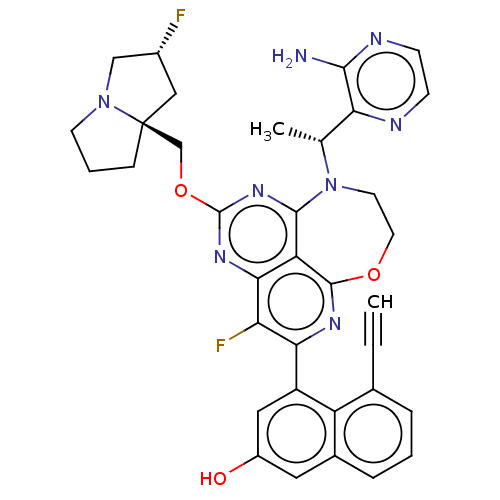
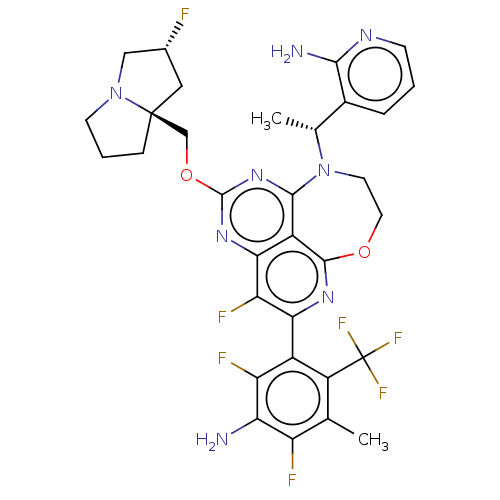
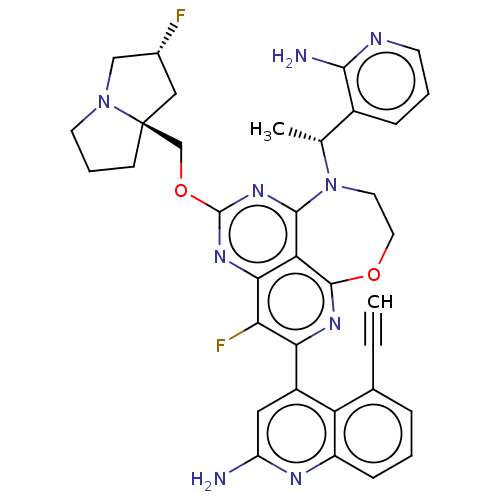
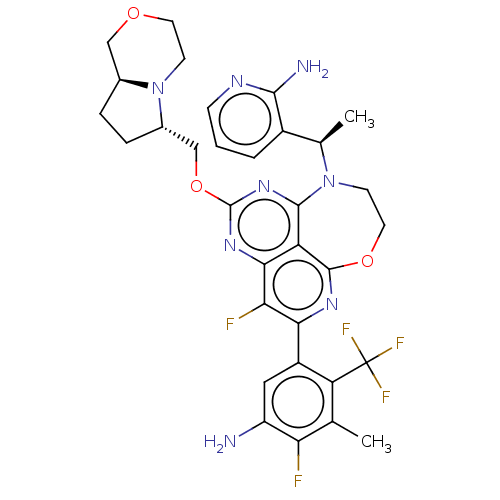
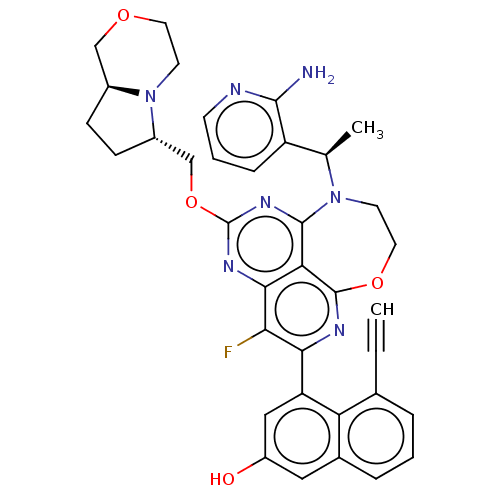
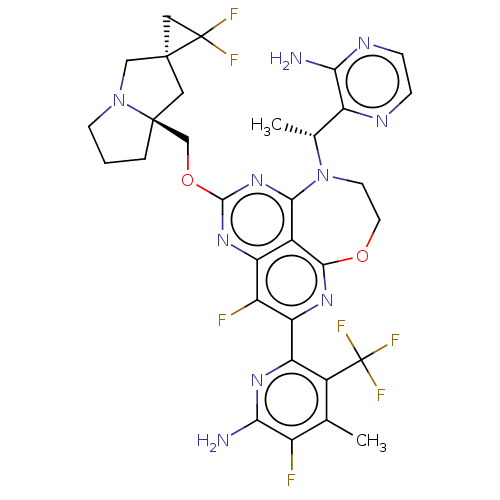
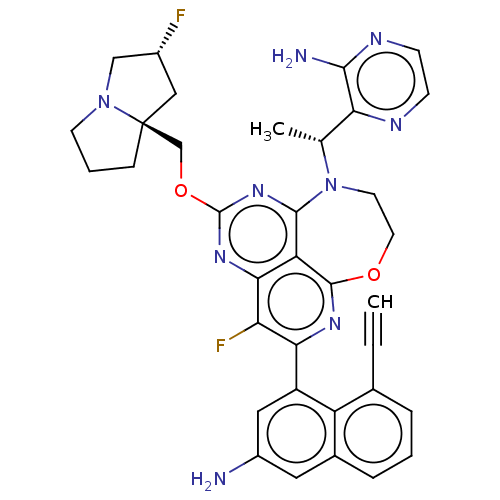
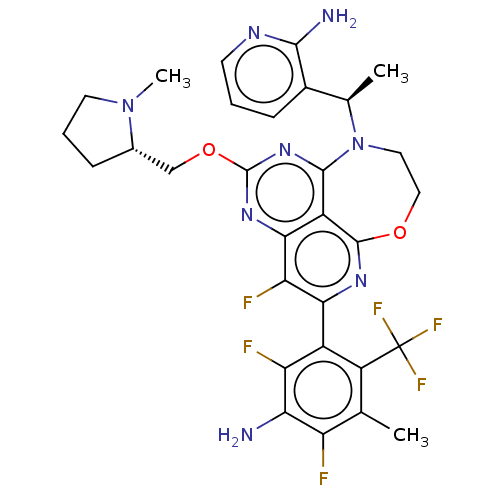
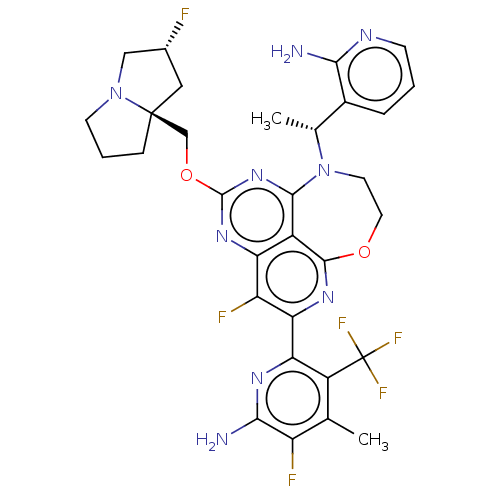
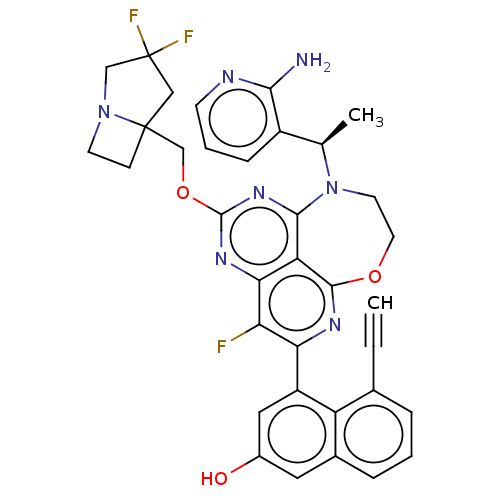
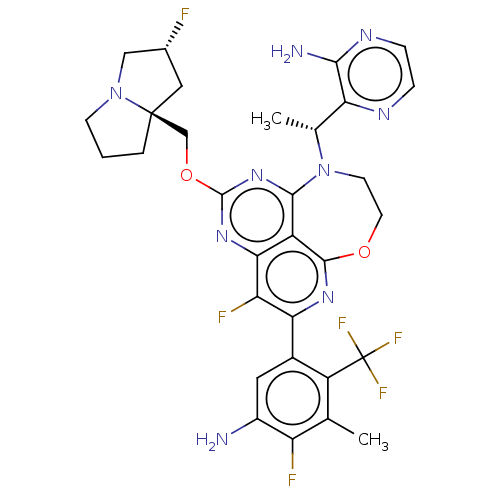
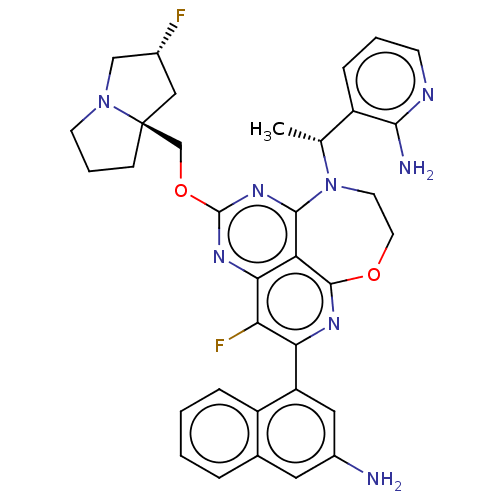
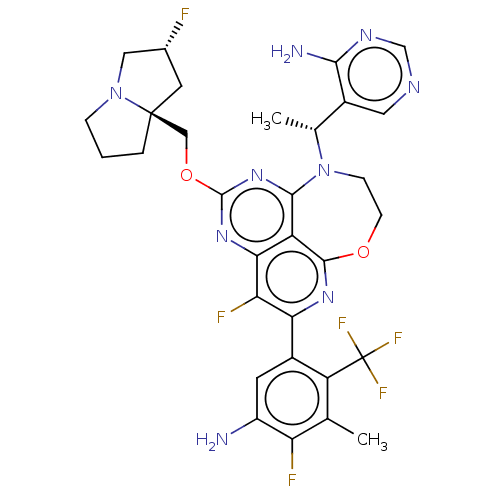
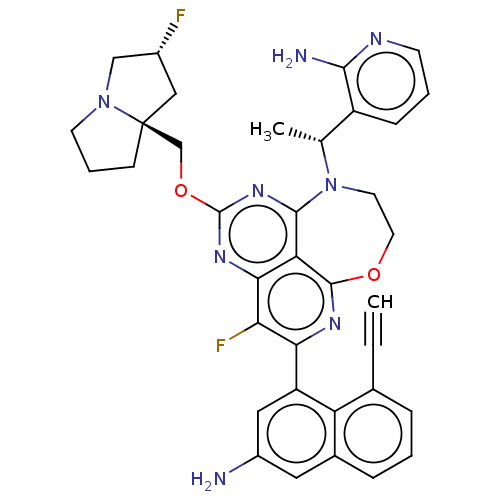
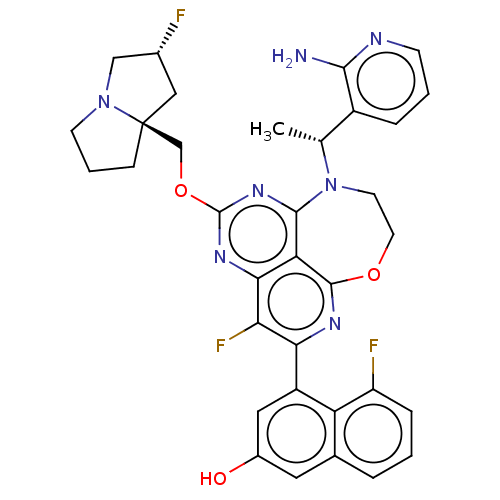
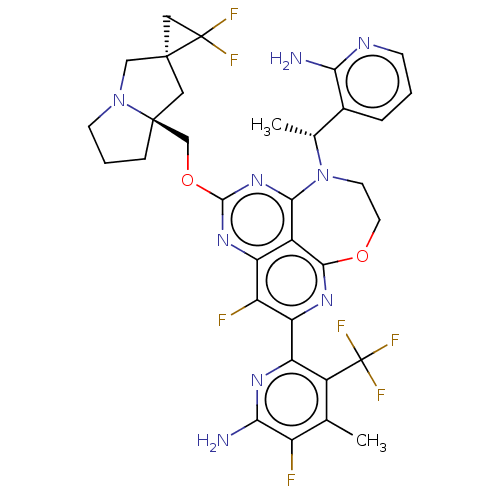
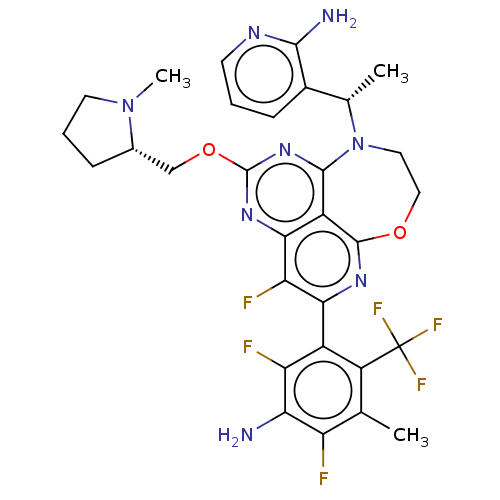
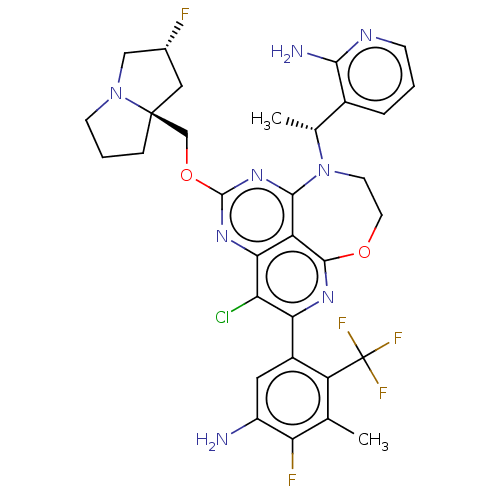
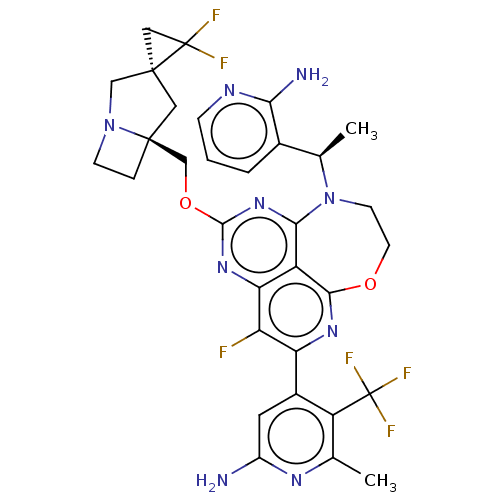
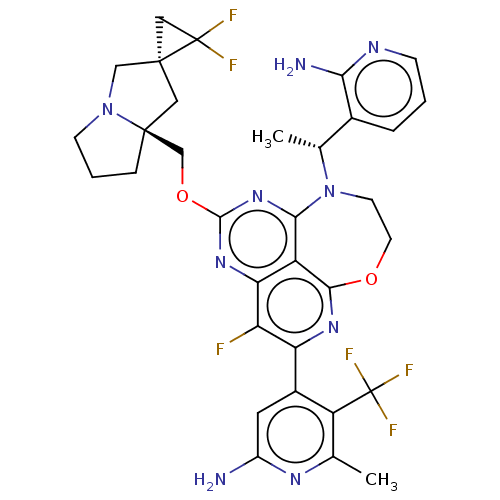
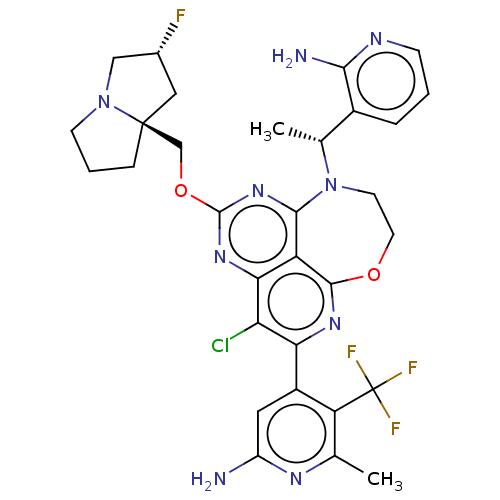
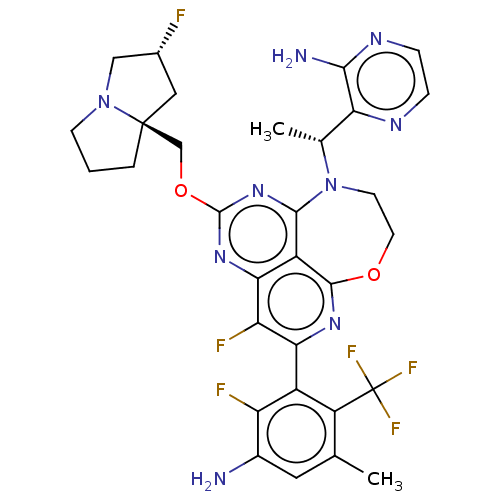
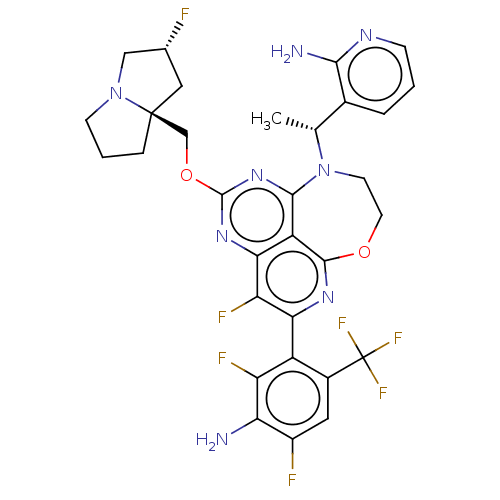
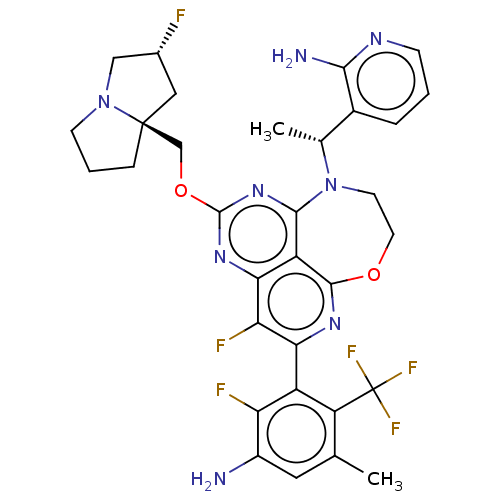
Report error Found 140 Enz. Inhib. hit(s) with all data for entry = 12185
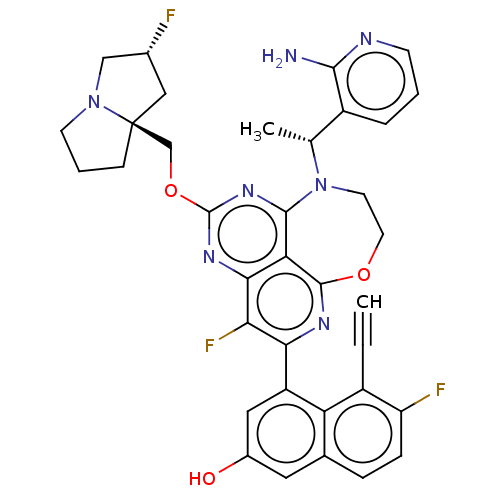
Affinity DataIC50: 0.0400nMAssay Description:Biochemical compound potencies were assessed by evaluating inhibition of SOS1-mediated nucleotide exchange in KRAS G12D. The SOS1-promoted exchange o...More data for this Ligand-Target Pair
Affinity DataIC50: 0.0400nMAssay Description:Biochemical compound potencies were assessed by evaluating inhibition of SOS1-mediated nucleotide exchange in KRAS G12D. The SOS1-promoted exchange o...More data for this Ligand-Target Pair
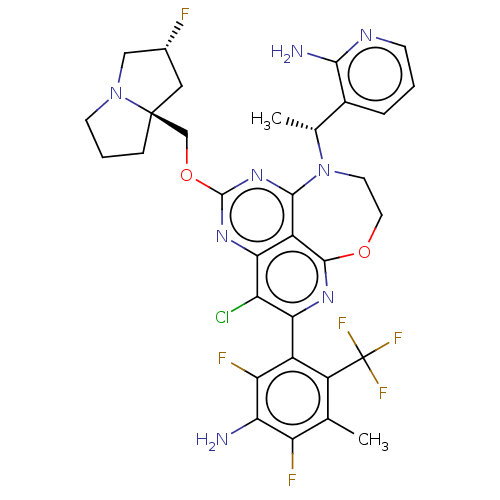
Affinity DataIC50: 0.0450nMAssay Description:Biochemical compound potencies were assessed by evaluating inhibition of SOS1-mediated nucleotide exchange in KRAS G12D. The SOS1-promoted exchange o...More data for this Ligand-Target Pair
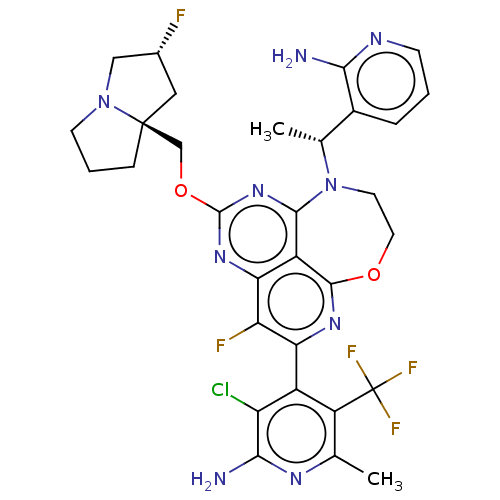
Affinity DataIC50: 0.0500nMAssay Description:Biochemical compound potencies were assessed by evaluating inhibition of SOS1-mediated nucleotide exchange in KRAS G12D. The SOS1-promoted exchange o...More data for this Ligand-Target Pair
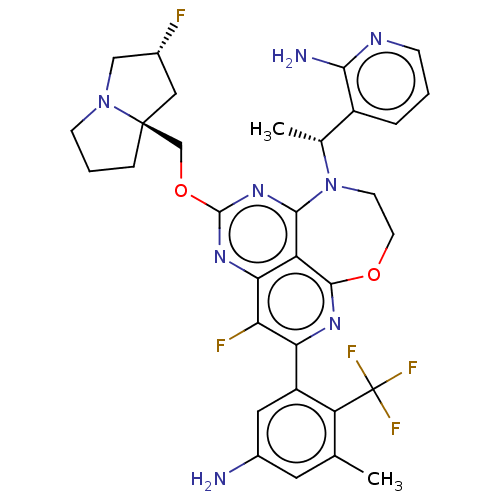
Affinity DataIC50: 0.0500nMAssay Description:Biochemical compound potencies were assessed by evaluating inhibition of SOS1-mediated nucleotide exchange in KRAS G12D. The SOS1-promoted exchange o...More data for this Ligand-Target Pair
Affinity DataIC50: 0.0530nMAssay Description:Biochemical compound potencies were assessed by evaluating inhibition of SOS1-mediated nucleotide exchange in KRAS G12D. The SOS1-promoted exchange o...More data for this Ligand-Target Pair
Affinity DataIC50: 0.0600nMAssay Description:Biochemical compound potencies were assessed by evaluating inhibition of SOS1-mediated nucleotide exchange in KRAS G12D. The SOS1-promoted exchange o...More data for this Ligand-Target Pair
Affinity DataIC50: 0.0600nMAssay Description:Biochemical compound potencies were assessed by evaluating inhibition of SOS1-mediated nucleotide exchange in KRAS G12D. The SOS1-promoted exchange o...More data for this Ligand-Target Pair
Affinity DataIC50: 0.0600nMAssay Description:Biochemical compound potencies were assessed by evaluating inhibition of SOS1-mediated nucleotide exchange in KRAS G12D. The SOS1-promoted exchange o...More data for this Ligand-Target Pair
Affinity DataIC50: 0.0600nMAssay Description:Biochemical compound potencies were assessed by evaluating inhibition of SOS1-mediated nucleotide exchange in KRAS G12D. The SOS1-promoted exchange o...More data for this Ligand-Target Pair
Affinity DataIC50: 0.0670nMAssay Description:Biochemical compound potencies were assessed by evaluating inhibition of SOS1-mediated nucleotide exchange in KRAS G12D. The SOS1-promoted exchange o...More data for this Ligand-Target Pair
Affinity DataIC50: 0.0690nMAssay Description:Biochemical compound potencies were assessed by evaluating inhibition of SOS1-mediated nucleotide exchange in KRAS G12D. The SOS1-promoted exchange o...More data for this Ligand-Target Pair
Affinity DataIC50: 0.0700nMAssay Description:Biochemical compound potencies were assessed by evaluating inhibition of SOS1-mediated nucleotide exchange in KRAS G12D. The SOS1-promoted exchange o...More data for this Ligand-Target Pair
Affinity DataIC50: 0.0700nMAssay Description:Biochemical compound potencies were assessed by evaluating inhibition of SOS1-mediated nucleotide exchange in KRAS G12D. The SOS1-promoted exchange o...More data for this Ligand-Target Pair
Affinity DataIC50: 0.0700nMAssay Description:Biochemical compound potencies were assessed by evaluating inhibition of SOS1-mediated nucleotide exchange in KRAS G12D. The SOS1-promoted exchange o...More data for this Ligand-Target Pair
Affinity DataIC50: 0.0800nMAssay Description:Biochemical compound potencies were assessed by evaluating inhibition of SOS1-mediated nucleotide exchange in KRAS G12D. The SOS1-promoted exchange o...More data for this Ligand-Target Pair
Affinity DataIC50: 0.0800nMAssay Description:Biochemical compound potencies were assessed by evaluating inhibition of SOS1-mediated nucleotide exchange in KRAS G12D. The SOS1-promoted exchange o...More data for this Ligand-Target Pair
Affinity DataIC50: 0.0800nMAssay Description:Biochemical compound potencies were assessed by evaluating inhibition of SOS1-mediated nucleotide exchange in KRAS G12D. The SOS1-promoted exchange o...More data for this Ligand-Target Pair
Affinity DataIC50: 0.0890nMAssay Description:Biochemical compound potencies were assessed by evaluating inhibition of SOS1-mediated nucleotide exchange in KRAS G12D. The SOS1-promoted exchange o...More data for this Ligand-Target Pair
Affinity DataIC50: 0.0900nMAssay Description:Biochemical compound potencies were assessed by evaluating inhibition of SOS1-mediated nucleotide exchange in KRAS G12D. The SOS1-promoted exchange o...More data for this Ligand-Target Pair
Affinity DataIC50: 0.0900nMAssay Description:Biochemical compound potencies were assessed by evaluating inhibition of SOS1-mediated nucleotide exchange in KRAS G12D. The SOS1-promoted exchange o...More data for this Ligand-Target Pair
Affinity DataIC50: 0.0900nMAssay Description:Biochemical compound potencies were assessed by evaluating inhibition of SOS1-mediated nucleotide exchange in KRAS G12D. The SOS1-promoted exchange o...More data for this Ligand-Target Pair
Affinity DataIC50: 0.0990nMAssay Description:Biochemical compound potencies were assessed by evaluating inhibition of SOS1-mediated nucleotide exchange in KRAS G12D. The SOS1-promoted exchange o...More data for this Ligand-Target Pair
Affinity DataIC50: 0.100nMAssay Description:Biochemical compound potencies were assessed by evaluating inhibition of SOS1-mediated nucleotide exchange in KRAS G12D. The SOS1-promoted exchange o...More data for this Ligand-Target Pair
Affinity DataIC50: 0.100nMAssay Description:Biochemical compound potencies were assessed by evaluating inhibition of SOS1-mediated nucleotide exchange in KRAS G12D. The SOS1-promoted exchange o...More data for this Ligand-Target Pair
Affinity DataIC50: 0.104nMAssay Description:Biochemical compound potencies were assessed by evaluating inhibition of SOS1-mediated nucleotide exchange in KRAS G12D. The SOS1-promoted exchange o...More data for this Ligand-Target Pair
Affinity DataIC50: 0.110nMAssay Description:Biochemical compound potencies were assessed by evaluating inhibition of SOS1-mediated nucleotide exchange in KRAS G12D. The SOS1-promoted exchange o...More data for this Ligand-Target Pair
Affinity DataIC50: 0.110nMAssay Description:Biochemical compound potencies were assessed by evaluating inhibition of SOS1-mediated nucleotide exchange in KRAS G12D. The SOS1-promoted exchange o...More data for this Ligand-Target Pair
Affinity DataIC50: 0.110nMAssay Description:Biochemical compound potencies were assessed by evaluating inhibition of SOS1-mediated nucleotide exchange in KRAS G12D. The SOS1-promoted exchange o...More data for this Ligand-Target Pair
Affinity DataIC50: 0.110nMAssay Description:Biochemical compound potencies were assessed by evaluating inhibition of SOS1-mediated nucleotide exchange in KRAS G12D. The SOS1-promoted exchange o...More data for this Ligand-Target Pair
Affinity DataIC50: 0.114nMAssay Description:Biochemical compound potencies were assessed by evaluating inhibition of SOS1-mediated nucleotide exchange in KRAS G12D. The SOS1-promoted exchange o...More data for this Ligand-Target Pair
Affinity DataIC50: 0.114nMAssay Description:Biochemical compound potencies were assessed by evaluating inhibition of SOS1-mediated nucleotide exchange in KRAS G12D. The SOS1-promoted exchange o...More data for this Ligand-Target Pair
Affinity DataIC50: 0.120nMAssay Description:Biochemical compound potencies were assessed by evaluating inhibition of SOS1-mediated nucleotide exchange in KRAS G12D. The SOS1-promoted exchange o...More data for this Ligand-Target Pair
Affinity DataIC50: 0.120nMAssay Description:Biochemical compound potencies were assessed by evaluating inhibition of SOS1-mediated nucleotide exchange in KRAS G12D. The SOS1-promoted exchange o...More data for this Ligand-Target Pair
Affinity DataIC50: 0.120nMAssay Description:Biochemical compound potencies were assessed by evaluating inhibition of SOS1-mediated nucleotide exchange in KRAS G12D. The SOS1-promoted exchange o...More data for this Ligand-Target Pair
Affinity DataIC50: 0.120nMAssay Description:Biochemical compound potencies were assessed by evaluating inhibition of SOS1-mediated nucleotide exchange in KRAS G12D. The SOS1-promoted exchange o...More data for this Ligand-Target Pair
Affinity DataIC50: 0.130nMAssay Description:Biochemical compound potencies were assessed by evaluating inhibition of SOS1-mediated nucleotide exchange in KRAS G12D. The SOS1-promoted exchange o...More data for this Ligand-Target Pair
Affinity DataIC50: 0.130nMAssay Description:Biochemical compound potencies were assessed by evaluating inhibition of SOS1-mediated nucleotide exchange in KRAS G12D. The SOS1-promoted exchange o...More data for this Ligand-Target Pair
Affinity DataIC50: 0.133nMAssay Description:Biochemical compound potencies were assessed by evaluating inhibition of SOS1-mediated nucleotide exchange in KRAS G12D. The SOS1-promoted exchange o...More data for this Ligand-Target Pair
Affinity DataIC50: 0.140nMAssay Description:Biochemical compound potencies were assessed by evaluating inhibition of SOS1-mediated nucleotide exchange in KRAS G12D. The SOS1-promoted exchange o...More data for this Ligand-Target Pair
Affinity DataIC50: 0.140nMAssay Description:Biochemical compound potencies were assessed by evaluating inhibition of SOS1-mediated nucleotide exchange in KRAS G12D. The SOS1-promoted exchange o...More data for this Ligand-Target Pair
Affinity DataIC50: 0.150nMAssay Description:Biochemical compound potencies were assessed by evaluating inhibition of SOS1-mediated nucleotide exchange in KRAS G12D. The SOS1-promoted exchange o...More data for this Ligand-Target Pair
Affinity DataIC50: 0.159nMAssay Description:Biochemical compound potencies were assessed by evaluating inhibition of SOS1-mediated nucleotide exchange in KRAS G12D. The SOS1-promoted exchange o...More data for this Ligand-Target Pair
Affinity DataIC50: 0.160nMAssay Description:Biochemical compound potencies were assessed by evaluating inhibition of SOS1-mediated nucleotide exchange in KRAS G12D. The SOS1-promoted exchange o...More data for this Ligand-Target Pair
Affinity DataIC50: 0.160nMAssay Description:Biochemical compound potencies were assessed by evaluating inhibition of SOS1-mediated nucleotide exchange in KRAS G12D. The SOS1-promoted exchange o...More data for this Ligand-Target Pair
Affinity DataIC50: 0.160nMAssay Description:Biochemical compound potencies were assessed by evaluating inhibition of SOS1-mediated nucleotide exchange in KRAS G12D. The SOS1-promoted exchange o...More data for this Ligand-Target Pair
Affinity DataIC50: 0.170nMAssay Description:Biochemical compound potencies were assessed by evaluating inhibition of SOS1-mediated nucleotide exchange in KRAS G12D. The SOS1-promoted exchange o...More data for this Ligand-Target Pair
Affinity DataIC50: 0.170nMAssay Description:Biochemical compound potencies were assessed by evaluating inhibition of SOS1-mediated nucleotide exchange in KRAS G12D. The SOS1-promoted exchange o...More data for this Ligand-Target Pair
Affinity DataIC50: 0.170nMAssay Description:Biochemical compound potencies were assessed by evaluating inhibition of SOS1-mediated nucleotide exchange in KRAS G12D. The SOS1-promoted exchange o...More data for this Ligand-Target Pair
Affinity DataIC50: 0.175nMAssay Description:Biochemical compound potencies were assessed by evaluating inhibition of SOS1-mediated nucleotide exchange in KRAS G12D. The SOS1-promoted exchange o...More data for this Ligand-Target Pair